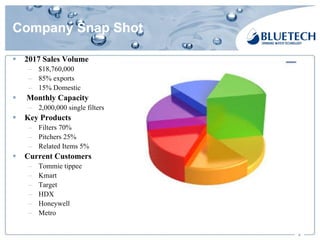
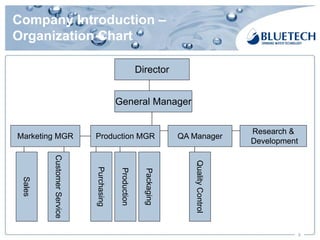
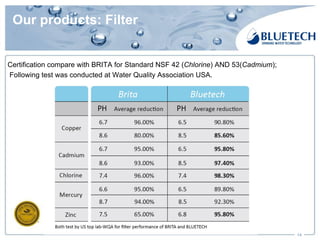
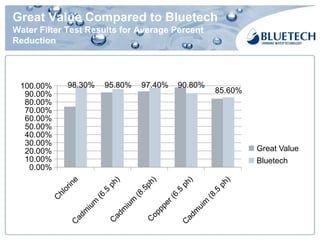
Bluetech, established in 2003 and based in Shanghai, specializes in manufacturing water filtration products, including filters and pitchers, and maintains a strong focus on quality standards, surpassing US and European regulations. The company has a monthly capacity of 2 million single filters and achieved 2017 sales of $18.76 million, primarily from exports. Bluetech is recognized for its innovative design and the ability to meet unique customer requirements, supported by a comprehensive quality control system and various international certifications.