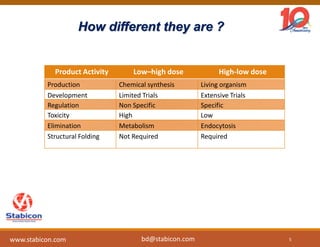
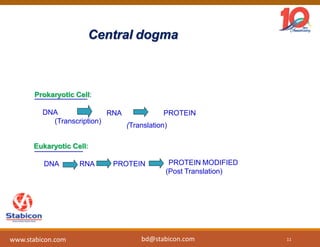
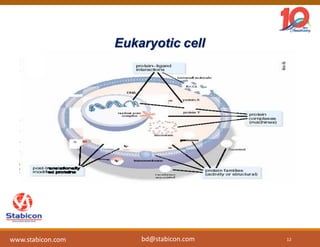
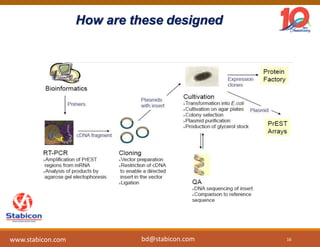
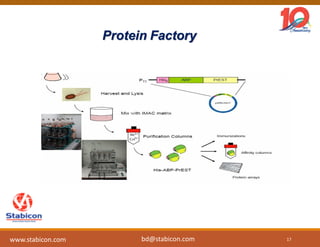
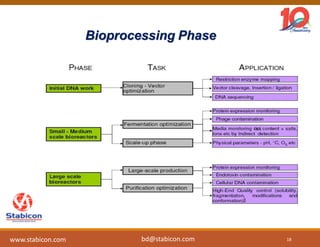
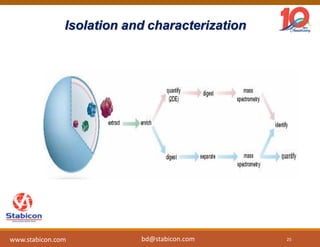
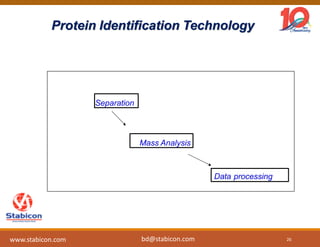
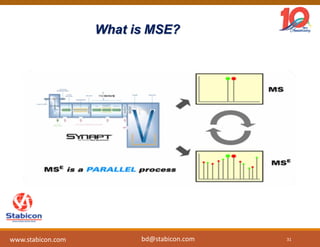
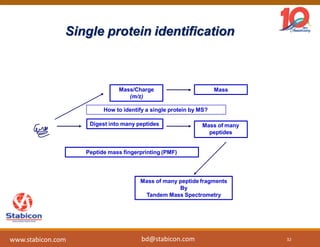
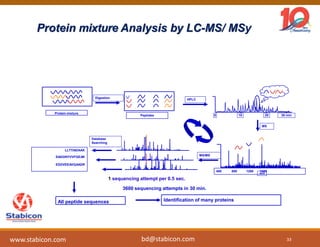
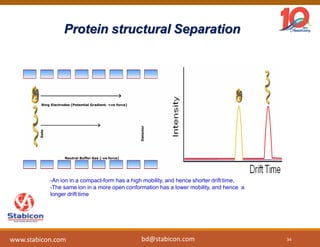
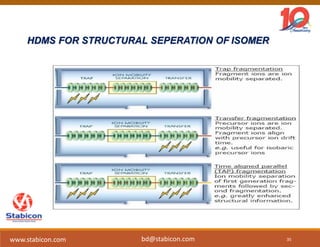
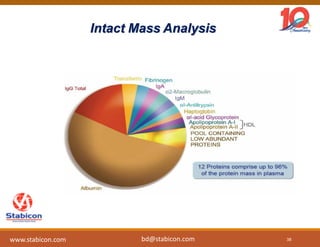
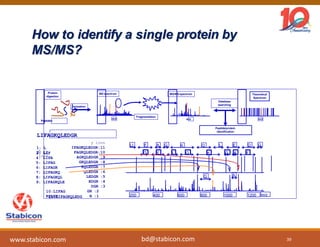
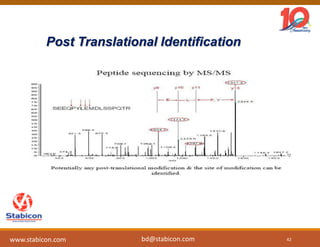
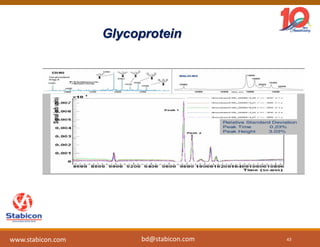
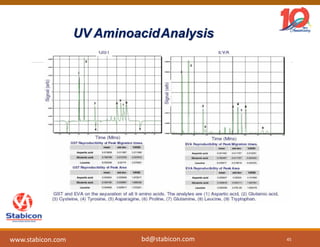
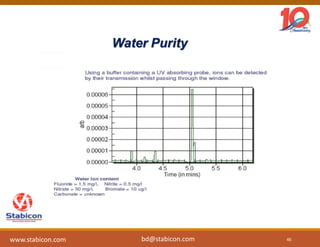
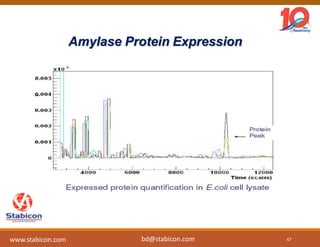
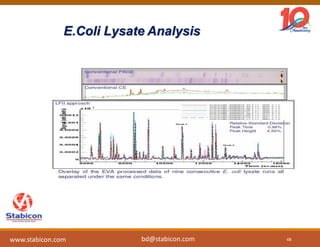
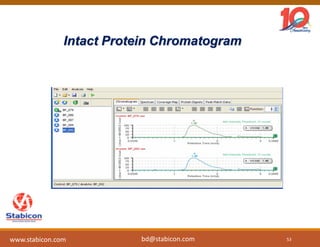
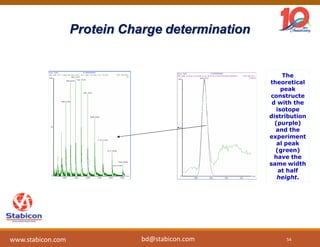
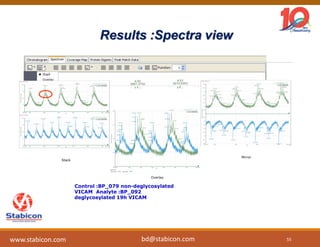
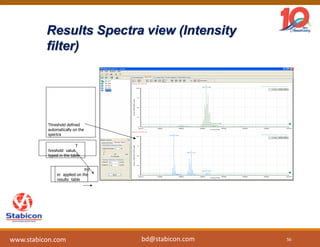
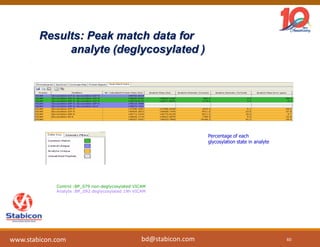
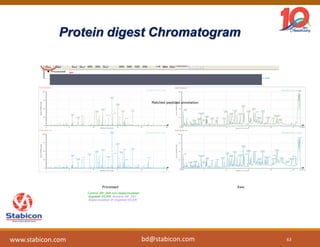
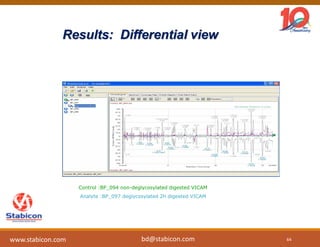
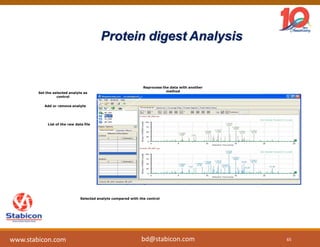
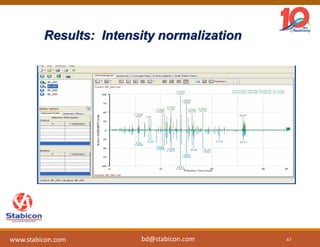
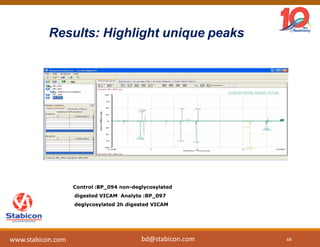
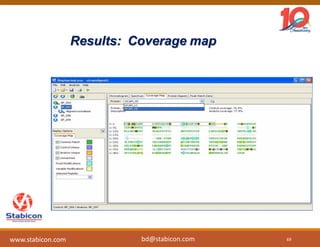
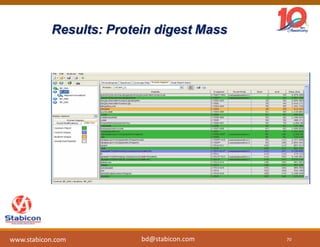
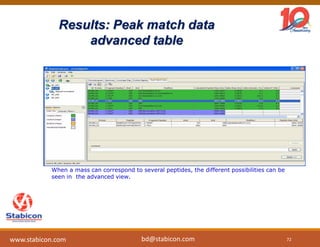
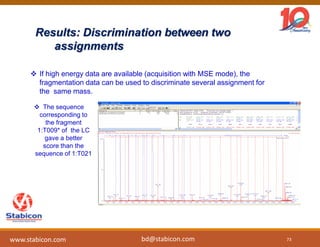
This document discusses the significance of biologics in therapeutics compared to traditional small molecules, detailing their production, characterization, and monitoring processes. It covers various chapters, including protein mechanisms, therapeutic protein production, and data processing methodologies in biopharmaceuticals. Key aspects include the complexity of protein structures, techniques for protein characterization, and the importance of post-translational modifications.