Bio based solvent KAT parameters
•Download as PPTX, PDF•
0 likes•105 views
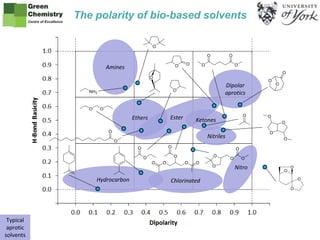
Polarity maps of bio-based solvents using Kamlet Abboud Taft solvatochromic parameters. Produced by the Green Chemistry Centre of Excellence at the University of York UK.
Report
Share
Report
Share
Recommended
More Related Content
Similar to Bio based solvent KAT parameters
Similar to Bio based solvent KAT parameters (20)
Lesson 6 Unit C - Carboxylic Acids and Esters (1).pptx

Lesson 6 Unit C - Carboxylic Acids and Esters (1).pptx
4-chem_109_alcohols_phenols_and_ethers_modified.ppt

4-chem_109_alcohols_phenols_and_ethers_modified.ppt
More from James Sherwood
More from James Sherwood (11)
Standards and regulations for a bio-based economy and a circular economy in E...

Standards and regulations for a bio-based economy and a circular economy in E...
Sustainable Solvent Selection Service (for bio-based solvents)

Sustainable Solvent Selection Service (for bio-based solvents)
Recirculation: A new concept to drive innovation in green product design

Recirculation: A new concept to drive innovation in green product design
Understanding the renewability of bio based products (james sherwood)

Understanding the renewability of bio based products (james sherwood)
Bio based products 2/2: Feedstocks and formulation, certification workshop [a...

Bio based products 2/2: Feedstocks and formulation, certification workshop [a...
Bio based products 1/2: Feedstocks and formulation, certification workshop [p...

Bio based products 1/2: Feedstocks and formulation, certification workshop [p...
Recently uploaded
THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptx

THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptxNandakishor Bhaurao Deshmukh
Bentham & Hooker's Classification. along with the merits and demerits of the ...

Bentham & Hooker's Classification. along with the merits and demerits of the ...Nistarini College, Purulia (W.B) India
Hot Sexy call girls in Moti Nagar,🔝 9953056974 🔝 escort Service

Hot Sexy call girls in Moti Nagar,🔝 9953056974 🔝 escort Service9953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptx

THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptx
Bentham & Hooker's Classification. along with the merits and demerits of the ...

Bentham & Hooker's Classification. along with the merits and demerits of the ...
Pests of jatropha_Bionomics_identification_Dr.UPR.pdf

Pests of jatropha_Bionomics_identification_Dr.UPR.pdf
Pests of safflower_Binomics_Identification_Dr.UPR.pdf

Pests of safflower_Binomics_Identification_Dr.UPR.pdf
Environmental Biotechnology Topic:- Microbial Biosensor

Environmental Biotechnology Topic:- Microbial Biosensor
Behavioral Disorder: Schizophrenia & it's Case Study.pdf

Behavioral Disorder: Schizophrenia & it's Case Study.pdf
GenBio2 - Lesson 1 - Introduction to Genetics.pptx

GenBio2 - Lesson 1 - Introduction to Genetics.pptx
OECD bibliometric indicators: Selected highlights, April 2024

OECD bibliometric indicators: Selected highlights, April 2024
Call Girls in Mayapuri Delhi 💯Call Us 🔝9953322196🔝 💯Escort.

Call Girls in Mayapuri Delhi 💯Call Us 🔝9953322196🔝 💯Escort.
The dark energy paradox leads to a new structure of spacetime.pptx

The dark energy paradox leads to a new structure of spacetime.pptx
Best Call Girls In Sector 29 Gurgaon❤️8860477959 EscorTs Service In 24/7 Delh...

Best Call Girls In Sector 29 Gurgaon❤️8860477959 EscorTs Service In 24/7 Delh...
Hot Sexy call girls in Moti Nagar,🔝 9953056974 🔝 escort Service

Hot Sexy call girls in Moti Nagar,🔝 9953056974 🔝 escort Service
Pests of castor_Binomics_Identification_Dr.UPR.pdf

Pests of castor_Binomics_Identification_Dr.UPR.pdf
Recombinant DNA technology( Transgenic plant and animal)

Recombinant DNA technology( Transgenic plant and animal)
Bio based solvent KAT parameters
- 1. Typical aprotic solvents Amines Dipolar aprotics ChlorinatedHydrocarbon Nitro Ethers Ester Ketones Nitriles The polarity of bio-based solvents
- 2. Ionic liquids Acids Alcohols Polyols Typical protic solvents The polarity of bio-based solvents
- 3. The Kamlet-Abboud-Taft solvatochromic parameters are measurements of solvent polarity. Dipolarity (π*) and hydrogen bond accepting ability (basicity, β) can be drawn on axes to create a polarity map. Instead of creating a three-dimensional plot with hydrogen bond donating ability (acidity, α) included, protic and aprotic solvents are separated onto separate charts for clarity. There are many protic solvents derived from biomass. Alcohols and carboxylic acids generally have only minor hazards. On the other hand, aprotic solvents can present significant hazards and are not straightforward to replace. Bio-based aprotic solvents tend to have unfamiliar structures in an attempt to convert bio-based chemicals into useful solvents without the same hazardous functionality.
