文献紹介20180525
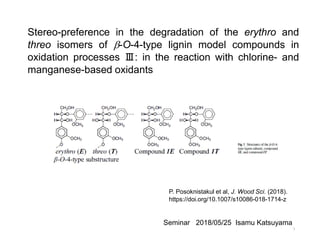
- 1. Stereo-preference in the degradation of the erythro and threo isomers of b-O-4-type lignin model compounds in oxidation processes Ⅲ: in the reaction with chlorine- and manganese-based oxidants P. Posoknistakul et al, J. Wood Sci. (2018). https://doi.org/10.1007/s10086-018-1714-z Seminar 2018/05/25 Isamu Katsuyama 1
- 2. Main points of this paper 1. Stereochemistry of b-O-4-type compounds The erythro and threo isomers 2. Chemical properties of some oxidants NaClO, NaClO2, MnO2, KMnO4 3. Degradation of each isomers in the presence of the oxidants Question: Is the reactivity same or different? 2
- 3. 3 1. Stereochemistry; erythro and threo isomers A B Erythro isomer
- 5. 2. Chemical properties of some oxidants NaClO A strong oxidant typically for oxidation of alcohols It is soluble in water. 5 NaClO H2O ClOHClO H HClO NaOH++ + (pka = 7.58) ClO ClO ClO pH = pka, [ClO - ] / [HClO] = 1 pH > pka, [ClO - ] / [HClO] > 1 pH < pka, [ClO- ] / [HClO] < 1 pH in solution 13.3 6.3 1.0 HClO < < HClO > HClO >>
- 6. NaClO2 A oxidant typically for oxidation of aldehydes, alcohols, etc. It is soluble in water. 6 NaClO2 ClO2 H2O HClO2 NaOH HClO2 H ++ + (pka = 1.94) ClO2 ClO2 ClO2 HClO2 < < HClO2 < HClO2 > pH in solution 13.3 6.3 1.0 pH = pka, [ClO2 - ] / [HClO2] = 1 pH > pka, [ClO2 - ] / [HClO2] > 1 pH < pka, [ClO2 - ] / [HClO2] < 1
- 7. 7 MnO2 • A mild oxidant typically for oxidation of allylic and benzylic alcohols, etc. → It is very useful for selective oxidation. • It is not soluble in organic solvent and water.
- 8. 8 KMnO4 • A powerful oxidant typically for oxidation of alcohols, aldehydes, alkenes, etc. • It is soluble in MeOH and water. • It is not soluble in most organic solvent.
- 9. 3. Degradation of isomers in the presence of the oxidants Example of previous work 9 CAN or Lignin peroxidase Fenton’s reagent No stereo-preference Stereo-preference in degradation of compound 1T C. Bohlin et al, J. Mol Catal B Enzym, 45, 21 (2007). C. Bohlin et al, ibid., 35, 100 (2005).
- 10. 10 This work Oxidants = NaClO, NaClO2, MnO2, KMnO4 Is stereo-preference in degradation of 1E and 1T observed?
- 11. ➀ Oxidation in hypochlorite systems (NaClO) (a) alkaline pH in solution (ClO-) Low reactivity No clear difference (b) neutral pH in solution (HClO) No clear difference ← Main reactive site may be aromatic ring; side chain was not affected. 11 pH 13.3 at 70 ℃ pH 6.3 at 70 ℃
- 12. ② Oxidation in chlorite systems (NaClO2) (a) alkaline pH in solution (ClO2 -) Low reactivity No clear difference (b) neutral pH in solution (ClO2 . generated from ClO2 -) No clear difference ←Main reactive site may be aromatic ring; side chain was not affected. (c) acidic pH in solution (HClO2) Degradation of compound 1T was slightly greater. 12 pH 13.3 at 70 ℃ pH 6.3 at 70 ℃ pH 1.0 at 70 ℃
- 13. 13 Reaction of alkenes with HClO, ClO2 .
- 14. ③ Oxidation in a manganese dioxide system (MnO2) acidic pH in solution Degradation of compound 1T was clearly more rapid than that of compound 1E. → It is presumed that MnO2 aggregates can approach 1T more easily than 1E. 14 pH 1.0 at room temp.
- 15. ④ Oxidation in permanganate systems (KMnO4) (a) acidic pH in solution Degradation of compound 1T was clearly grater than that of compound 1E. →Large size of MnO4 - may be related to the T preference. (b) neutral pH in solution Degradation of 1T was also grater than that of 1E. But, reactivity was not great. 15 pH 2.0 at room temp. pH 6.3 at room temp.
- 16. Conclusions Oxidation in hypochlorite systems (NaClO) and chlorite systems (NaClO2) No clear stereo-preference was observed. Oxidation in a manganese dioxide system (MnO2) and permanganate systems (KMnO4) A clear 1T stereo-preference was observed. 16
Editor's Notes
- In H2O Fenton reagent Fe2+ + H2O2 → HO・ + OH- + Fe3+ pH 4.0 at room temp.
- General rule is not clear. Stereo-preference may be observed under mild condition, and in the presence of large size of species (from oxidants)
