The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA Immunotherapy, and Unique Bispecific Platforms
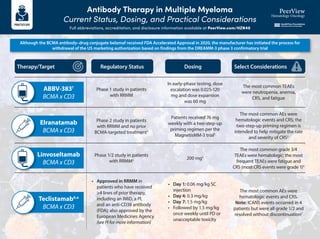
- 1. Antibody Therapy in Multiple Myeloma Current Status, Dosing, and Practical Considerations Full abbreviations, accreditation, and disclosure information available at PeerView.com/HZN40 Regulatory Status Dosing Phase 1 study in patients with RRMM Phase 2 study in patients with RRMM and no prior BCMA-targeted treatment2 • Approved in RRMM in patients who have received ≥4 lines of prior therapy, including an IMiD, a PI, and an anti-CD38 antibody (FDA); also approved by the European Medicines Agency (see PI for more information) • Day 1: 0.06 mg/kg SC injection • Day 4: 0.3 mg/kg • Day 7: 1.5 mg/kg • Followed by 1.5 mg/kg once weekly until PD or unacceptable toxicity Phase 1/2 study in patients with RRMM4 200 mg5 Patients received 76 mg weekly with a two-step-up priming regimen per the MagnetisMM-3 trial3 The most common AEs were hematologic events and CRS; the two-step-up priming regimen is intended to help mitigate the rate and severity of CRS3 The most common grade 3/4 TEAEs were hematologic; the most frequent TEAEs were fatigue and CRS (most CRS events were grade 1)4 The most common AEs were hematologic events and CRS. Note: ICANS events occurred in 4 patients but were all grade 1/2 and resolved without discontinuation7 In early-phase testing, dose escalation was 0.025-120 mg and dose expansion was 60 mg The most common TEAEs were neutropenia, anemia, CRS, and fatigue Select Considerations Therapy/Target Elranatamab BCMA x CD3 Teclistamab6,a BCMA x CD3 ABBV-3831 BCMA x CD3 Linvoseltamab BCMA x CD3 Although the BCMA antibody–drug conjugate belamaf received FDA Accelerated Approval in 2020, the manufacturer has initiated the process for withdrawal of the US marketing authorization based on findings from the DREAMM-3 phase 3 confirmatory trial
- 2. Antibody Therapy in Multiple Myeloma Current Status, Dosing, and Practical Considerations Full abbreviations, accreditation, and disclosure information available at PeerView.com/HZN40 • Approved in NDMM in combination with Rd and VMP in patients who are transplant ineligible and VTd in patients who are transplant eligible • Approved in RRMM in combination with Rd, Vd, and pom/dex; as monotherapy for more heavily pretreated patients; and in combination with carfilzomib and dex for patients who have received 1 to 3 prior lines of therapy (see PI for more information) • Approved in NDMM in combination with Rd in patients who are transplant ineligible, in combination with VMP in patients who are transplant ineligible, and in combination with VTd in patients who are transplant eligible • Approved in RRMM in combination with Rd, Vd, Kd, and pom/dex, and as monotherapy for more heavily pretreated patients (see PI for more information) 1,800 mg daratumumab and 30,000 units hyaluronidase SC in the abdomen over ~3 to 5 min 16 mg/kg IV actual body weight • Monitor for - infusion-related reactions - neutropenia - thrombocytopenia • Inform blood banks • Monitor for hypersensitivity reactions; screen for hepatitis B core antibody and surface antigen prior to initiation and initiate prophylaxis if needed • Monitor for - neutropenia - thrombocytopenia • Inform blood banks • Monitor for hypersensitivity reactions; screen for hepatitis B core antibody and surface antigen prior to initiation and initiate prophylaxis if needed Select Considerations Daratumumab- hyaluronidase– fihj9 CD38 Daratumumab8 CD38 Therapy/Target Dosing Regulatory Status
- 3. Antibody Therapy in Multiple Myeloma Current Status, Dosing, and Practical Considerations Full abbreviations, accreditation, and disclosure information available at PeerView.com/HZN40 a Currently available under accelerated approval. 1. D'Souza A et al. J Clin Oncol. 2022;40:3576-3586. 2. https://clinicaltrials.gov/ct2/show/NCT04649359. 3. Lesokhin A et al. ASCO 2022. Abstract 8006. 4. Zonder JA et al. EHA 2022. Abstract S189. 5. Bumma N et al. ASH 2022. Abstract 4555. 6. Tecvayli (teclistamab-cqyv) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/761291s000lbl.pdf. 7. Moreau P et al. ASH 2021. Abstract 896. 8. Darzalex (daratumumab) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/761036s033lbl.pdf. 9. Faspro (daratumumab-hyaluronidase–fihj) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/761145s000lbl.pdf. 10. Sarclisa (isatuximab-irfc) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/761113s000lbl.pdf. 11. Empliciti (elotuzumab) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/761035s008lbl.pdf. 12. Wong SW et al. ASH 2022. Abstract 162. • Approved in RRMM in combination with Rd and pom/ dex (see PI for more information) • Approved in RRMM in combination with pom/dex after ≥2 prior therapies, including lenalidomide and a PI, and in combination with carfilzomib and dex for patients who have received 1 to 3 prior lines of therapy (see PI for more information) • With Rd: 10 mg/kg IV every wk for 2 cycles and every 2 wk thereafter • With pom/dex: 10 mg/kg IV every wk for 2 cycles and 20 mg/kg every 4 wk thereafter 10 mg/kg IV every wk for 4 wk and every 2 wk thereafter • Monitor for - infusion-related reactions - neutropenia - second primary malignancies - M protein (can affect determination of CR) • Monitor CBC periodically during treatment, and monitor patients with neutropenia for signs of infection • Premedication required for infusion-related reactions • Monitor for - fever and infection - second primary malignancies - hepatotoxicity - M protein (can affect determination of CR) Select Considerations Isatuximab10 CD38 Elotuzumab11 SLAMF7 Other bispecific BCMA and non-BCMA agents are also in rapid development, including TNB383b, talquetamab, cevostamab, and HPN 217 Other new developments: Alnuctamab is an emerging BCMA x CD3 antibody that has demonstrated clinical activity in patients with RRMM who have been treated with ≥3 prior lines of therapy12 Therapy/Target Dosing Regulatory Status
- 4. CAR-T Cell Therapy in Multiple Myeloma Current Status, Dosing, and Practical Considerations Full abbreviations, accreditation, and disclosure information available at PeerView.com/HZN40 1. Carvykti (ciltacabtagene autoleucel) Prescribing Information. https://www.fda.gov/media/156560/download. 2. Abecma (idecabtagene vicleucel) Prescribing Information. https://www.fda.gov/media/147055/download. REMS Cilta-Cel1 Approved in RRMM after ≥4 prior therapies, including an anti-CD38 mAb, a proteasome inhibitor, and an IMiD Recommended Dose Range 0.5-1.0 × 106 CAR-positive viable T cells (maximum dose of 1.0 × 108 CAR-positive viable T cells per single-dose infusion) Recommended Dose Range 300-460 × 106 CAR-positive viable T cells Ide-Cel2 Approved in RRMM after ≥4 prior therapies, including an anti-CD38 mAb, a proteasome inhibitor, and an IMiD General Principles for CAR-T Therapy Referral to a certified healthcare facility is required for collection of patient’s cells and administration of CAR-T therapy Avoid prophylactic use of dexamethasone or other systemic corticosteroids Premedicate with acetaminophen and an H1 antihistamine Monitor for CRS and ICANS and confirm tocilizumab availability before infusion Ide-cel and cilta-cel are available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) Monitor for neurologic events, hemophagocytic lymphohistiocytosis/ macrophage activation syndrome, and cytopenias Administer a lymphodepleting chemotherapy regimen of cyclophosphamide and fludarabine before CAR-T infusion Do not use a leukodepleting filter when administering
- 5. Resources for Encouraging Patient Engagement in Care From HealthTree Foundation for Multiple Myeloma Full abbreviations, accreditation, and disclosure information available at PeerView.com/HZN40 HealthTree Foundation for Multiple Myeloma offers a wide range of resources that can be used to encourage patients to learn more about their disease or a planned course of treatment Patient education and navigation tools Programs that create patient connections and build community A living, real-world evidence data portal that delivers researchers the data they need to accelerate a cure Scan to visit: healthtree.org/myeloma Scan to visit: healthtree.org/myeloma/coach Free one-on-one help from experienced patients and caregivers Help patients get answers to basic questions about myeloma Scan to visit: healthtree.org/myeloma/university Selected Resources
