Novel Induction Options in AML: Assessing the Implications for HCT-Eligible Patients
•
1 like•94 views
Jorge E. Cortes, MD, and Stephen A. Strickland, MD, MSCI, prepared useful practice aids pertaining to acute myeloid leukemia for this CME activity titled "Novel Induction Options in AML: Assessing the Implications for HCT-Eligible Patients." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2u4yk5p. CME credit will be available until April 3, 2019.
Report
Share
Report
Share
Download to read offline
Recommended
Alexander Perl, MD, and James M. Foran, MD, FRCPC, prepared useful practice aids pertaining to leukemia for this CME activity titled "Integrating Innovative Therapeutics With Allogeneic HCT in AML: Insights and Evidence From Induction to Maintenance." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2EZE2I6. CME credit will be available until March 28, 2020.Integrating Innovative Therapeutics With Allogeneic HCT in AML: Insights and ...

Integrating Innovative Therapeutics With Allogeneic HCT in AML: Insights and ...PVI, PeerView Institute for Medical Education
Harry P. Erba, MD, PhD, and James M. Foran, MD, FRCPC, prepared useful Practice Aids pertaining to acute myeloid leukemia for this CME activity titled "The Continuing Wave of Innovation in AML: Getting the Most From the Convergence of Novel Therapy and Allogeneic Transplant." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2vdoO1j. CME credit will be available until March 12, 2021.The Continuing Wave of Innovation in AML: Getting the Most From the Convergen...

The Continuing Wave of Innovation in AML: Getting the Most From the Convergen...PVI, PeerView Institute for Medical Education
Harry P. Erba, MD, PhD, Naval Daver, MD, Courtney D. DiNardo, MD, MSCE, and Gail J. Roboz, MD prepared useful Practice Aids pertaining to acute myeloid leukemia for this CME/MOC activity titled Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Diverse Patient Populations. For the full presentation, complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3fsa7Jh. CME/MOC credit will be available until July 9, 2021.Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Div...

Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Div...PVI, PeerView Institute for Medical Education
Recommended
Alexander Perl, MD, and James M. Foran, MD, FRCPC, prepared useful practice aids pertaining to leukemia for this CME activity titled "Integrating Innovative Therapeutics With Allogeneic HCT in AML: Insights and Evidence From Induction to Maintenance." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2EZE2I6. CME credit will be available until March 28, 2020.Integrating Innovative Therapeutics With Allogeneic HCT in AML: Insights and ...

Integrating Innovative Therapeutics With Allogeneic HCT in AML: Insights and ...PVI, PeerView Institute for Medical Education
Harry P. Erba, MD, PhD, and James M. Foran, MD, FRCPC, prepared useful Practice Aids pertaining to acute myeloid leukemia for this CME activity titled "The Continuing Wave of Innovation in AML: Getting the Most From the Convergence of Novel Therapy and Allogeneic Transplant." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2vdoO1j. CME credit will be available until March 12, 2021.The Continuing Wave of Innovation in AML: Getting the Most From the Convergen...

The Continuing Wave of Innovation in AML: Getting the Most From the Convergen...PVI, PeerView Institute for Medical Education
Harry P. Erba, MD, PhD, Naval Daver, MD, Courtney D. DiNardo, MD, MSCE, and Gail J. Roboz, MD prepared useful Practice Aids pertaining to acute myeloid leukemia for this CME/MOC activity titled Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Diverse Patient Populations. For the full presentation, complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3fsa7Jh. CME/MOC credit will be available until July 9, 2021.Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Div...

Transforming Modern Care in AML: Clinical Solutions With Novel Agents for Div...PVI, PeerView Institute for Medical Education
Sagar Lonial, MD, FACP, Shaji Kumar, MD, Robert Z. Orlowski, MD, PhD, and Nina Shah, MD, prepared useful practice aids pertaining to multiple myeloma for this CME/MOC activity titled "How I Think, How I Treat: Understanding Innovation in Multiple Myeloma—New Models for Attacking Disease Across the Clinical Spectrum". For the full presentation, complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/30sAs5I. CME/MOC credit will be available until July 16, 2021.How I Think, How I Treat: Understanding Innovation in Multiple Myeloma—New Mo...

How I Think, How I Treat: Understanding Innovation in Multiple Myeloma—New Mo...PVI, PeerView Institute for Medical Education
Robert Z. Orlowski, MD, PhD, prepared useful practice aids pertaining to multiple myeloma management for this CME/CNE activity titled "Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-Generation Therapeutics." For the full presentation, monograph, complete CME/CNE information, and to apply for credit, please visit us at http://bit.ly/2SPKaZZ. CME/CNE credit will be available until August 7, 2020.Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-G...

Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-G...PVI, PeerView Institute for Medical Education
Despite remarkable progress in the treatment of breast cancer in recent years, management of estrogen receptor (ER)-positive disease remains a challenge. Through didactic and case presentations, expert faculty will address clinical debates regarding optimal treatment selection and the incorporation of new classes of targeted therapies into practice to improve outcomes and overcome resistance in the management of ER-positive breast cancer
Target Audience
This activity has been designed to meet the educational needs of medical oncologists and other healthcare providers who are involved in the care and treatment of patients with advanced estrogen receptor (ER)-positive breast cancer.
Purpose
The goal is to optimize the clinical decision-making of clinicians involved in the treatment of advanced ER-positive breast cancer by providing updates on emerging data.
Slide Deck Disclaimer
This slide deck in its original and unaltered format is for educational purposes and is current as of August 2012. All materials contained herein reflect the views of the faculty, and not those of IMER, the CME provider, or the commercial supporter. These materials may discuss therapeutic products that have not been approved by the US Food and Drug Administration and off-label uses of approved products. Readers should not rely on this information as a substitute for professional medical advice, diagnosis, or treatment. The use of any information provided is solely at your own risk, and readers should verify the prescribing information and all data before treating patients or employing any therapeutic products described in this educational activity.
Usage Rights
This slide deck is provided for educational purposes and individual slides may be used for personal, non-commercial presentations only if the content and references remain unchanged. No part of this slide deck may be published in print or electronically as a promotional or certified educational activity without prior written permission from IMER. Additional terms may apply.
More information:
http://imeronline.com/864dsd
Community Oncology Clinical Debates in Breast Cancer: Advanced ER-Positive Di...

Community Oncology Clinical Debates in Breast Cancer: Advanced ER-Positive Di...Institute For Medical Education and Research (IMER)
Steven Brem, MD, Manmeet Ahluwalia, MD, FACP, MBA, and Steven A. Toms, MD, MPH, FAANS, FACS, prepared useful Practice Aids pertaining to glioblastoma for this CME activity titled "Rethinking Glioblastoma Care in an Evolving Therapeutic Landscape: Expert Perspectives on Modern Modalities, Novel Agents, and Combinations." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at https://bit.ly/3n4CSAb. CME credit will be available until December 28, 2021.Rethinking Glioblastoma Care in an Evolving Therapeutic Landscape: Expert Per...

Rethinking Glioblastoma Care in an Evolving Therapeutic Landscape: Expert Per...PVI, PeerView Institute for Medical Education
More Related Content
What's hot
Sagar Lonial, MD, FACP, Shaji Kumar, MD, Robert Z. Orlowski, MD, PhD, and Nina Shah, MD, prepared useful practice aids pertaining to multiple myeloma for this CME/MOC activity titled "How I Think, How I Treat: Understanding Innovation in Multiple Myeloma—New Models for Attacking Disease Across the Clinical Spectrum". For the full presentation, complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/30sAs5I. CME/MOC credit will be available until July 16, 2021.How I Think, How I Treat: Understanding Innovation in Multiple Myeloma—New Mo...

How I Think, How I Treat: Understanding Innovation in Multiple Myeloma—New Mo...PVI, PeerView Institute for Medical Education
Robert Z. Orlowski, MD, PhD, prepared useful practice aids pertaining to multiple myeloma management for this CME/CNE activity titled "Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-Generation Therapeutics." For the full presentation, monograph, complete CME/CNE information, and to apply for credit, please visit us at http://bit.ly/2SPKaZZ. CME/CNE credit will be available until August 7, 2020.Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-G...

Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-G...PVI, PeerView Institute for Medical Education
Despite remarkable progress in the treatment of breast cancer in recent years, management of estrogen receptor (ER)-positive disease remains a challenge. Through didactic and case presentations, expert faculty will address clinical debates regarding optimal treatment selection and the incorporation of new classes of targeted therapies into practice to improve outcomes and overcome resistance in the management of ER-positive breast cancer
Target Audience
This activity has been designed to meet the educational needs of medical oncologists and other healthcare providers who are involved in the care and treatment of patients with advanced estrogen receptor (ER)-positive breast cancer.
Purpose
The goal is to optimize the clinical decision-making of clinicians involved in the treatment of advanced ER-positive breast cancer by providing updates on emerging data.
Slide Deck Disclaimer
This slide deck in its original and unaltered format is for educational purposes and is current as of August 2012. All materials contained herein reflect the views of the faculty, and not those of IMER, the CME provider, or the commercial supporter. These materials may discuss therapeutic products that have not been approved by the US Food and Drug Administration and off-label uses of approved products. Readers should not rely on this information as a substitute for professional medical advice, diagnosis, or treatment. The use of any information provided is solely at your own risk, and readers should verify the prescribing information and all data before treating patients or employing any therapeutic products described in this educational activity.
Usage Rights
This slide deck is provided for educational purposes and individual slides may be used for personal, non-commercial presentations only if the content and references remain unchanged. No part of this slide deck may be published in print or electronically as a promotional or certified educational activity without prior written permission from IMER. Additional terms may apply.
More information:
http://imeronline.com/864dsd
Community Oncology Clinical Debates in Breast Cancer: Advanced ER-Positive Di...

Community Oncology Clinical Debates in Breast Cancer: Advanced ER-Positive Di...Institute For Medical Education and Research (IMER)
Steven Brem, MD, Manmeet Ahluwalia, MD, FACP, MBA, and Steven A. Toms, MD, MPH, FAANS, FACS, prepared useful Practice Aids pertaining to glioblastoma for this CME activity titled "Rethinking Glioblastoma Care in an Evolving Therapeutic Landscape: Expert Perspectives on Modern Modalities, Novel Agents, and Combinations." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at https://bit.ly/3n4CSAb. CME credit will be available until December 28, 2021.Rethinking Glioblastoma Care in an Evolving Therapeutic Landscape: Expert Per...

Rethinking Glioblastoma Care in an Evolving Therapeutic Landscape: Expert Per...PVI, PeerView Institute for Medical Education
What's hot (20)
Enzalutamide in Metastatic Prostate Cancer Before Chemotherapy

Enzalutamide in Metastatic Prostate Cancer Before Chemotherapy
How I Think, How I Treat: Understanding Innovation in Multiple Myeloma—New Mo...

How I Think, How I Treat: Understanding Innovation in Multiple Myeloma—New Mo...
Treatment strategies in patients with statin intolerance

Treatment strategies in patients with statin intolerance
Optimal integration of new treatments for castration resistant prostate cancer 

Optimal integration of new treatments for castration resistant prostate cancer
L. berarducci new cholesterol management guidelines

L. berarducci new cholesterol management guidelines
Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-G...

Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-G...
Renard_et_al-2015-British_Journal_of_Clinical_Pharmacology

Renard_et_al-2015-British_Journal_of_Clinical_Pharmacology
Community Oncology Clinical Debates in Breast Cancer: Advanced ER-Positive Di...

Community Oncology Clinical Debates in Breast Cancer: Advanced ER-Positive Di...
Rethinking Glioblastoma Care in an Evolving Therapeutic Landscape: Expert Per...

Rethinking Glioblastoma Care in an Evolving Therapeutic Landscape: Expert Per...
MCO 2011 - Slide 4 - K. Fizazi - Castration-refractory prostate cancer

MCO 2011 - Slide 4 - K. Fizazi - Castration-refractory prostate cancer
Similar to Novel Induction Options in AML: Assessing the Implications for HCT-Eligible Patients
Triple-negative breast cancer is an area of very active research, with many exciting new agents and approaches in the pipeline.
What’s New in Biology, Treatment and Clinical Trials for Metastatic Triple-N...

What’s New in Biology, Treatment and Clinical Trials for Metastatic Triple-N...Dana-Farber Cancer Institute
Chair & Presenter, Robert Z. Orlowski, MD, PhD, Noa Biran, MD, and Ajay K. Nooka, MD, MPH, FACP, prepared useful Practice Aids pertaining to multiple myeloma for this CME/MOC/AAPA activity titled “The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA Immunotherapy, and Unique Bispecific Platforms.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA information, and to apply for credit, please visit us at https://bit.ly/3MPq140. CME/MOC/AAPA credit will be available until July 3, 2024.The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...

The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...PVI, PeerView Institute for Medical Education
John P. Leonard, MD, John C. Byrd, MD, Professor Dr. med Georg Lenz, and David G. Maloney, MD, PhD, discuss non-Hodgkin lymphoma management in this CME activity titled, "The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal Guide to Integrating New Therapy Into Patient Care." For the full presentation, downloadable Practice Aids, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2M7vUqT. CME credit will be available until July 4, 2019.The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal...

The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal...PVI, PeerView Institute for Medical Education
Chair & Presenter, Beth Faiman, PhD, MSN, APN-BC, AOCN, BMTCN, FAAN, FAPO, Donna D. Catamero, ANP-BC, OCN, CCRC, and Tiffany A. Richards, PhD, ANP-BC, AOCNP, prepared useful Practice Aids pertaining to multiple myeloma for this NCPD/ILNA activity titled “Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering Effective Care With BCMA Antibodies and Cellular Therapy.” For the full presentation, downloadable Practice Aids, and complete NCPD/ILNA information, and to apply for credit, please visit us at https://bit.ly/3ZSymre. NCPD/ILNA credit will be available until May 27, 2024.Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering...

Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering...PVI, PeerView Institute for Medical Education
Richard S. Finn, MD, Anthony El-Khoueiry, MD, and Josep M. Llovet, MD, PhD, prepared useful practice aids pertaining to hepatocellular carcinoma for this CME activity titled "Breaking the Paradox: Expanding Options and New Questions in HCC Management: Mapping the Pathways to Better Patient Outcomes." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2HU6L5K. CME credit will be available until February 14, 2020.Breaking the Paradox: Expanding Options and New Questions in HCC Management—M...

Breaking the Paradox: Expanding Options and New Questions in HCC Management—M...PVI, PeerView Institute for Medical Education
Naval Daver, MD, prepared useful practice aids pertaining to acute myeloid leukemia for this CME/CE activity titled "Groundbreaking Treatment Options for AML: How to Personalize Patient Care With New and Emerging Therapies." For the full presentation, monograph, complete CME/CE information, and to apply for credit, please visit us at http://bit.ly/2FCo09Y. CME/CE credit will be available until March 20, 2019.Groundbreaking Treatment Options for AML: How to Personalize Patient Care Wit...

Groundbreaking Treatment Options for AML: How to Personalize Patient Care Wit...PVI, PeerView Institute for Medical Education
Chair, Amit Singal, MD, MS, Anthony El-Khoueiry, MD, Ahmed Omar Kaseb, MD, CMQ, and Anjana Pillai, MD, prepared useful Practice Aids pertaining to hepatocellular carcinoma for this CME/MOC activity titled “It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collaboration & Modern Therapeutics.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3AbNtC5. CME/MOC credit will be available until December 4, 2023.
It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...

It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...PVI, PeerView Institute for Medical Education
Chair & Presenter, Kenneth R. Cooke, MD, Megan Burris, MSN, CPNP-PC/AC, and Megan Burris, MSN, CPNP-PC/AC, prepared useful Practice Aids pertaining to VOD/SOS for this CME/MOC/NCPD/CPE activity titled “Collaborative Strategies for Managing VOD/SOS: Interprofessional Insights on Advances in Diagnosis, Severity Grading, and Treatment.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/CPE information, and to apply for credit, please visit us at https://bit.ly/2TeQSga. CME/MOC/NCPD/CPE credit will be available until September 23, 2022.Collaborative Strategies for Managing VOD/SOS: Interprofessional Insights on ...

Collaborative Strategies for Managing VOD/SOS: Interprofessional Insights on ...PVI, PeerView Institute for Medical Education
Ghassan Abou-Alfa, MD, MBA, Anthony El-Khoueiry, MD, and R. Kate Kelley, MD, prepared useful Practice Aids pertaining to liver cancer for this CME/CE activity titled "Teaming Up to Improve Outcomes in Advanced Hepatocellular Carcinoma: A Tumor Board Evaluating the Potential of Immunotherapy and Novel Targeted Approaches." For the full presentation, monograph, complete CME/CE information, and to apply for credit, please visit us at http://bit.ly/2FG0J75. CME/CE credit will be available until March 25, 2019.Teaming Up to Improve Outcomes in Advanced Hepatocellular Carcinoma: A Tumor ...

Teaming Up to Improve Outcomes in Advanced Hepatocellular Carcinoma: A Tumor ...PVI, PeerView Institute for Medical Education
Similar to Novel Induction Options in AML: Assessing the Implications for HCT-Eligible Patients (20)
What’s New in Biology, Treatment and Clinical Trials for Metastatic Triple-N...

What’s New in Biology, Treatment and Clinical Trials for Metastatic Triple-N...
The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...

The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...
The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal...

The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal...
Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering...

Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering...
Breaking the Paradox: Expanding Options and New Questions in HCC Management—M...

Breaking the Paradox: Expanding Options and New Questions in HCC Management—M...
Groundbreaking Treatment Options for AML: How to Personalize Patient Care Wit...

Groundbreaking Treatment Options for AML: How to Personalize Patient Care Wit...
Shorter oral bedaquiline regimen 2022 NTEP guidelines

Shorter oral bedaquiline regimen 2022 NTEP guidelines
New Thinking, New Strategies in Advanced Urothelial Carcinoma

New Thinking, New Strategies in Advanced Urothelial Carcinoma
It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...

It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...
Collaborative Strategies for Managing VOD/SOS: Interprofessional Insights on ...

Collaborative Strategies for Managing VOD/SOS: Interprofessional Insights on ...
Teaming Up to Improve Outcomes in Advanced Hepatocellular Carcinoma: A Tumor ...

Teaming Up to Improve Outcomes in Advanced Hepatocellular Carcinoma: A Tumor ...
More from PVI, PeerView Institute for Medical Education
Chair A. Michael Lincoff, MD, discusses obesity in this CME activity titled “Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Weight Loss Pharmacotherapy.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/3KAO98K. CME credit will be available until April 25, 2025.Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...

Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...PVI, PeerView Institute for Medical Education
Co-Chairs and Planners Saakshi Khattri, MBBS, MD, FAAD, FACR, Marla Dubinsky, MD, Emma Guttman-Yassky, MD, PhD, and Alexis Ogdie, MD, MSCE, discuss immune-mediated inflammatory diseases in this CME/AAPA activity titled “Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Diseases: Addressing Shared Pathophysiology With JAK Inhibitors.” For the full presentation, downloadable Practice Aids, and complete CME/AAPA information, and to apply for credit, please visit us at https://bit.ly/3JhsIZ7. CME/AAPA credit will be available until April 24, 2025.Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...

Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...PVI, PeerView Institute for Medical Education
Co-Chairs Alicia K. Morgans, MD, MPH, and Neal D. Shore, MD, FACS, discuss prostate cancer in this CME/MOC/NCPD/CPE/AAPA/IPCE activity titled “Treatment Advances and Individualized Therapeutic Strategies in Prostate Cancer: Expert Insights on Key Evidence, Practical Tips for Personalized Therapy, and Clinical Integration Approaches.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/CPE/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SQrJ6G. CME/MOC/NCPD/CPE/AAPA/IPCE credit will be available until April 24, 2025.Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...

Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...PVI, PeerView Institute for Medical Education
Co-Chairs Prof. Nicolas Girard, MD, PhD, and Aaron Lisberg, MD, discuss NSCLC in this CME/MOC/NCPD/AAPA/IPCE activity titled “Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Cancer: Unveiling Potential, Shaping Tomorrow.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3RmX3dU. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 24, 2025.Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...

Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...PVI, PeerView Institute for Medical Education
Co-Chairs, Joseph K. Han, MD, and Seth J. Isaacs, MD, prepared useful Practice Aids pertaining to chronic rhinosinusitis with nasal polyps for this CME/MOC/CC/AAPA/IPCE activity titled “Biologics in CRSwNP: Putting a Paradigm Shift Into Practice.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3Tq6n1G. CME/MOC/CC/AAPA/IPCE credit will be available until May 6, 2025.Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into PracticePVI, PeerView Institute for Medical Education
Co-Chairs, Joseph K. Han, MD, and Seth J. Isaacs, MD, discuss chronic rhinosinusitis with nasal polyps in this CME/MOC/CC/AAPA/IPCE activity titled “Biologics in CRSwNP: Putting a Paradigm Shift Into Practice.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3Tq6n1G. CME/MOC/CC/AAPA/IPCE credit will be available until May 6, 2025.Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into PracticePVI, PeerView Institute for Medical Education
Co-Chairs, R. Donald Harvey, PharmD, BCOP, FCCP, FHOPA, FASCO, Zahra Mahmoudjafari, PharmD, MBA, BCOP, FHOPA, and James Davis, PharmD, BCOP, discuss multiple myeloma in this CME/CPE/IPCE activity titled “Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering Effective Therapy With Antibody Platforms.” For the full presentation, downloadable Practice Aids, and complete CME/CPE/IPCE information, and to apply for credit, please visit us at https://bit.ly/4aa0iMX. CME/CPE/IPCE credit will be available until May 2, 2025.Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...

Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...PVI, PeerView Institute for Medical Education
Co-Chairs, Carlos G. Romo, MD, and Aimee Sato, MD, discuss Neurofibromatosis in this CME/MOC activity titled “Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & Multimodal Care for NF1 pNF and Other Tumors.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3SZRz8p. CME/MOC credit will be available until May 2, 2025.Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...

Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...PVI, PeerView Institute for Medical Education
Chair and Presenters Kathleen N. Moore, MD, MS, Floor J. Backes, MD, and Bhavana Pothuri, MD, MS, prepared useful Practice Aids pertaining to endometrial cancer for this CME/MOC/NCPD/AAPA/IPCE activity titled “Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Potential of Immunotherapy, ADCs, PARP Inhibitors, and Other Emerging Treatment Strategies.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SjJyuH. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 17, 2025.Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...PVI, PeerView Institute for Medical Education
Chair and Presenters Kathleen N. Moore, MD, MS, Floor J. Backes, MD, and Bhavana Pothuri, MD, MS, discuss endometrial cancer in this CME/MOC/NCPD/AAPA/IPCE activity titled “Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Potential of Immunotherapy, ADCs, PARP Inhibitors, and Other Emerging Treatment Strategies.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SjJyuH. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 17, 2025.Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...PVI, PeerView Institute for Medical Education
Chair and Presenters Bradley J. Monk, MD, FACS, FACOG, Kathleen N. Moore, MD, MS, and Ana Oaknin, MD, PhD, discuss gynecologic cancers in this CME/MOC/NCPD/AAPA/IPCE activity titled “Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Implementation Strategies, and Patient Care.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/4a56tly. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 16, 2025.Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...

Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...PVI, PeerView Institute for Medical Education
Chair Lecia V. Sequist, MD, MPH, and Patrick Nana-Sinkam, MD, FCCP, prepared useful Practice Aids pertaining to lung cancer for this CME/MOC/AAPA/IPCE activity titled “Screening and Early Intervention as the Keys to Success in Lung Cancer: A Practical Approach to Implementing Lung Cancer Screening for High-Risk Individuals.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/46VvwoP. CME/MOC/AAPA/IPCE credit will be available until April 16, 2025.Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...PVI, PeerView Institute for Medical Education
Chair Lecia V. Sequist, MD, MPH, and Patrick Nana-Sinkam, MD, FCCP, discuss lung cancer screening in this CME/MOC/AAPA/IPCE activity titled “Screening and Early Intervention as the Keys to Success in Lung Cancer: A Practical Approach to Implementing Lung Cancer Screening for High-Risk Individuals.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/46VvwoP. CME/MOC/AAPA/IPCE credit will be available until April 16, 2025.Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...PVI, PeerView Institute for Medical Education
Chair and Presenter, Olalekan Oluwole, MBBS, MPH, Veronika Bachanova, MD, PhD, and David L. Porter, MD, prepared useful Practice Aids pertaining to CAR-T therapy for this CME/NCPD activity titled “Democratizing the CAR-T Experience: The Principles and Practice of Outpatient Cellular Therapy.” For the full presentation, downloadable Practice Aids, and complete CME/NCPD information, and to apply for credit, please visit us at https://bit.ly/3TfIABM. CME/NCPD credit will be available until April 15, 2025.Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...PVI, PeerView Institute for Medical Education
Chair and Presenter, Olalekan Oluwole, MBBS, MPH, Veronika Bachanova, MD, PhD, and David L. Porter, MD, discuss CAR-T therapy in this CME/NCPD activity titled “Democratizing the CAR-T Experience: The Principles and Practice of Outpatient Cellular Therapy.” For the full presentation, downloadable Practice Aids, and complete CME/NCPD information, and to apply for credit, please visit us at https://bit.ly/3TfIABM. CME/NCPD credit will be available until April 15, 2025.Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...PVI, PeerView Institute for Medical Education
Co-Chairs Lipika Goyal, MD, MPhil, and Riad Salem, MD, MBA, discuss HCC in this CME activity titled “The Convergence of Interventional Radiologists and Oncologists in HCC: Shared Decision-Making and Care Coordination at the Center of Personalized Care Across the Disease Continuum.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/48BAasz. CME credit will be available until April 26, 2025.The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...

The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...PVI, PeerView Institute for Medical Education
Chair, Richard K. Bogan, MD, FCCP, FAASM, discusses sleep disorders in this CME/MOC activity titled “Navigating Narcolepsy in Family Practice: Patient-Centered Strategies to Optimize the Experience and Outcomes of Treatment.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/48QOONd. CME/MOC credit will be available until April 9, 2025.
Navigating Narcolepsy in Family Practice: Patient-Centered Strategies to Opti...

Navigating Narcolepsy in Family Practice: Patient-Centered Strategies to Opti...PVI, PeerView Institute for Medical Education
Chair and Presenter, Jennifer Wargo, MD, MMSc, Charlotte E. Ariyan, MD, PhD, and Hussein Tawbi, MD, PhD, discuss melanoma in this CME/MOC/AAPA/IPCE activity titled “New Chapters in the Immunotherapy Story for Melanoma: Collaborative Care and Next Steps With Adjuvant and Neoadjuvant Therapy.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3T70bfi. CME/MOC/AAPA/IPCE credit will be available until April 21, 2025.New Chapters in the Immunotherapy Story for Melanoma: Collaborative Care and ...

New Chapters in the Immunotherapy Story for Melanoma: Collaborative Care and ...PVI, PeerView Institute for Medical Education
Chair and Presenters Laura S. Dominici, MD, FACS, Albert Henry Diehl, III, MD, FACS, and Jane L. Meisel, MD, discuss breast cancer in this CME/MOC/CC activity titled “Unraveling the Complex Choices in Early Breast Cancer: A Roadmap to Informed Multidisciplinary Decisions About Assessment and Treatment.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CC information, and to apply for credit, please visit us at https://bit.ly/42BZBZE. CME/MOC/CC credit will be available until April 21, 2025.Unraveling the Complex Choices in Early Breast Cancer: A Roadmap to Informed ...

Unraveling the Complex Choices in Early Breast Cancer: A Roadmap to Informed ...PVI, PeerView Institute for Medical Education
Chair, Meghan C. Thompson, MD, discusses chronic lymphocytic leukemia in this CME/MOC/NCPD/CPE/AAPA/IPCE activity titled “From Resistance to Resilience in R/R CLL: Sequencing Strategies for Achieving Effective Continuous Care.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/CPE/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3uoWOWG. CME/MOC/NCPD/CPE/AAPA/IPCE credit will be available until April 4, 2025.From Resistance to Resilience in R/R CLL: Sequencing Strategies for Achieving...

From Resistance to Resilience in R/R CLL: Sequencing Strategies for Achieving...PVI, PeerView Institute for Medical Education
More from PVI, PeerView Institute for Medical Education (20)
Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...

Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...
Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...

Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...
Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...

Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...
Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...

Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...
Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into Practice
Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into Practice
Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...

Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...
Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...

Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...
Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...
Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...
Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...

Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...
Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...
Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...
Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...
Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...
The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...

The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...
Navigating Narcolepsy in Family Practice: Patient-Centered Strategies to Opti...

Navigating Narcolepsy in Family Practice: Patient-Centered Strategies to Opti...
New Chapters in the Immunotherapy Story for Melanoma: Collaborative Care and ...

New Chapters in the Immunotherapy Story for Melanoma: Collaborative Care and ...
Unraveling the Complex Choices in Early Breast Cancer: A Roadmap to Informed ...

Unraveling the Complex Choices in Early Breast Cancer: A Roadmap to Informed ...
From Resistance to Resilience in R/R CLL: Sequencing Strategies for Achieving...

From Resistance to Resilience in R/R CLL: Sequencing Strategies for Achieving...
Recently uploaded
Model Call Girl Services in Delhi reach out to us at 🔝 9953056974 🔝✔️✔️
Our agency presents a selection of young, charming call girls available for bookings at Oyo Hotels. Experience high-class escort services at pocket-friendly rates, with our female escorts exuding both beauty and a delightful personality, ready to meet your desires. Whether it's Housewives, College girls, Russian girls, Muslim girls, or any other preference, we offer a diverse range of options to cater to your tastes.
We provide both in-call and out-call services for your convenience. Our in-call location in Delhi ensures cleanliness, hygiene, and 100% safety, while our out-call services offer doorstep delivery for added ease.
We value your time and money, hence we kindly request pic collectors, time-passers, and bargain hunters to refrain from contacting us.
Our services feature various packages at competitive rates:
One shot: ₹2000/in-call, ₹5000/out-call
Two shots with one girl: ₹3500/in-call, ₹6000/out-call
Body to body massage with sex: ₹3000/in-call
Full night for one person: ₹7000/in-call, ₹10000/out-call
Full night for more than 1 person: Contact us at 🔝 9953056974 🔝. for details
Operating 24/7, we serve various locations in Delhi, including Green Park, Lajpat Nagar, Saket, and Hauz Khas near metro stations.
For premium call girl services in Delhi 🔝 9953056974 🔝. Thank you for considering us!Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7![Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X79953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
Best Rate (Hyderabad) Call Girls Jahanuma ⟟ 8250192130 ⟟ High Class Call Girl...

Best Rate (Hyderabad) Call Girls Jahanuma ⟟ 8250192130 ⟟ High Class Call Girl...
Top Rated Bangalore Call Girls Mg Road ⟟ 9332606886 ⟟ Call Me For Genuine S...

Top Rated Bangalore Call Girls Mg Road ⟟ 9332606886 ⟟ Call Me For Genuine S...
♛VVIP Hyderabad Call Girls Chintalkunta🖕7001035870🖕Riya Kappor Top Call Girl ...

♛VVIP Hyderabad Call Girls Chintalkunta🖕7001035870🖕Riya Kappor Top Call Girl ...
Top Rated Bangalore Call Girls Ramamurthy Nagar ⟟ 9332606886 ⟟ Call Me For G...

Top Rated Bangalore Call Girls Ramamurthy Nagar ⟟ 9332606886 ⟟ Call Me For G...
Best Rate (Patna ) Call Girls Patna ⟟ 8617370543 ⟟ High Class Call Girl In 5 ...

Best Rate (Patna ) Call Girls Patna ⟟ 8617370543 ⟟ High Class Call Girl In 5 ...
Night 7k to 12k Chennai City Center Call Girls 👉👉 7427069034⭐⭐ 100% Genuine E...

Night 7k to 12k Chennai City Center Call Girls 👉👉 7427069034⭐⭐ 100% Genuine E...
All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...

All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...
Call Girls Haridwar Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Haridwar Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Agra Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Agra Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Aurangabad Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Aurangabad Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Kochi Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Kochi Just Call 8250077686 Top Class Call Girl Service Available
VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...

VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...
Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7![Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7
Call Girls Gwalior Just Call 8617370543 Top Class Call Girl Service Available

Call Girls Gwalior Just Call 8617370543 Top Class Call Girl Service Available
Call Girls Guntur Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Guntur Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Nagpur Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Nagpur Just Call 9907093804 Top Class Call Girl Service Available
(👑VVIP ISHAAN ) Russian Call Girls Service Navi Mumbai🖕9920874524🖕Independent...

(👑VVIP ISHAAN ) Russian Call Girls Service Navi Mumbai🖕9920874524🖕Independent...
Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...

Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...
Call Girls Tirupati Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Tirupati Just Call 8250077686 Top Class Call Girl Service Available
Top Quality Call Girl Service Kalyanpur 6378878445 Available Call Girls Any Time

Top Quality Call Girl Service Kalyanpur 6378878445 Available Call Girls Any Time
Novel Induction Options in AML: Assessing the Implications for HCT-Eligible Patients
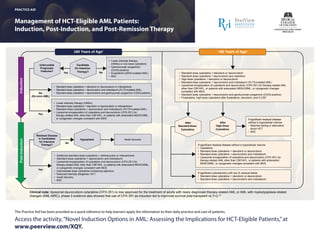
- 1. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Management of HCT-Eligible AML Patients: Induction, Post-Induction, and Post-Remission Therapy PRACTICE AID Access the activity,“Novel Induction Options in AML: Assessing the Implications for HCT-Eligible Patients,”at www.peerview.com/XQY. ≥60 Years of Age1 <60 Years of Age1 Induction • Standard-dose cytarabine + idarubicin or daunorubicin • Standard-dose cytarabine + daunorubicin and cladribine • High-dose cytarabine + idarubicin or daunorubicin • Standard-dose cytarabine + daunorubicin and midostaurin (FLT3-mutated AML) • Liposomal encapsulation of cytarabine and daunorubicin (CPX-351) for therapy-related AML other than CBF/APL, or patients with antecedent MDS/CMML, or cytogenetic changes consistent with MDS • Standard-dose cytarabine + daunorubicin and gemtuzumab ozogamicin (CD33-positive) • Fludarabine, high-dose cytarabine after fludarabine, idarubicin, and G-CSF • Lower intensity therapy (HMAs) or low-dose cytarabine • Gemtuzumab ozogamicin (CD33-positive) • Enasidenib (IDH2-mutated AML) • BSC • Additional standard-dose cytarabine + anthracycline or mitoxantrone • Standard-dose cytarabine + daunorubicin and midostaurin • Liposomal encapsulation of cytarabine and daunorubicin (CPX-351) for therapy-related AML other than CBF/APL, or patients with antecedent MDS/CMML, or cytogenetic changes consistent with MDS • Intermediate-dose cytarabine-containing regimens • Reduced-intensity allogeneic HCT • Await recovery • BSC If significant cytoreduction with low % residual blasts • Standard-dose cytarabine + idarubicin or daunorubicin • Standard-dose cytarabine + daunorubicin and midostaurin Post-Induction Yes No Unfavorable Prognostic Features? Yes No (De novo AML) Residual Disease in Candidates for Intensive Therapy? Hypoplasia Await recovery After Standard-Dose Cytarabine After High-Dose Cytarabine If significant residual disease without a hypocellular marrow • Matched sibling or alternative donor HCT • BSC • Standard-dose cytarabine + idarubicin or daunorubicin or mitoxantrone • Standard-dose cytarabine + daunorubicin and midostaurin (FLT3-mutated AML) • Standard-dose cytarabine + daunorubicin and gemtuzumab ozogamicin (CD33-positive) • Lower-intensity therapy (HMAs) • Standard-dose cytarabine + idarubicin or daunorubicin or mitoxantrone • Standard-dose cytarabine + daunorubicin and midostaurin (FLT3-mutated AML) • Liposomal encapsulation of cytarabine and daunorubicin (CPX-351) for therapy-related AML other than CBF/APL, or patients with antecedent MDS/CMML, or cytogenetic changes consistent with MDS No Yes Clinical note: liposomal daunorubicin-cytarabine (CPX-351) is now approved for the treatment of adults with newly diagnosed therapy-related AML or AML with myelodysplasia-related changes (AML-MRC); phase 3 evidence also showed that use of CPX-351 as induction led to improved survival post-transplant vs 7+3.2,3 If significant residual disease without a hypocellular marrow • Cytarabine • Standard-dose cytarabine + idarubicin or daunorubicin • Standard-dose cytarabine + daunorubicin and midostaurin • Liposomal encapsulation of cytarabine and daunorubicin (CPX-351) for therapy-related AML other than CBF/APL, or patients with antecedent MDS/CMML, or cytogenetic changes consistent with MDS Candidate for Intensive Therapy?
- 2. Management of HCT-Eligible AML Patients: Induction, Post-Induction, and Post-Remission Therapy AML: acute myeloid leukemia; APL: acute promyelocytic leukemia; BSC: best supportive care; CBF: core-binding factor; CD: cluster of differentiation; CMML: chronic myelomonocytic leukemia; FLT3: fms-like tyrosine kinase 3; G-CSF: granulocyte colony-stimulating factor; HCT: hematopoietic stem cell transplantation; HMAs: hypomethylating agents; IDH2: isocitrate dehydrogenase 2; MDS: myelodysplastic syndrome; PS: performance status. 1. National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology. Acute Myeloid Leukemia. Version 1.2018. 2. Lancet JE et al. 2017 Annual BMT Tandem Meetings (BMT Tandem 2017). Abstract 19. 3. Vyxeos (daunorubicin and cytarabine) Prescribing Information. https://www.accessdata.fda. gov/drugsatfda_docs/label/2017/209401s000lbl.pdf. Accessed February 6, 2018. PRACTICE AID Access the activity,“Novel Induction Options in AML: Assessing the Implications for HCT-Eligible Patients,”at www.peerview.com/XQY. ≥60 Years of Age1 <60 Years of Age1 • Matched sibling/alternative donor HCT • High-dose cytarabine • High-dose cytarabine and midostaurin (FLT3-mutated AML) • Liposomal encapsulation of cytarabine and daunorubicin (CPX-351) for therapy-related AML other than CBF/APL, or patients with antecedent MDS/CMML, or cytogenetic changes consistent with MDS • BSC Post-Remission(Consolidation)Therapy Previous Intensive Therapy Yes Yes No No Response? Previous Low-Intensity Therapy Poor-Risk Cytogenetics/ Molecular Abnormalities • Reduced-intensity HCT • Clinical trial • Standard-dose cytarabine ± anthracycline (idarubicin or daunorubicin) • Consider intermediate-dose cytarabine in patients with good PS, normal renal function, and better-risk/normal karyotype with favorable molecular markers • Intermediate-dose cytarabine with midostaurin • Liposomal encapsulation of cytarabine and daunorubicin (CPX-351) for therapy-related AML other than CBF/APL, or patients with antecedent MDS/CMML, or cytogenetic changes consistent with MDS • Standard-dose cytarabine + daunorubicin and gemtuzumab ozogamicin (CD33-positive) • Maintenance with HMAs until progression (if patient received HMAs during induction) • Observation Induction failure • Allogeneic HCT (preferably in clinical trial) • BSC CBF Translocations Treatment- Related DiseaseOr Complete Response? • High-dose cytarabine • Standard-dose cytarabine + daunorubicin and gemtuzumab ozogamicin (CD33-positive) No KIT Mutation or Favorable-Risk Molecular Abnormalities • Matched sibling or alternative donor HCT • High-dose cytarabine • High-dose cytarabine and midostaurin (FLT3-mutated AML) • Standard-dose cytarabine + daunorubicin and gemtuzumab ozogamicin (CD33-positive) Intermediate-Risk Cytogenetics/ Molecular Abnormalities • Reduced-intensity HCT • Continue HMA therapy every 4-6 weeks until progression • Gemtuzumab ozogamicin (CD33-positive) • Continue enasidenib until progression (IDH2-mutated AML)
- 3. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. A Snapshot of Innovative Therapies in AML: Current Status, Dosing, and Other Considerations a For additional induction use d 1 and 3 for subsequent cycles, if needed; for consolidation: daunorubicin 29 mg/m2 and cytarabine 65 mg/m2 liposome IV over 90 mins on d 1 and 3. AE: adverse event; AML: acute myeloid leukemia; AML-MRC: AML with myelodysplasia-related changes; CD: cluster of differentiation; CT: chemotherapy; FLT3: fms-like tyrosine kinase 3; FN: febrile neutropenia; IDH1: isocitrate dehydrogenase 1; IDH2: isocitrate dehydrogenase 2; R/R: relapsed or refractory; t-AML: therapy-related acute myeloid leukemia. 1. Lancet JE et al. 2016 Annual Meeting of the American Society of Clinical Oncology (ASCO 2016). Abstract 7000. 2. Lancet JE et al. 2017 Annual BMT Tandem Meetings (BMT Tandem 2017). Abstract 19. 3. Vyxeos (daunorubicin and cytarabine) Prescribing Information. https://www.accessdata.fda. gov/drugsatfda_docs/label/2017/209401s000lbl.pdf. Accessed February 6, 2018. 4. Rydapt (midostaurin) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/207997s000lbl.pdf. Accessed February 6, 2018. 5. Idhifa (enasidenib) Prescribing Information. https:// www.accessdata.fda.gov/drugsatfda_docs/label/2017/209606s000lbl.pdf. Accessed February 6, 2018. 6. https://clinicaltrials.gov/ct2/show/NCT03173248. Accessed February 6, 2018. 7. Mylotarg (gemtuzumab ozogamicin) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/ label/2017/761060lbl.pdf. Accessed February 6, 2018. PRACTICE AID Access the activity,“Novel Induction Options in AML: Assessing the Implications for HCT-Eligible Patients,”at www.peerview.com/XQY. DRUG Midostaurin4 STATUS TARGET/ FORMULATION DOSE CONSIDERATIONS Approved Plus CT in FLT3-mutant AML FLT3 50 mg orally twice daily with food q GI events most common q Promote therapy adherence q Be mindful of potential drug–drug interactions Enasidenib5 Approved R/R IDH2-mutation–positive AML IDH2 100 mg orally daily Monitor for: q IDH-differentiation syndrome q GI events q Elevated bilirubin Ivosidenib6 Phase 3; under FDA review R/R IDH1-mutation–positive AML IDH1 500 mg orally daily Most common AEs in trials: q Diarrhea, leukocytosis, nausea, fatigue, and FN q IDH-differentiation syndrome also reported CPX-3511-3 Approved Adults with newly diagnosed t-AML or AML-MRC Liposomal cytarabine + daunorubicin 5:1 molar ratio Induction: daunorubicin 44 mg/m2 and cytarabine 100 mg/m2 liposome IV over 90 mins d 1, 3, and 5a q Monitor blood counts regularly until recovery q Not recommended in pts with cardiac function less than normal Gemtuzumab ozogamicin7 Approved Newly diagnosed CD33-positive AML in adults, R/R CD33-positive AML in adults, and in pediatric pts aged ≥2 y CD33 Induction: 3 mg/m2 (up to one 4.5 mg vial) d 1, 4, and 7 in combination with daunorubicin and cytarabine q Infusion-related reactions q Premedicate with corticosteroid, antihistamine, and acetaminophen q Monitor platelet counts
