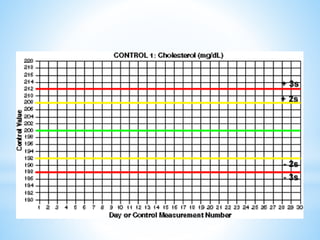
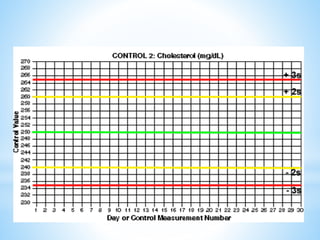
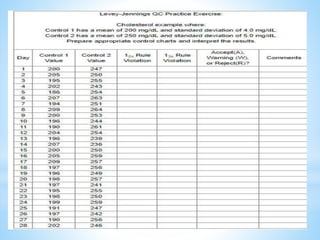
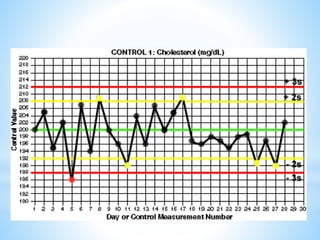
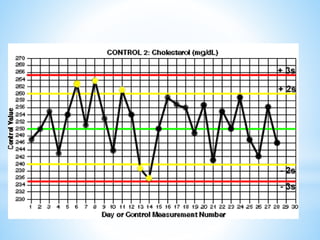
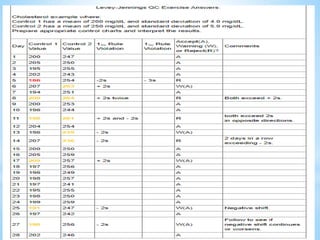
This document describes how to construct Levey-Jennings control charts to monitor the performance of a cholesterol testing method. It provides details on calculating control limits from control sample measurements and plotting control results on charts to identify out-of-control runs. Applying the 12s, 13s, 22s and R4s rules can help determine whether runs should be rejected, with the 13s/22s/R4s multirule procedure balancing sensitivity and false rejections. Worked examples are given of interpreting control charts and identifying problems based on rule violations.