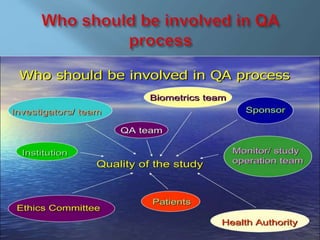
The document discusses audit and inspection in the context of local authorities in England. It summarizes the key findings of a 2019 parliamentary report on the topic. The report found that the 2014 Local Audit and Accountability Act fragmented accountability and that the new audit regime is not as effective as the previous Audit Commission in ensuring value for money and fairness. It also discusses limitations of audits and inspections in improving outcomes and ensuring food safety. The document covers the purpose and goals of public audits, inspector independence, the impact of changing regulations, and ways auditing could be improved.