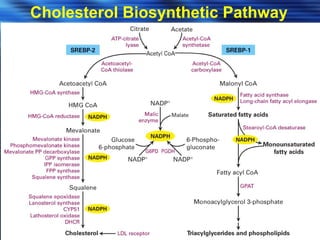
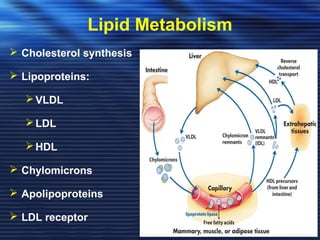
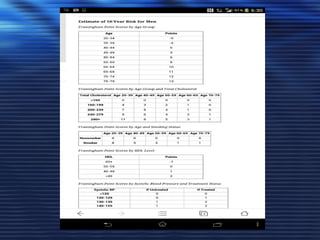
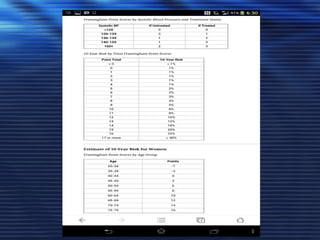
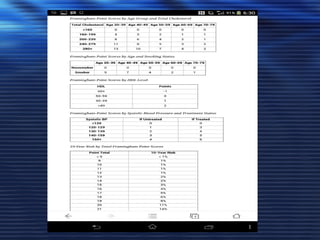
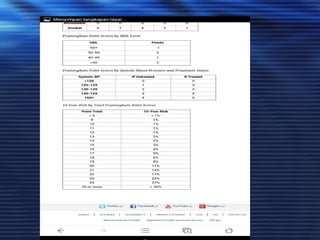
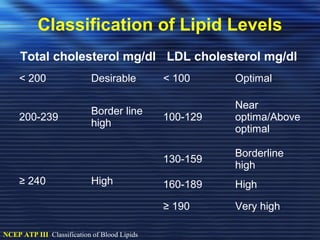
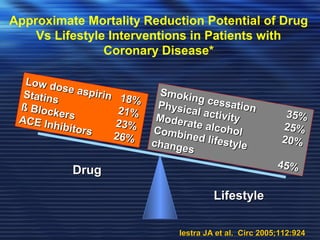
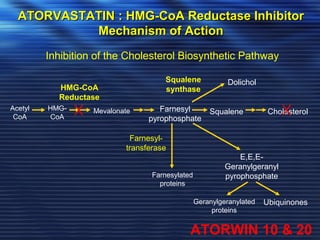
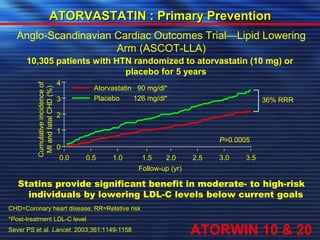
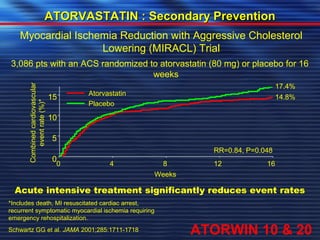
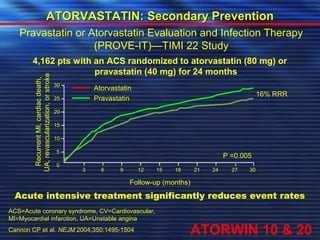
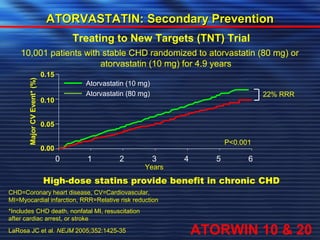
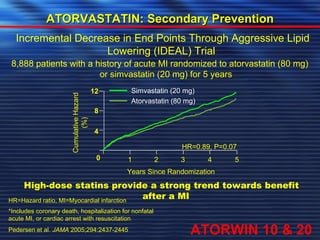
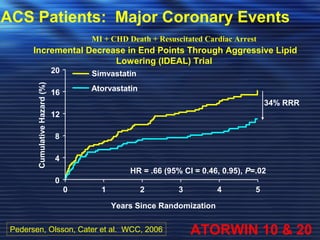
- Studies have shown that lowering LDL cholesterol through statin therapy such as atorvastatin provides significant cardiovascular benefits in both primary and secondary prevention. Atorvastatin in particular has been shown in clinical trials to reduce cardiovascular events and mortality when used for acute coronary syndromes, stable coronary heart disease, and among those at high risk of cardiovascular disease. Atorvastatin may be a good choice of statin due to its proven efficacy in improving cardiovascular outcomes.