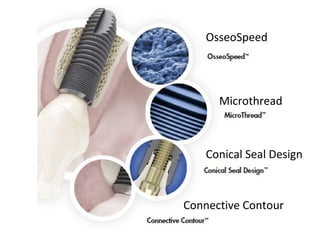
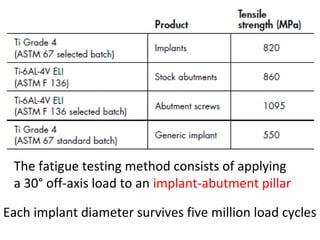
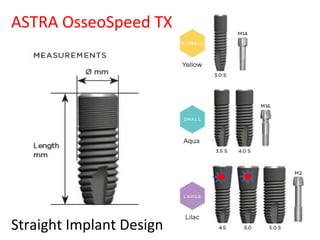
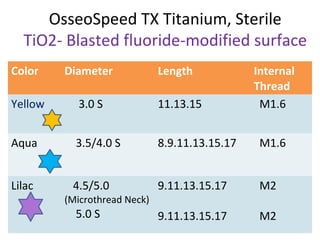
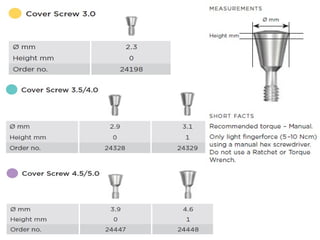
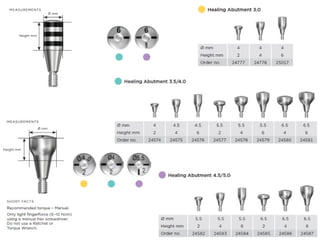
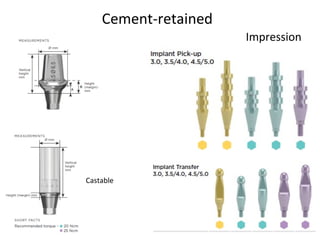
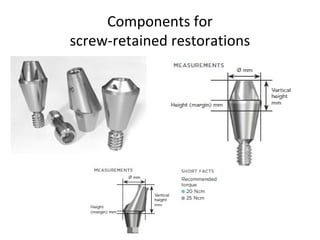
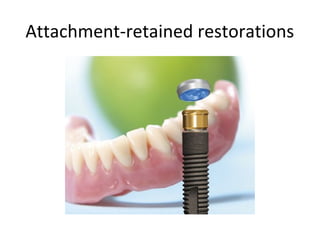
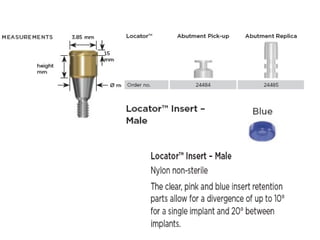
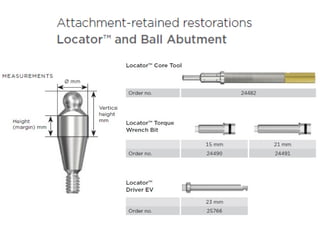
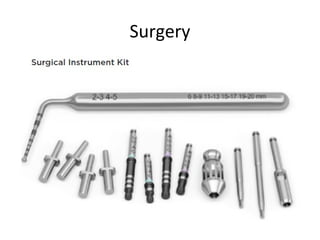
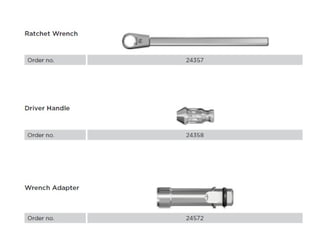
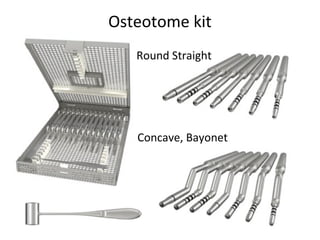
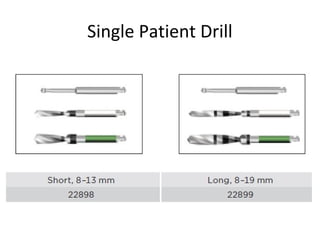
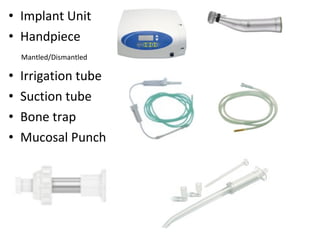
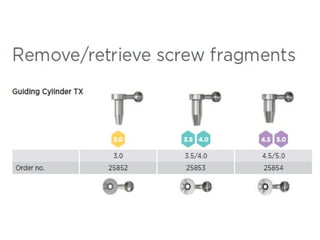
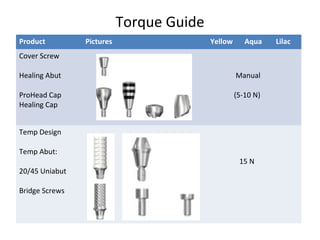
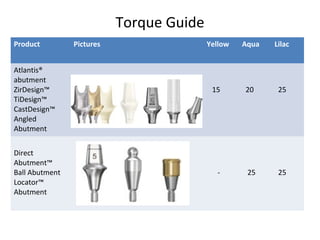
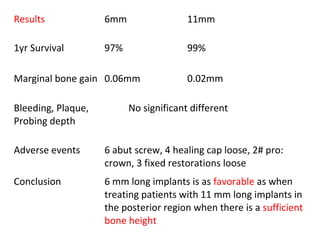
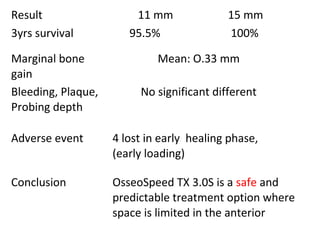
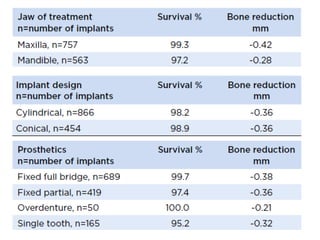
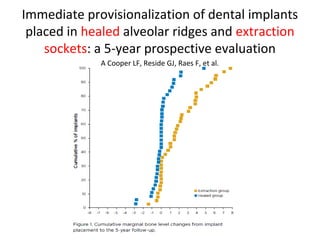
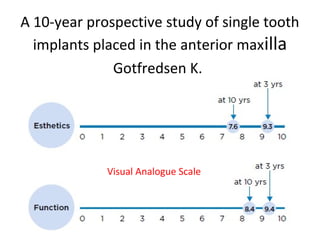
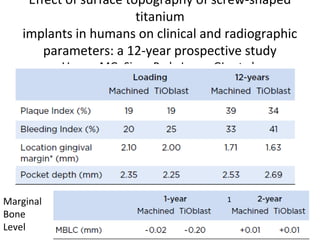
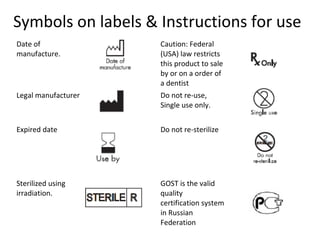
The document outlines the collaboration of the Myanmar Dental Association and the Myanmar Society of Oral Implantology in a seminar focused on Astra Tech dental implants, detailing the desirable characteristics, design, fatigue testing, and research surrounding these implants. It highlights various studies related to implant success, immediate loading, and the performance of different implant types in various clinical scenarios, emphasizing the importance of design and material selection for optimal outcomes. The comprehensive overview includes specific product details, testing methodologies, and clinical trial results relevant to dental implant technology.