APAR terminology flowcharts
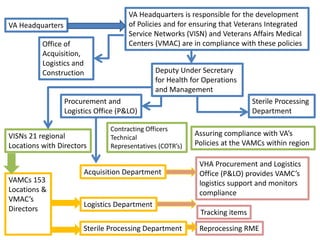
- 1. VA Headquarters is responsible for the development of Policies and for ensuring that Veterans Integrated Service Networks (VISN) and Veterans Affairs Medical Centers Office of (VMAC) are in compliance with these policies Acquisition, Logistics and Construction Deputy Under Secretary VA Headquarters Procurement and Logistics Office (P&LO) VISNs 21 regional Locations with Directors VAMCs 153 Locations & VMAC’s Directors for Health for Operations and Management Contracting Officers Technical Representatives (COTR’s) Acquisition Department Logistics Department Sterile Processing Department Assuring compliance with VA’s Policies at the VAMCs within region VHA Procurement and Logistics Office (P&LO) provides VAMC’s logistics support and monitors compliance Tracking items Sterile Processing Department Reprocessing RME
- 2. Facility Location Warehouse Inventory Primary Inventory Secondary Inventory Normal Stock Level Emergency Stock Level Temporary Stock Level Standard Reorder Point Optional Reorder Point Generate order Kicks back to Purchasing tab General Inventory Categories Functional Areas Requiring GIP Expendable Medical Equipment Reusable Medical Equipment
- 3. Items Primary inventory models 1) Posted Stock 2) Inventory with Distribution Points 3) Stand Alone Inventory Standard Items On-Demand (Just In Case) Item An average of no more than 30 to 90 days stock on hand is required, those types of stock levels are; 1) Normal Stock Level 2) Emergency Stock Level 3) Temporary Stock Level 4) Standard Reorder Point Level 5) Optional Reorder Point Level Primary inventory 1) History of Distribution report 2) Inactive Item report 3) Packaging or Procurement Source Discrepancy report 4) Stock Status report 5) Due-In Item report 6) Days of Stock on Hand report 7) Limited Access Expendable Medical Equipment Reusable Medical Equipment
- 4. IFCAP Systems GIP a portion of IFCAP IFCAP Maintenance of supplies from Outside Vendor & Supply Warehouse PIP integrates with IFCAP & CPRS Computerized Patient Record System GIP Maintenance of supplies to facilities; there is three levels 1) Warehouse maintains a supply of items that are repetitively used by multiple services and is funded by Supply Fund 2) The primary inventory is the main inventory for a using department 3) The secondary inventories are the points of distribution. NOTE: Supply Processing and Distribution (SPD) is typically the main inventory for medical and surgical supplies Manage the receipt, distribution Identifies candidates items for and stock maintenance standardization, tracks and promotes compliance, generates usage cost reports, and provides a mechanism to evaluate vendor delivery performance Pharmacy PO/Inventory system Electronic Contract Management System (eCMS) actions valued at $25,000 or more One Sourcedocs database to obtain manufacturer’s IFU VAMC’s must enter information on all RME entered into the AEMS/MERS Federal Supply Services
- 5. IFCAP Maintenance of supplies from Outside Vendor & Supply Warehouse GIP Maintenance of supplies to facilities; there is three levels 1) Warehouse 2) The primary inventory 3) The secondary inventories Assumption: GIP will be the Expendable Medical Equipment DB Electronic Contract Management System (eCMS) actions valued at $25,000 or more Assumption: eCMS manages Purchase card and contract relationships with vendors PIP Prosthetics, Assumption: RME – Surgical and Dental inventory management system One Sourcedocs database to obtain manufacturer’s IFU Assumption: Sourcedocs manages sterilization in-stock and pm-stock also tracking of all inventory specifications and recalls. Pharmacy PO/Inventory system - Assumption: in patient and out patient Pharmacy PO process Purchase card Process & Federal Supply Schedule Program AEMS/MERS Assumption: manages all preventive maintenance and audits. Computerized Patient Record System