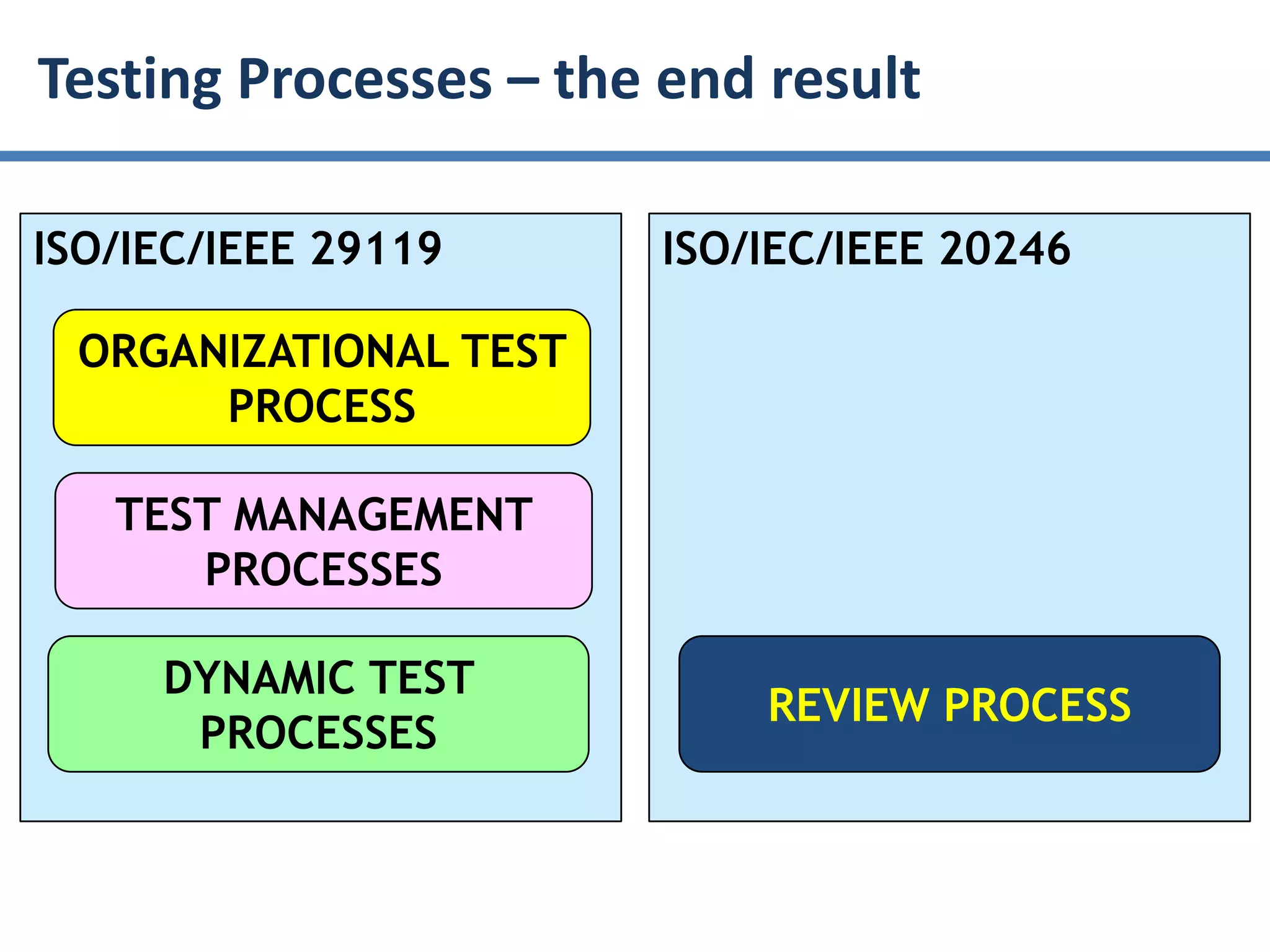
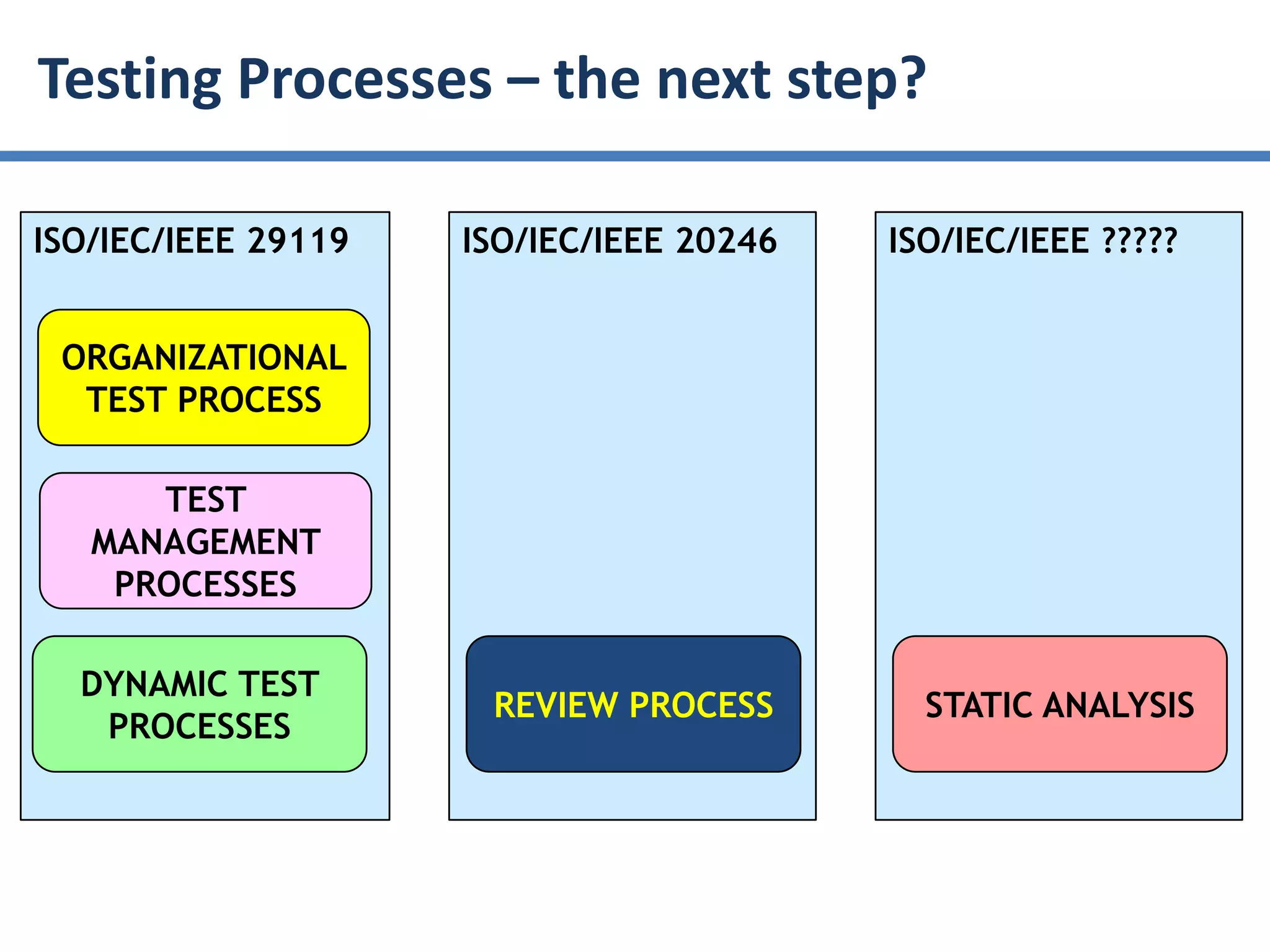
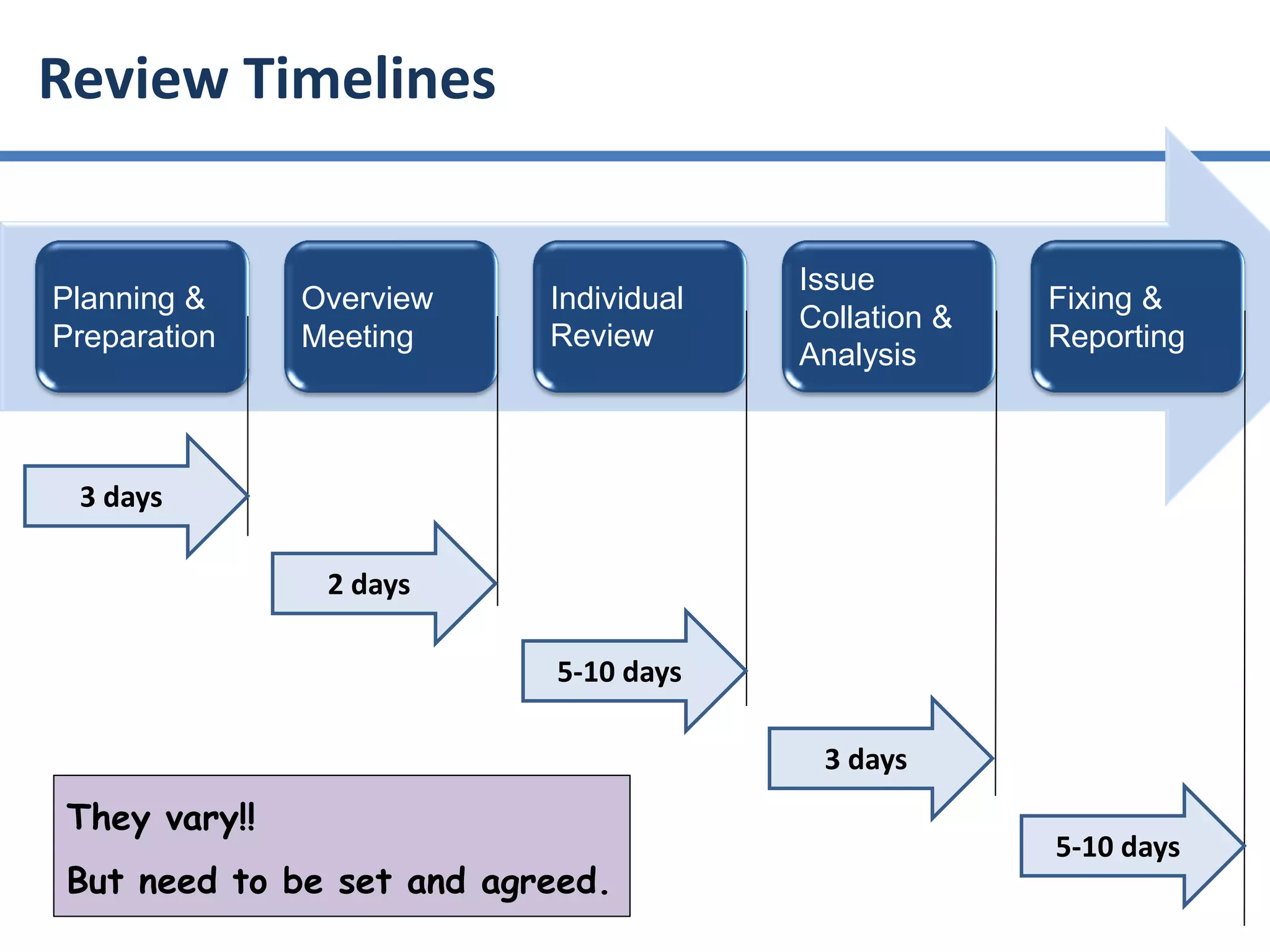
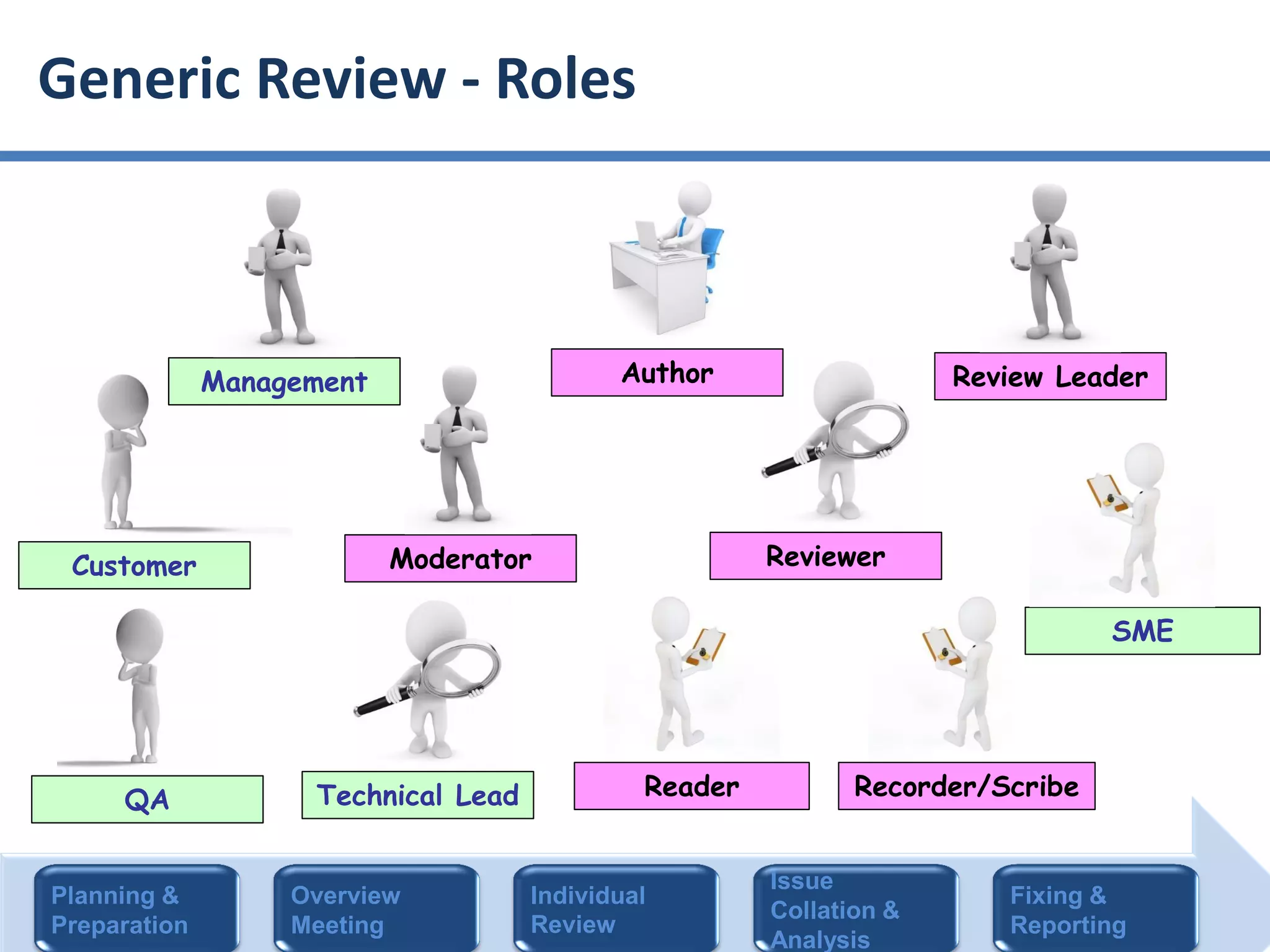
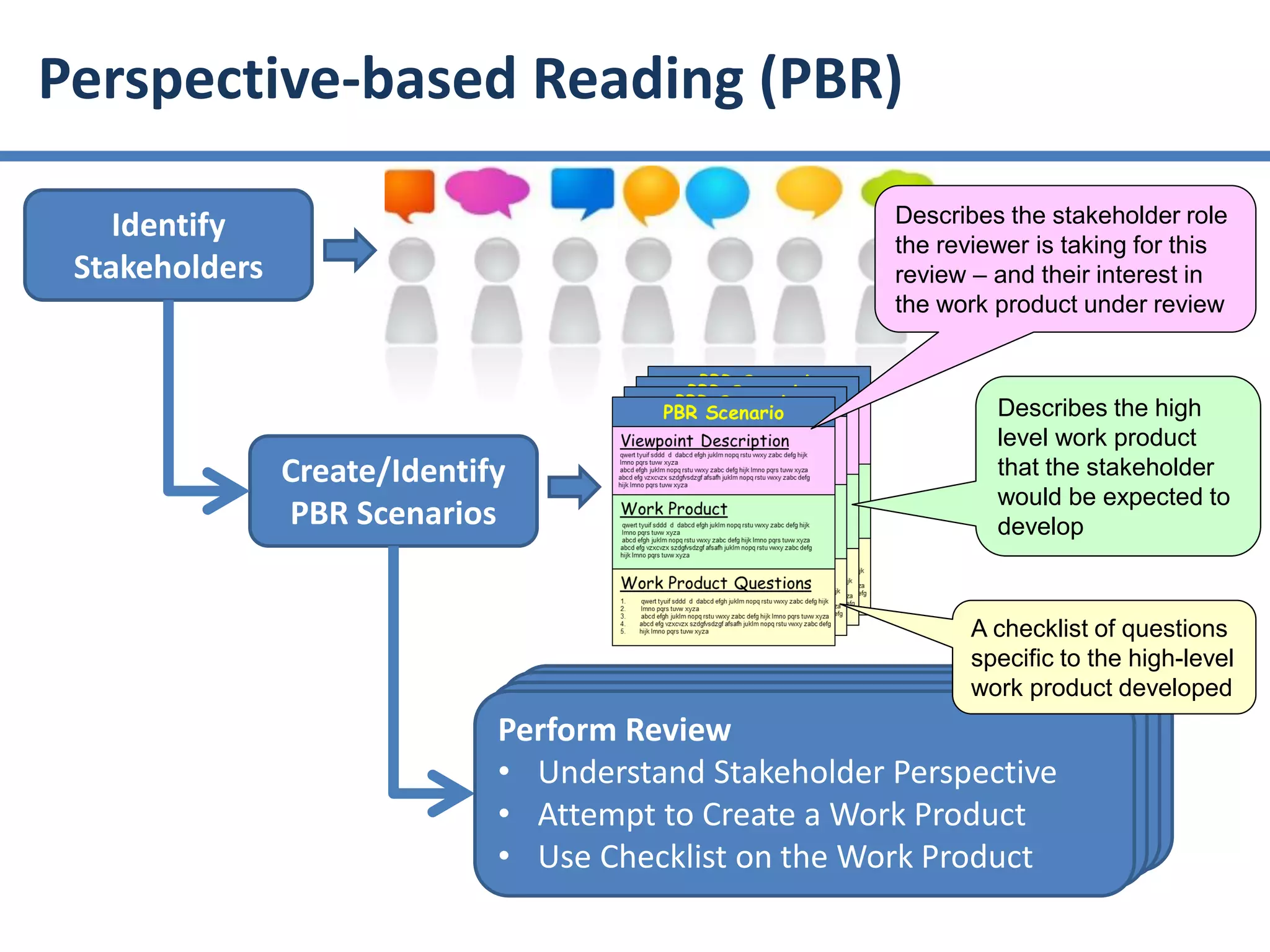
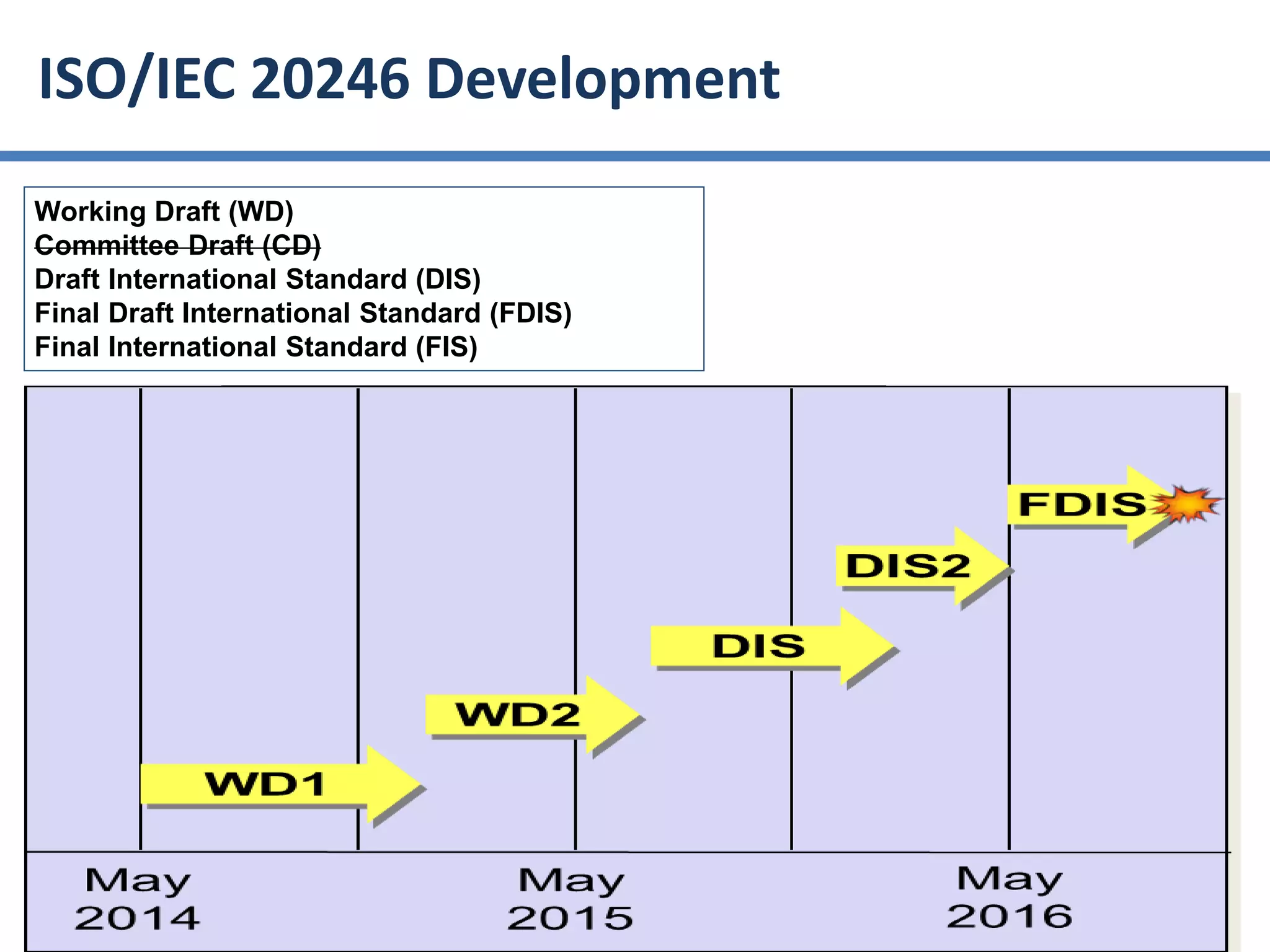
The document summarizes ISO/IEC 20246, which provides guidance on work product reviews. It describes the generic review process, including planning, individual reviews, issue collation, and reporting. It also covers different review techniques like checklist-based and scenario-based reviewing. Finally, it discusses factors to consider when choosing a review approach and provides an overview of the ISO 20246 standard development process and opportunities to get involved.