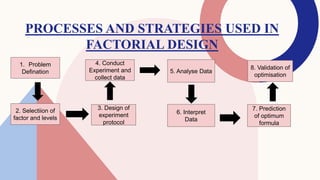
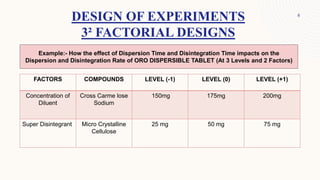
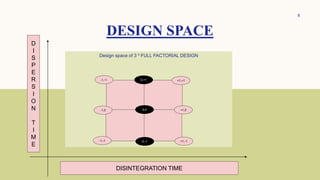
The document discusses the concept of 32 full factorial design in experimental research, focusing on optimizing processes to minimize experimental runs, manpower, and costs. It details the steps involved in factorial design, such as problem definition, selection of factors, conducting experiments, and analyzing results. Additionally, examples of applying factorial designs to determine the effects of various factors on tablet characteristics are provided.