Parts of the Atom Labeling
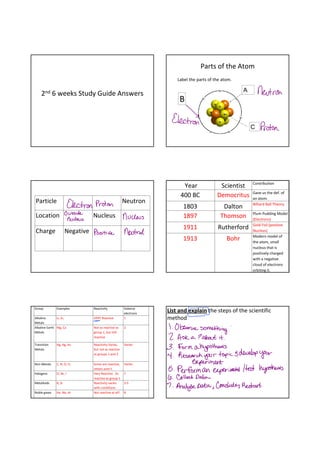
- 1. Parts of the Atom Label the parts of the atom. A 2nd 6 weeks Study Guide Answers B C Contribution Year Scientist Gave us the def. of 400 BC Democritus an atom. Particle Neutron Billiard Ball Theory 1803 Dalton Plum Pudding Model Location Nucleus 1897 Thomson (Electrons) Gold Foil (positive 1911 Rutherford Charge Negative Nucleus) Modern model of 1913 Bohr the atom, small nucleus that is positively charged with a negative cloud of electrons orbiting it. Group Examples Reactivity Valence electrons List and explain the steps of the scientific Alkaline Li, Sr, VERY Reactive 1 method Metals Alkaline Earth Mg, Ca Not as reactive as 2 Metals group 1, but still reactive. Transition Hg, Ag, Au Reactivity Varies, Varies Metals but not as reactive as groups 1 and 2 Non-Metals C, N, O, H, Some are reactive, Varies others aren’t Halogens Cl, Br, I Very Reactive. As 7 reactive as group 1 Metalloids B, Si Reactivity varies 3-5 with conditions Noble gases He, Ne, Ar Not reactive at all! 8
- 2. •Write out the formula for density and complete the following •A student finds a rock on the way to calculations: SHOW ALL WORK. school. In the laboratory he determines that •Calculate the density of a material that has a mass of 52.457 g the volume of the rock is 22.7 cm3, and the and a volume of 13.5 cm3. mass in 39.943 g. What is the density of the rock? •The density of silver is 10.49 g/cm3. If a sample of •How many grams of tin would occupy 5.5 L, if it pure silver has a volume of 12.993 cm3, what would has a density of 7.265 g/cm3? the mass? A car is driving 70 mph down the Pure gold has a density of 19.32 g/cm3. How large would a piece of gold be if it had a mass of 318.97 g? highway. How many m/s is this? There are 1.6 km in a mile.
- 3. 3.650g =_________kg 450000000mL=________kL Describe the differences and 1.3x107 cm=_________m similarities between physical and chemical properties. How did Dmitri Mendeleev organize his periodic table? • Glass Breaking • Ice Melting • Burning Wood • Air Freshener Evaporating
- 4. How did Henry Mosely organize his What is the difference between a periodic Table? period and a group? How do you calculate the number of How many electrons, neutrons and neutrons in an atom? protons are in the following elements? Element Protons Neutrons Electrons Oxygen Nitrogen Argon Lithium Boron Ms. Milsaps Demo Day • Types of reactions