2015-CLSA-Report-Recent-Trends-in-FDA-Med-Device-Regulation-Final
•
0 likes•122 views
The document summarizes recent trends in FDA medical device regulation. It finds that after plateauing in previous years, 510(k) clearance times have recently decreased, suggesting improvements. PMA review times have also improved markedly over the past few years, with the exception of 2013 which saw an increase in panel reviews. While progress has been made, continued efforts are needed to refine review processes and ensure consistent performance across the FDA's divisions.
Report
Share
Report
Share
Download to read offline
Recommended
Fda Cms Pro Industry Initiatives

Are FDA/CMS "pro-industry" initiatives beneficial or harmful to emerging technologies?
FDA Data Integrity: Misconceptions of 21 CFR Part 11 

Martin Browning, co-author of FDA's 21 CFR Part 11 regulation for electronic records and signatures, explains FDA's expectations for data integrity and common industry misconceptions about how to comply with the rule.
The CRO-Sponsor Interaction: Regulatory Affairs

In this presentation, Chris Ingram discusses the key factors for positive CRO-Sponsor relationships.
The Use of EDC in Canadian Clinical Trials

Presentation at CHEO Research Rounds on a study to estimate the proportion of Canadian clinical trials that are using an Electronic Data Capture system during the period 2006-2007.
Presentation investing-in-medical-device-safety

Investing in medical device safety: The importance of a functional Quality Management System
Conducting Clinical Trials in Asia: Global Engage.May.2013

Medpace Director of Clinical Operations Mainfong Ang, presented a current view of best practices in Asia at the Global Engage conference, May 2013
Recommended
Fda Cms Pro Industry Initiatives

Are FDA/CMS "pro-industry" initiatives beneficial or harmful to emerging technologies?
FDA Data Integrity: Misconceptions of 21 CFR Part 11 

Martin Browning, co-author of FDA's 21 CFR Part 11 regulation for electronic records and signatures, explains FDA's expectations for data integrity and common industry misconceptions about how to comply with the rule.
The CRO-Sponsor Interaction: Regulatory Affairs

In this presentation, Chris Ingram discusses the key factors for positive CRO-Sponsor relationships.
The Use of EDC in Canadian Clinical Trials

Presentation at CHEO Research Rounds on a study to estimate the proportion of Canadian clinical trials that are using an Electronic Data Capture system during the period 2006-2007.
Presentation investing-in-medical-device-safety

Investing in medical device safety: The importance of a functional Quality Management System
Conducting Clinical Trials in Asia: Global Engage.May.2013

Medpace Director of Clinical Operations Mainfong Ang, presented a current view of best practices in Asia at the Global Engage conference, May 2013
Top Regulatory Trends in the Life Sciences Industry in 2017

Almost every major life sciences company has been under a settlement action or monetary penalty situation
Lab data integrity 

Data Integrity in pharmaceutical laboratories is a must, the attached ppt shall help the QC members to understand and develop an integral analytical culture
Routine Diagnostics | Advance Diagnostics | Pathology Lab | Blood Test | Molq

MolQ pathology lab provides you both of diagnostics Routine and Advance with the team of experts and researchers.
What Happens After Your Device is Approved? Collecting Data in the Real World

In this workshop, Medpace will discuss key considerations for generating real-world evidence and how to apply critical insights in order to drive late-stage clinical research. To listen to this presentation, visit https://vimeo.com/168768256
Synergy CRO capabilities as of 4 Aug 2016

Excellent presentation prepared by our COO John Barker for American client. Right-to-the-point, simple and crystal clear. Great job!
A New Regulatory Model for India

Summary of key changes and its impacts on medical device industry in India due to the introduction of Indian Medical Rules, 2017
Regulatory Concerns When Running Virtual/Paperless Clinical Trials

With drug and device manufacturers and the U.S. Food and Drug Administration (FDA) supplying much of the push, so-called paperless clinical trials are gaining momentum. In this eClinical Forum webinar, Dr. Jules Mitchel, President of Target Health, facilitated the discussion on the future landscape and regulatory concerns of paperless clinical trials and clinical trial design incorporating mobile tools.
Rx15 tpp tues_330_1_gavin_2nader_3jeter

Third-Party Payer Track: Rx Drugs and Urine Testing: Knowing What’s Too Much, Too Little and Just Right
A Pharma/CRO Partnership in the Design and Execution of Paperless Clinical Tr...

DIA 2019 presentation by Dr. Jules Mitchel with Michelle Eli (Lilly) and Tom Haag (ex-Novartis) based on their experience with Lilly collaborating on Target Health's paperless clinical trial system.
Data Integrity in a GxP-regulated Environment - Pauwels Consulting Academy

On Tuesday, December 6, 2016, our colleague Angelo Rossi, Senior Regulatory Compliance Consultant, gave an interesting presentation about “Data Integrity in a GxP-regulated Environment” at the Brussels Office of Pauwels Consulting in Diegem.
In his presentation, Angelo covered definitions and concepts of data integrity, the change in regulatory focus, lessons learned from recent FDA warning letters, importants highlights of regulations and guidelines. Angelo also presented a practical example of data integrity for a computerized system.
Please contact us at contact@pauwelsconsulting.com or +32 9 324 70 80 if you have any further questions regarding our consulting services in this area.
Surveyors are Here

Do you feel like there are more regulatory and accrediting surveys being conducted than ever before? Have you been the host of a CMS survey…or two… in the last several years? Do you feel like the surveys are becoming more onerous or prescriptive?
2019-07 James Sullivan - Case Study - Endomechanicals

James Sullivan: Case Study - Endomechanicals Analysis
RAC US Exam Questions

Now you don’t need to take any stress about the RAC US Exam. We provide you real exam questions along with updated Test Engine. You can pass your exam in first attempt with 100% passing assurance and money back guarantee. Get amazing flat 15% discount on RAC US exam and pass your RAC US Dumps Exam. Visit us for more information and RAC US Exam Questions.
https://www.certs4you.com/raps/rac-us-dumps.html
Data integrity - Regulatory Perspective and Challenges: 

Data Integrity: Regulatory Perspective and Challenges.
This presentation will present the importance of data integrity in the pharma industry with regards to the quality compliance and its impact on industry. This presentation also emphasizes on importance of regulatory affairs department in ensuring the data integrity and regulatory challenges.
How eSource Solutions are Impacting Clinical Research Sites, Patients, Regula...

Presented at DIA 2017 by Jules Mitchel, Mitchell Efros, and Jonathan Helfgott.
FDA Electronic Health Record Data in Clinical Research

Dr. Jules Mitchel, President of Target Health, delivered this presentation on the new FDA Guidance on the use of the electronic health record for clinical research at the North American eClinical Forum Autumn Meeting on October 2, 2018.
Life Science Fast Track Faculty and Mentor Directory

Are you a physician, innovator, or entrepreneur with a commercial idea and ready to make it a fundable company? Join us for this full-day program to understand today’s unique challenges for life science, med device, and healthcare entrepreneurs starting companies and raising capital. Learn from and meet some of the leading life science investors, entrepreneurs, and early-stage experts in New England.
Building on the success of The Capital Network’s annual Fast Track Boot Camp for Entrepreneurs, this Life Science Fast Track is an intensive, interactive full-day seminar designed to provide entrepreneurs and innovators an in-depth understanding of what it takes to raise early stage capital for a life science startup.
Modern SEO Techniques Part 1:New SEO Methods for Medical Device Marketers

This two-part series on Modern SEO Techniques for Med Device Marketers can be used by any medical device marketer to understand SEO. Part one will cover modern link building and part two will cover user experience as a search engine ranking factor.
Search Engines. We love them when we’re looking for the hottest sushi restaurant in town. But when it comes to getting our business to rank at the top, they can be a pain in the wasabi.
More Related Content
What's hot
Top Regulatory Trends in the Life Sciences Industry in 2017

Almost every major life sciences company has been under a settlement action or monetary penalty situation
Lab data integrity 

Data Integrity in pharmaceutical laboratories is a must, the attached ppt shall help the QC members to understand and develop an integral analytical culture
Routine Diagnostics | Advance Diagnostics | Pathology Lab | Blood Test | Molq

MolQ pathology lab provides you both of diagnostics Routine and Advance with the team of experts and researchers.
What Happens After Your Device is Approved? Collecting Data in the Real World

In this workshop, Medpace will discuss key considerations for generating real-world evidence and how to apply critical insights in order to drive late-stage clinical research. To listen to this presentation, visit https://vimeo.com/168768256
Synergy CRO capabilities as of 4 Aug 2016

Excellent presentation prepared by our COO John Barker for American client. Right-to-the-point, simple and crystal clear. Great job!
A New Regulatory Model for India

Summary of key changes and its impacts on medical device industry in India due to the introduction of Indian Medical Rules, 2017
Regulatory Concerns When Running Virtual/Paperless Clinical Trials

With drug and device manufacturers and the U.S. Food and Drug Administration (FDA) supplying much of the push, so-called paperless clinical trials are gaining momentum. In this eClinical Forum webinar, Dr. Jules Mitchel, President of Target Health, facilitated the discussion on the future landscape and regulatory concerns of paperless clinical trials and clinical trial design incorporating mobile tools.
Rx15 tpp tues_330_1_gavin_2nader_3jeter

Third-Party Payer Track: Rx Drugs and Urine Testing: Knowing What’s Too Much, Too Little and Just Right
A Pharma/CRO Partnership in the Design and Execution of Paperless Clinical Tr...

DIA 2019 presentation by Dr. Jules Mitchel with Michelle Eli (Lilly) and Tom Haag (ex-Novartis) based on their experience with Lilly collaborating on Target Health's paperless clinical trial system.
Data Integrity in a GxP-regulated Environment - Pauwels Consulting Academy

On Tuesday, December 6, 2016, our colleague Angelo Rossi, Senior Regulatory Compliance Consultant, gave an interesting presentation about “Data Integrity in a GxP-regulated Environment” at the Brussels Office of Pauwels Consulting in Diegem.
In his presentation, Angelo covered definitions and concepts of data integrity, the change in regulatory focus, lessons learned from recent FDA warning letters, importants highlights of regulations and guidelines. Angelo also presented a practical example of data integrity for a computerized system.
Please contact us at contact@pauwelsconsulting.com or +32 9 324 70 80 if you have any further questions regarding our consulting services in this area.
Surveyors are Here

Do you feel like there are more regulatory and accrediting surveys being conducted than ever before? Have you been the host of a CMS survey…or two… in the last several years? Do you feel like the surveys are becoming more onerous or prescriptive?
2019-07 James Sullivan - Case Study - Endomechanicals

James Sullivan: Case Study - Endomechanicals Analysis
RAC US Exam Questions

Now you don’t need to take any stress about the RAC US Exam. We provide you real exam questions along with updated Test Engine. You can pass your exam in first attempt with 100% passing assurance and money back guarantee. Get amazing flat 15% discount on RAC US exam and pass your RAC US Dumps Exam. Visit us for more information and RAC US Exam Questions.
https://www.certs4you.com/raps/rac-us-dumps.html
Data integrity - Regulatory Perspective and Challenges: 

Data Integrity: Regulatory Perspective and Challenges.
This presentation will present the importance of data integrity in the pharma industry with regards to the quality compliance and its impact on industry. This presentation also emphasizes on importance of regulatory affairs department in ensuring the data integrity and regulatory challenges.
How eSource Solutions are Impacting Clinical Research Sites, Patients, Regula...

Presented at DIA 2017 by Jules Mitchel, Mitchell Efros, and Jonathan Helfgott.
FDA Electronic Health Record Data in Clinical Research

Dr. Jules Mitchel, President of Target Health, delivered this presentation on the new FDA Guidance on the use of the electronic health record for clinical research at the North American eClinical Forum Autumn Meeting on October 2, 2018.
What's hot (20)
Top Regulatory Trends in the Life Sciences Industry in 2017

Top Regulatory Trends in the Life Sciences Industry in 2017
Routine Diagnostics | Advance Diagnostics | Pathology Lab | Blood Test | Molq

Routine Diagnostics | Advance Diagnostics | Pathology Lab | Blood Test | Molq
What Happens After Your Device is Approved? Collecting Data in the Real World

What Happens After Your Device is Approved? Collecting Data in the Real World
Regulatory Concerns When Running Virtual/Paperless Clinical Trials

Regulatory Concerns When Running Virtual/Paperless Clinical Trials
A Pharma/CRO Partnership in the Design and Execution of Paperless Clinical Tr...

A Pharma/CRO Partnership in the Design and Execution of Paperless Clinical Tr...
Data Integrity in a GxP-regulated Environment - Pauwels Consulting Academy

Data Integrity in a GxP-regulated Environment - Pauwels Consulting Academy
2019-07 James Sullivan - Case Study - Endomechanicals

2019-07 James Sullivan - Case Study - Endomechanicals
Data integrity - Regulatory Perspective and Challenges: 

Data integrity - Regulatory Perspective and Challenges:
How eSource Solutions are Impacting Clinical Research Sites, Patients, Regula...

How eSource Solutions are Impacting Clinical Research Sites, Patients, Regula...
FDA Electronic Health Record Data in Clinical Research

FDA Electronic Health Record Data in Clinical Research
Viewers also liked
Life Science Fast Track Faculty and Mentor Directory

Are you a physician, innovator, or entrepreneur with a commercial idea and ready to make it a fundable company? Join us for this full-day program to understand today’s unique challenges for life science, med device, and healthcare entrepreneurs starting companies and raising capital. Learn from and meet some of the leading life science investors, entrepreneurs, and early-stage experts in New England.
Building on the success of The Capital Network’s annual Fast Track Boot Camp for Entrepreneurs, this Life Science Fast Track is an intensive, interactive full-day seminar designed to provide entrepreneurs and innovators an in-depth understanding of what it takes to raise early stage capital for a life science startup.
Modern SEO Techniques Part 1:New SEO Methods for Medical Device Marketers

This two-part series on Modern SEO Techniques for Med Device Marketers can be used by any medical device marketer to understand SEO. Part one will cover modern link building and part two will cover user experience as a search engine ranking factor.
Search Engines. We love them when we’re looking for the hottest sushi restaurant in town. But when it comes to getting our business to rank at the top, they can be a pain in the wasabi.
Promoting Medical Devices Prior to FDA Approval

Most med device companies will agree with Tom Petty: the waiting is the hardest part. The time period from FDA submission to clearance or approval can feel like an eternity. But what can companies do to get hospitals and providers excited about their technology before launch?
View the presentation for more information on how to promote medical devices prior to FDA approval.
Visit us at www.GreyMatterMarketing.com
How to Start a Med Device Startup From Your Garage - Vancouver Edition

New and improved!
This is a Canada-localized, updated version of my talk on starting a medical device startup with no outside capital.
Given on March 31, 2016 at HxD Vancouver.
Med Device Vendors Have Big Opportunities in Health IT Software, Services, an...

If you’re in the medical device manufacturing or hardware sales business your revenue growth (CAGR) is under pressure like never before. You’re being asked to do more with less but you’re probably going to find that hard to accomplish because of one or more of the following challenges:
* Longer product development timelines caused by more FDA and other government regulations
* Increased demand by customers to have your devices deliver user experiences that are more like “consumer” devices such as cell phones and tablets
* Lower margins as a reaction to commodity competition (your sensor hardware business will be commoditized faster and faster over time)
* More complex and longer sales cycles because devices are now being approved for sale not by facilities and clinical executives alone but increasingly by CIOs and IT teams
* Increased cost of risk management and compliance caused by connectivity requirements
Any one of these challenges is difficult to meet but these days you’re probably being asked to meet more than one simultaneously. The solutions are not simple but the good news is that medical device manufacturers have many revenue generation opportunities today that can fund the new strategic imperatives you’ll need to put into place to meet the challenges listed above.
This briefing, presented by Netspective CEO Shahid Shah, describes some of the opportunities and how device vendors can take advantage of them.
Healthcare & Med Device - LiveWorx Recommended Agenda

LiveWorx17 Recommended Agenda – May 22-25, 2017 – Boston
LiveWorx is the premier Internet of Things (IoT) Business Event. Learn more-> http://ptc.co/yFPV30barOi
Top 10 lies of Venture Capitalists

From Garage Technology resource section
www.garage.com
Guy Kawasaki's Lecture Series
Venture Capital Term Sheets (April 7, 2009)

Presentation regarding the basics of venture capital term sheets
Startup Metrics (Ignite Velocity)

Startup Metrics for Pirates / Startup Metrics 101, presented at Ignite / Velocity Conference (June 2008)
SteadyBudget's Seed Funding Pitch Deck

This is the pitch deck we used in the spring of 2015 to pursue seed funding for our pay-per-click optimization platform SteadyBudget.com.
Zenpayroll Pitch Deck Template

The Pitch Deck That Helped A Startup Land $6 Million From The Founders Of Dropbox, Yelp, Box, Yammer, And Gmail
AppVirality.com - Investor Pitch Deck

AppVirality - Plug & Play growth hacking toolkit for Mobile apps.
We help app developers implement Uber/Lyft like in-app referrals under 30 minutes. No coding required.
Checkout - http://appvirality.com
How Wealthsimple raised $2M in 2 weeks

How Wealthsimple raised a $2M seed round in 2 weeks. Presentation from the Mobile Learning Lab hosted by Ward Technology Solutions.
500’s Demo Day Batch 16 >> Podozi

Contact: http://500demoday.co
Want MOAR? Subscribe for our newsletter & event updates: http://bit.ly/1IVVcP4
Swipes pitch deck for Beta Pitch 2013 Finals in Berlin

This was our pitch deck for Beta Pitch 2013 Finals in Berlin. http://swipesapp.com
The deck we used to raise $270k for our startup Castle

Castle (entercastle.com) is a Detroit-based real estate startup that lets rental owners put their properties on autopilot. In April 2015, we closed a $270,000 angel round using this deck.
Questions? Comments? I'd love to hear from you. Email me at max@entercastle.com.
The Deck We Used to Raise $1M Seed Round

Mapme Investor Deck.
The deck we originally used to raise our seed round of $1M. See the progress we've made at www.mapme.com. Welcome to try it out and create a map.
Any questions? I'm at ben@mapme.com
Viewers also liked (20)
Life Science Fast Track Faculty and Mentor Directory

Life Science Fast Track Faculty and Mentor Directory
Modern SEO Techniques Part 1:New SEO Methods for Medical Device Marketers

Modern SEO Techniques Part 1:New SEO Methods for Medical Device Marketers
How to Start a Med Device Startup From Your Garage - Vancouver Edition

How to Start a Med Device Startup From Your Garage - Vancouver Edition
Med Device Vendors Have Big Opportunities in Health IT Software, Services, an...

Med Device Vendors Have Big Opportunities in Health IT Software, Services, an...
Healthcare & Med Device - LiveWorx Recommended Agenda

Healthcare & Med Device - LiveWorx Recommended Agenda
Swipes pitch deck for Beta Pitch 2013 Finals in Berlin

Swipes pitch deck for Beta Pitch 2013 Finals in Berlin
The deck we used to raise $270k for our startup Castle

The deck we used to raise $270k for our startup Castle
Similar to 2015-CLSA-Report-Recent-Trends-in-FDA-Med-Device-Regulation-Final
The Minute: Volume 11 Issue 1 - Winter 2011

Crossroads: U.S. Medical Device Regulation vs. Innovation
The U.S. medical device industry is at a regulatory and potentially economic crossroad as the FDA continues to refine its 510(k) regulatory submission requirements and guidelines. Medical device manufacturers have been urging the FDA and Congress to expedite new product review processes to spur innovation and bring new medical technologies to market faster. However, supporters of stricter FDA regulations claim that a faster regulatory review process causes unsafe devices to enter the market.
As a result of numerous exchanges between both sides of the issue, CDRH (FDA) recently issued multiple updates to its initiatives for the 510(k) approval process. To shed light on key changes, we obtained the support of the office of Dr. Jeffrey Shuren MD JD, Director of CDRH and Dr. John Smith MD JD, of Hogan Lovells, a prominent international law firm with a medical regulatory specialty, on their interpretations of the 510(k) regulatory guidelines and the impact these guidelines will have on medical device manufacturers.
Listen to the Dr. John Smith podcast interview here:
http://youtu.be/iHVpwwXi7dY
The MarkeTech Group
502 Mace Blvd.
Davis, CA 95618
http://www.themarketechgroup.com
Washington Update: Changes at the FDA and Health Reform Implementation - Thom...

Washington Update: Changes at the FDA and Health Reform Implementation - Thom...marcus evans Network
Thomas Novelli, Medical Device Manufacturers Association - Speaker at the marcus evans Medical Device Manufacturing Summit June 2012, held in Las Vegas, NV delivered his presentation entitled Washington Update: Changes at the FDA and Health Reform ImplementationMercer Capital's Value Focus: Laboratory Services | Mid-Year 2015

Mercer Capital's Laboratory Services Industry newsletter provides perspective on valuation issues. Each newsletter also includes a macroeconomic trends, industry trends, and guideline public company metrics.
Shedding Some Light on the Insights Lurking in the PMA Database

May 28, 2016 marked forty years of modern day federal authority over medical devices in the U.S. Not only is this period brief in and of itself, but it’s also shorter by half compared to the duration of federal authority over pharmaceuticals, which began with the Food, Drug and Cosmetics Act of 1938. In the past several years the FDA has been the target of much criticism with regards to the approval of high-risk medical devices. Some of that critique is likely merited, but it is important to keep in mind that the medical device arm of the FDA is a work-in-progress that has had considerably less time to invent itself in the larger context of history.
This paper compares and contrasts different stakeholders’ perspectives and takes a deeper dive into the data. In doing so, this paper outlines practical changes and enhancements to the PMA database that can be carried out in the immediate present to increase transparency between the FDA and Industry.
The deeper dive also identifies several potential avenues for follow-on research, including PMAs that do not reach a positive conclusion and PMAs that are sponsored by early-stage and/or inexperienced filers. Insights from such research may hold the key to longer-term regulatory process improvements within the existing framework to promote high-risk medical device innovation and shorten these devices’ time-to-market without compromising the higher standards of the domestic regulatory system or the safety of patients.
Influencing Payer Coverage for Advanced Genomic Testing

Presentation at the San Diego Q1 Diagnostics Conference in Feb 2016 on Influencing Payer Coverage for Advanced Genomic Testing
FDA Draft 510(K) Guidance July 27, 2011

7/2711: FDA released draft guidance clarifying when medical device changes warrant new premarket review.
The value of early asset development and commercialization

Worldwide pharmaceutical R&D spend is increasing each year, and the competition for a share of that investment is becoming fiercer. Companies need to demonstrate the potential value of their asset in commercial as well as scientific and clinical terms.
The slides in this deck define and identify value from a financial, clinical and commercial perspective while also exploring how to derive value for patients.
Integrate RWE into clinical development

With greater application of RWE throughout the pharmaceutical
lifecycle, learnings are emerging that offer guidance for
approaches to derive the maximum value. This article captures
the author’s experience at a leading international biotech, with
insights for smoothing RWE assimilation into clinical
development and realizing the benefits it brings.
Pharma Uptoday Monthly Magazine - Volume 18; Issue: Sep 2015

To recap the August 2015 month's pharma highlights to Pharma Uptoday members, Monthly magazine Volume 18 has been released with the following content.
News Uptoday
New Guidance
Audit Findings
483 Observations
- 483 of PharMEDium Services, LLC (Outsourcing facility)
- 483 of "Walgreens Home Care, Inc. dba Walgreens Infusion Services
EU Non Compliance Report
- EU Non-Compliance Report: TXCELL - BESANCON, France Warning Letters
- Warning letter : Sipra Labs Limited, Hyderabad
- Warning letter : Mylan Laboratories Limited, India
Health Canada Non Compliance Report
- Procter & Gamble Inc., Canada.
Regulations of the Month
- Sec. 211.28 Personnel responsibilities (b) & (c)
- Sec. 211.42 Design and construction features (a) & (b)
Regulatory Compliance in Pharmaceutical DevelopmentGL.docx

Regulatory Compliance in Pharmaceutical Development:
GLP & GMP
Jeffrey G. Sarver, Ph.D.
MBC 3100
March 8, 2016
email questions to:
[email protected]
*
Food and Drug Administration (FDA)Agency of U.S. Department of Health and Human Services (HHS)Protect public health and provide essential public servicesOther HHS agencies include: CDC, NIH, Medicare/MedicaidFDA responsible for assuring safety and efficacy of: Human Drugs - Center for Drug Evaluation and Research (CDER)Veterinary Drugs - Center for Veterinary Medicine (CVM)Biological Agents - Center for Biologic Evaluation and Research (CBER)Medical Devices - Center for Devices and Radiological Health (CDRH)Food/Supplements/Cosmetics - Center for Food Safety and Applied Nutrition (CFSAN)
Manufacture
Market Drug
NDA
FDA Review
Clinical
Trials
I, II, III
IND
FDA Review
Preclinical
Testing
Drug
Discovery
Drug Development/Approval ProcessIND – Investigational New Drug application (3-6 yr, $5M-$10M)FDA Approval → Proceed into Clinical Trials (~30% from preclinical tests)Clinical Hold → Collect More data or End DevelopmentNDA – New Drug Application (9-12 yr, $500M-$1B)FDA Approval → Drug Enters Market (~8% from preclinical tests)Not Approved → More Data or Adjust Application or End Development
Basic
Research
+
Target
Discovery
Preclinical Testing RequirementsMechanism of Action (in vitro) and Efficacy (in vivo)General Toxicology: Single and Repeated Dose (in vivo)Genotoxicity/Mutagenicity (in vitro/in vivo)Carcinogenicity (in vivo)Reproductive Toxicology/Teratology (in vivo)ADME (in vitro)/Pharmacokinetics (in vivo)Additional Safety TestingCore: Cardiovascular (hERG), Respiratory, CNSOther tests as needed based on structure, mechanism, general tox
Additional Information for INDChemistry, Manufacturing, and Controls (CMC)Structure, physical propertiesSynthetic method and scale-upPurity, identification of impuritiesDosage form/route, formulation, preparation, packagingClinical Study ProtocolsPrevious Human Experience (if available)
FDA Guidance Documents
Information on suggested: testing methods, analyzing and summarizing data
Can be found at: http://www.fda.gov/RegulatoryInformation/Guidances/Searching can be difficult/tedious, use appropriate filters:Product → Drugs (or Biologics)FDA Organization → CDER (or CBER)Document Type → Guidance DocumentsExample Guidance Documents available on Blackboard:Genotoxicity TestingCardiotoxicity (hERG) TestingChemistry, Manufacturing, and Controls (CMC) for Phase I DrugMaximum Safe Starting Dose for Clinical Testing
International Council on Harmonization (ICH)Harmonize procedures for evaluating/reporting safety, efficacy, CMC in multiple regions/countriesRegulators and industry representatives from participating regions collaborate to generate internationally acceptable guidelinesImprove efficiency of drug testing/reporting requirements for approval in multiple countriesOriginally Europe, Japan, U.S.Other countries adopting IC ...
Practical Guide - www.devicematters.com

A Practical Guide presentation created for DeviceMatters (www.devicematters.com) by vr4U Global Presentation Team
The 21st Century Cures Act a focus on Title III Subtitle F – Medical Device I...

The 21st century cures act a focus on title iii subtitle f – medical device innovations
Unique Device Identification and GS1: Defining Elements in the Future of Glob...

This white paper, as the title suggests, is about new national and international mandates for a global standard to be used
in the Unique Device Identification (UDI) of medical devices and other healthcare products. It examines the global trading
opportunities, on an enormous scale, that can be captured by early adopters or forfeited by default by those who wait,
dismiss the idea, or discount the powerful market and competitive forces that UDI developments are driving. Also offered
is a starting blueprint for regulatory compliance professionals, packaging engineers, C-suite executives and manufacturing experts who agree the time to start meeting the UDI opportunity has clearly arrived.
Similar to 2015-CLSA-Report-Recent-Trends-in-FDA-Med-Device-Regulation-Final (20)
CHI-Report-Taking-the-Pulse-of-Medical-Device-Regulation-Innovation_Oct-2014

CHI-Report-Taking-the-Pulse-of-Medical-Device-Regulation-Innovation_Oct-2014
CLSA-BCG-Report-Tracking-FDA-Drug-Review-Performance-March-2016-Final2

CLSA-BCG-Report-Tracking-FDA-Drug-Review-Performance-March-2016-Final2
Washington Update: Changes at the FDA and Health Reform Implementation - Thom...

Washington Update: Changes at the FDA and Health Reform Implementation - Thom...
Future Challenges of Clinical Development; a View from the CRO - Hani Zaki

Future Challenges of Clinical Development; a View from the CRO - Hani Zaki
Future Challenges of Clinical Development; a View from the CRO - Hani Zaki

Future Challenges of Clinical Development; a View from the CRO - Hani Zaki
Mercer Capital's Value Focus: Laboratory Services | Mid-Year 2015

Mercer Capital's Value Focus: Laboratory Services | Mid-Year 2015
Shedding Some Light on the Insights Lurking in the PMA Database

Shedding Some Light on the Insights Lurking in the PMA Database
Influencing Payer Coverage for Advanced Genomic Testing

Influencing Payer Coverage for Advanced Genomic Testing
The value of early asset development and commercialization

The value of early asset development and commercialization
Pharma Uptoday Monthly Magazine - Volume 18; Issue: Sep 2015

Pharma Uptoday Monthly Magazine - Volume 18; Issue: Sep 2015
Regulatory Compliance in Pharmaceutical DevelopmentGL.docx

Regulatory Compliance in Pharmaceutical DevelopmentGL.docx
The 21st Century Cures Act a focus on Title III Subtitle F – Medical Device I...

The 21st Century Cures Act a focus on Title III Subtitle F – Medical Device I...
Unique Device Identification and GS1: Defining Elements in the Future of Glob...

Unique Device Identification and GS1: Defining Elements in the Future of Glob...
More from Will Zasadny
More from Will Zasadny (8)
CLSA & PwC 2017 CA Life Sciences Industry Report Final

CLSA & PwC 2017 CA Life Sciences Industry Report Final
FINAL-CHI-Report-Innovation-in-Hepatitis-C-Treatment-July-2014

FINAL-CHI-Report-Innovation-in-Hepatitis-C-Treatment-July-2014
2015-CHI-PwC-California-Biomedical-Industry-Report_Final

2015-CHI-PwC-California-Biomedical-Industry-Report_Final
2015-CLSA-Report-Recent-Trends-in-FDA-Med-Device-Regulation-Final
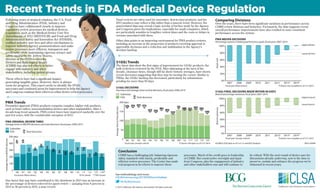
- 1. PAUL HORNSOURCE:FDA data as of 5.31.15 and BCG Analysis Recent Trends in FDA Medical Device Regulation 510(k) Trends The latest data show the first signs of improvement for 510(k) products, the bulk of devices reviewed by the FDA. After plateauing at the turn of the decade, clearance times, though still far above historic averages, have shown recent decreases suggesting that they may be turning the corner. Similar to PMAs, the 510(k) backlog has decreased, particularly for submissions pending for more than 90 days. Other data points to an improving environment for PMA product reviews, including an increase in the proportion of products receiving approval or approvable decisions and a reduction and stabilization in the Agency’s decision backlog. Submitter FDA Total decisionsXX ’13*’12*’11*’10’09*’08’07’06’05’04’03’02’01’00 ’14* *Cohorts still open as of 3.31.2014 510(k) DECISIONS Decisions and average times to final decisions,fiscal years 2000-2014 Averagetimetofinaldecision 200 150 100 50 0 days 4204 4254 3394 4322 4225 3632 3550 3853 3656 3348 4101 3880 3833 3992 3865 Panel reviews are often used for innovative, first-in-class products, and the 2013 numbers may reflect a blip rather than a nascent trend. However, the panel-related data may reveal a topic worthy of further study by the Agency and Congress given the implications, especially for small companies, which are particularly sensitive to lengthier review times and the costs or delays in revenue associated with them. Following years of strained relations, the U.S. Food and Drug Administration (FDA), industry and Congress have collaborated closely to improve regulatory review processes for medical devices. Legislation, such as the Medical Device User Fee Amendments of 2012 (MDUFA III) and Food and Drug Administration Safety and Innovation Act (FDASIA), codified industry user fees and other mechanisms to improve industry-Agency communications and make review processes more efficient, transparent and predictable while maintaining rigorous science and safety standards. Dr. Jeffrey Shuren, director of the FDA’s Center for Devices and Radiological Health (CDRH) has also led efforts to better engage with industry and other stakeholders, including patient groups. These efforts have had a significant impact, generating tangible gains. However, there is always room for progress. This report seeks to identify the FDA’s successes and continued areas for improvement to help the Agency and Congress continue their efforts to refine device review processes. One factor that may have contributed to the slowdown in 2013 was an increase in the percentage of devices referred for panel review — jumping from 8 percent in 2012 to 36 percent in 2013, a near record. 500 400 300 200 100 0 ’13*’12’11’10’09’08’07’06’05’04’03’02’01’00 ’14** AveragetimetoMDUFAdecision days Submitter FDA Total decisionsXX PMA ORIGINAL REVIEW TIMES Average times to MDUFA decisions and total decisions,fiscal years 2000-2014 *97% closed **79% closed 65 66 41 43 53 47 39 35 30 32 43 43 24 28 22 PMA Trends Premarket approval (PMA) products comprise complex, higher risk products, such as heart valves, neuromodulation devices and other implantables. After a decade-long trend upwards, PMA review times have improved markedly over the past few years, with the considerable exception of 2013. Comparing Divisions Over the years, there have been significant variations in performance across CDRH review divisions and branches. Fortunately, the data suggests recent overall performance improvements have also resulted in more consistent performance across the system. 2013*201220112010200920082007 2014* PMA MDUFA DECISIONS Percentage variance meeting performance goals,fiscal years 2007-2014 100% 80% 60% 40% 20% 0% *Cohorts not complete as of 5.31.2015 Goalsmet *Cohorts not complete as of 5.31.2015 510(k) FINAL DECISIONS MADE WITHIN 90 DAYS Branch percentage variances,fiscal years 2007-2014 2013*2012*201120102009*20082007 2014* Branchvariances 100% 80% 60% 40% 20% 0% Highest branch Lowest branch Highestdivision Lowest division See methodology and more: CALifeSciences.org/2015FDADeviceUpdate @CALifeSciences © 2015 California Life Sciences Association.All rights reserved. Conclusion CDRH has a challenging job: balancing rigorous safety standards with timely, predictable and efficient review processes. The Center has made real progress in its efforts to improve these processes. Much of the credit goes to leadership of CDRH. But constructive oversight and input from Congress, plus the engagement of industry and other stakeholders was and will continue to be critical. With the next round of device user fee discussions already underway, now is the time to preserve, sustain and enhance the progress we’ve witnessed in recent years. Fiscal year (filed cohort) Fiscal year (receipt cohort) Fiscal year (filed cohort) Fiscal year (receipt cohort)