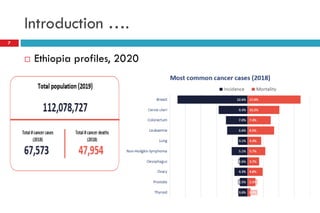
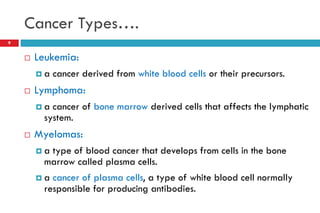
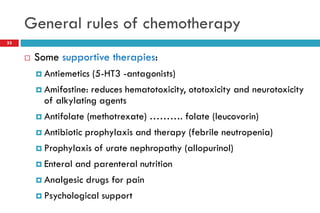
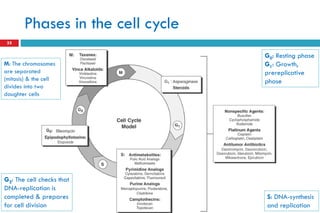
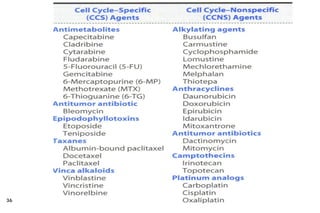
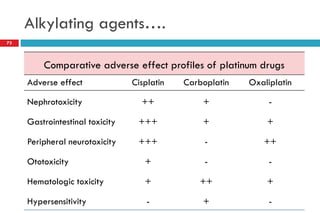
This document provides information about cancer treatment and chemotherapy. It discusses the types and characteristics of cancer cells, principles of chemotherapy including drug combinations and treatment cycles, methods for calculating chemotherapy doses, and types of chemotherapeutic drugs including antimetabolites like 5-FU and capecitabine. The document contains details on the mechanisms of action, administration methods, and side effects of specific chemotherapy drugs.


![Introduction ….
3
The changes are the result of the interaction b/n a person's
genetic factors and external agents;
physical carcinogens [eg, UV and ionizing radiation]
chemical carcinogens [asbestos, components of tobacco smoke,
food contaminants)
biological carcinogens, such as infections from certain viruses,
bacteria, or parasites
Ageing ….. also a factor for the development of cancer.](https://image.slidesharecdn.com/01-caintro2016-240226082451-23f79bcd/85/01-Ca-Intro_2016-pdf-pharmacy-student-ca-3-320.jpg)