ENZYMES CATALYZE
- 1. ENZYMES Enzymes are biocatalysts – the catalysts of life. A catalyst is defined as a substance that increases the velocity or rate of a chemical reaction without itself undergoing any change in the overall process. Enzymes may be defined as biocatalysts synthesized by living cells. They are protein in nature (exception – RNA acting as ribozyme), colloidal and thermolabile in character, and specific in their action. Berzelius in 1836 coined the term catalysis (Greek : to dissolve). In 1878, Kuhne used the word enzyme (Greek : in yeast) to indicate the catalysis taking place in the biological systems. NOMENCLATURE AND CLASSIFICATION The suffix-ase was added to the substrate for naming the enzymes e.g. lipase acts on lipids; nuclease on nucleic acids; lactase on lactose. Enzymes are sometimes considered under two broad categories : (a) Intracellular enzymes – They are functional within cells where they are synthesized. (b) Extracellular enzymes – These enzymes are active outside the cell; The International Union of Biochemistry (IUB) appointed an Enzyme Commission in 1961. Since 1964, the IUB system of enzyme classification has been in force. Enzymes are divided into six major classes.
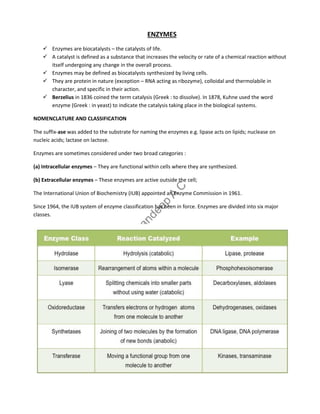
- 2. 1. Oxidoreductases : Enzymes involved in oxidation-reduction reactions. 2. Transferases : Enzymes that catalyse the transfer of functional groups. 3. Hydrolases : Enzymes that bring about hydrolysis of various compounds. 4. Lyases : Enzymes specialised in the addition or removal of water, ammonia, CO2 etc. 5. Isomerases : Enzymes involved in all the isomerization reactions. 6. Ligases : Enzymes catalysing the synthetic reactions (Greek : ligate—to bind) where two molecules are joined together and ATP is used. PROPERTIES OF ENZYMES All the enzymes are invariably proteins. Each enzyme has its own tertiary structure and specific conformation which is very essential for its catalytic activity. The functional unit of the enzyme is known as holoenzyme The enzyme is made up of apoenzyme (the protein part) and a coenzyme (non-protein organic part). Holoenzyme Apoenzyme + Coenzyme (active enzyme) (protein part) (non-protein part) FACTORS AFFECTING ENZYME ACTIVITY 1. Concentration of enzyme - As the concentration of the enzyme is increased, the velocity of the reaction proportionately increases. 2. Concentration of substrate - Increase in the substrate concentration gradually increases the velocity of enzyme reaction within the limited range of substrate levels.
- 3. 3. Effect of temperature - Velocity of an enzyme reaction increases with increase in temperature up to a maximum and then declines. A bell-shaped curve is usually observed. 4. Effect of pH - Increase in the hydrogen ion concentration (pH) considerably influences the enzyme activity and a bell-shaped curve is normally obtained. 5. Effect of product concentration - The accumulation of reaction products generally decreases the enzyme velocity. 6. Effect of activators - Some of the enzymes require certain inorganic metallic cations like Mg2+, Mn2+, Zn2+, Ca2+, Co2+, Cu2+, Na+, K+ etc. for their optimum activity. Rarely, anions are also needed for enzyme activity. 7. Effect of time - Under ideal and optimal conditions (like pH, temperature etc.), the time required for an enzyme reaction is less. Variations in the time of the reaction are generally related to the alterations in pH and temperature. ACTIVE SITE The active site (or active centre) of an enzyme represents as the small region at which the substrate(s) binds and participates in the catalysis. Salient features of active site The existence of active site is due to the tertiary structure of protein resulting in three- dimensional native conformation.
- 4. The active site is made up of amino acids (known as catalytic residues) which are far from each other in the linear sequence of amino acids. Active sites are regarded as clefts or crevices or pockets occupying a small region in a big enzyme molecule. The active site is not rigid in structure and shape. It is rather flexible to promote the specific substrate binding. Generally, the active site possesses a substrate binding site and a catalytic site. The coenzymes or cofactors on which some enzymes depend are present as a part of the catalytic site. The substrate(s) binds at the active site by weak noncovalent bonds. Enzymes are specific in their function due to the existence of active sites. ENZYME INHIBITION Enzyme inhibitor is defined as a substance which binds with the enzyme and brings about a decrease in catalytic activity of that enzyme. The inhibitor may be organic or inorganic in nature. There are three broad categories of enzyme inhibition 1. Reversible inhibition. 2. Irreversible inhibition. 3. Allosteric inhibition. 1. Reversible inhibition The inhibitor binds non-covalently with enzyme and the enzyme inhibition can be reversed if the inhibitor is removed. The reversible inhibition is further sub-divided into I. Competitive inhibition II. Non-competitive inhibition Competitive inhibition : The inhibitor (I) which closely resembles the real substrate (S) is regarded as a substrate analogue. The inhibitor competes with substrate and binds at the active site of the enzyme but does not undergo any catalysis. As long as the competitive inhibitor holds the active site, the enzyme is not available for the substrate to bind. The relative concentration of the substrate and inhibitor and their respective affinity with the enzyme determines the degree of competitive inhibition. In competitive inhibition, the Km value increases whereas Vmax remains unchanged Non-competitive inhibition : The inhibitor binds at a site other than the active site on the enzyme surface. This binding impairs the enzyme function. The inhibitor has no structural resemblance with the substrate. However, there usually exists a strong affinity for the inhibitor to bind at the second site. In fact, the inhibitor does not interfere with the enzyme-substrate binding. But the catalysis is prevented, possibly due to a distortion in the enzyme conformation. Irreversible inhibition - The inhibitors bind covalently with the enzymes and inactivate them, which is irreversible. These inhibitors are usually toxic substances that poison enzymes.
- 5. COENZYMES The non-protein, organic, low molecular weight and dialysable substance associated with enzyme function is known as coenzyme. The protein part of the enzyme, on its own, is not always adequate to bring about the catalytic activity. Many enzymes require certain non- protein small additional factors, collectively referred to as cofactors for catalysis. Coenzymes are second substrates : Coenzymes are often regarded as the second substrates or co- substrates, since they have affinity with the enzyme comparable with that of the substrates. Coenzymes participate in various reactions involving transfer of atoms or groups like hydrogen, aldehyde, keto, amino, acyl, methyl, carbon dioxide etc. MECHANISM OF ENZYME ACTION Enzymes lower activation energy : The energy required by the reactants to undergo the reaction is known as activation energy. The reactants when heated attain the activation energy. The catalyst (or the enzyme in the biological system) reduces the activation energy and this causes the reaction to proceed at a lower temperature. The enzyme lowers energy barrier of reactants, thereby making the reaction go faster. The prime requisite for enzyme catalysis is that the substrate (S) must combine with the enzyme (E) at the active site to form enzyme- substrate complex (ES) which ultimately results in the product formation (P). MECHANISM OF ENZYME CATALYSIS The formation of an enzyme-substrate complex (ES) is very crucial for the catalysis to occur, and for the product formation. It is estimated that an enzyme catalysed reaction. The enhancement in the rate of the reaction is mainly due to four processes : 1. Acid-base catalysis; 2. Substrate strain; 3. Covalent catalysis; 4. Entropy effects. Isoenzyme - The multiple forms of an enzyme catalyzing the same reaction are isoenzymes or isozymes. They, however, differ in their physical and chemical properties which include the structure, electrophoretic and immunological properties, Km and Vmax values, pH optimum, relative susceptibility to inhibitors and degree of denaturation.