Report
Share
Recommended
Recommended
More Related Content
Similar to stereomers.pptx
Similar to stereomers.pptx (20)
Stereochemistry Lecture.pptx by university of sargodha

Stereochemistry Lecture.pptx by university of sargodha
Stereoisomers are isomeric molecules that have th.pdf

Stereoisomers are isomeric molecules that have th.pdf
Constitional You can tell if a pair of molecules.pdf

Constitional You can tell if a pair of molecules.pdf
Structural isomers and stereoisomers configuration | types of isomers | isome...

Structural isomers and stereoisomers configuration | types of isomers | isome...
Recently uploaded
Cara menggugurkan kandungan paling ampuh 08561234742

Cara menggugurkan kandungan paling ampuh 08561234742Jual obat penggugur 08561234742 Cara menggugurkan kandungan 08561234742
Abortion pills in Kuwait (+918133066128) Abortion clinic pills in Kuwait

Abortion pills in Kuwait (+918133066128) Abortion clinic pills in KuwaitAbortion pills in Kuwait Cytotec pills in Kuwait
Abortion pills in Abu Dhabi ௵+918133066128௹Un_wandted Pregnancy Kit in Dubai UAE

Abortion pills in Abu Dhabi ௵+918133066128௹Un_wandted Pregnancy Kit in Dubai UAEAbortion pills in Kuwait Cytotec pills in Kuwait
Recently uploaded (20)
Leadership Style - Code and Rapid Response Workshop

Leadership Style - Code and Rapid Response Workshop
The 2024 Outlook for Older Adults: Healthcare Consumer Survey

The 2024 Outlook for Older Adults: Healthcare Consumer Survey
Cara menggugurkan kandungan paling ampuh 08561234742

Cara menggugurkan kandungan paling ampuh 08561234742
Pulse Check Decisions - RRT and Code Blue Workshop

Pulse Check Decisions - RRT and Code Blue Workshop
Abortion pills in Kuwait (+918133066128) Abortion clinic pills in Kuwait

Abortion pills in Kuwait (+918133066128) Abortion clinic pills in Kuwait
Jual obat aborsi Tuban Wa 081225888346 obat aborsi Cytotec asli Di Tuban

Jual obat aborsi Tuban Wa 081225888346 obat aborsi Cytotec asli Di Tuban
Session-5-Birthing-Practices-Breastfeeding (1).ppt

Session-5-Birthing-Practices-Breastfeeding (1).ppt
Abortion pills in Abu Dhabi ௵+918133066128௹Un_wandted Pregnancy Kit in Dubai UAE

Abortion pills in Abu Dhabi ௵+918133066128௹Un_wandted Pregnancy Kit in Dubai UAE
ITM HOSPITAL The hospital has also been recognised as the best emerging hosp...

ITM HOSPITAL The hospital has also been recognised as the best emerging hosp...
Top 20 Famous Indian Female Pornstars Name List 2024

Top 20 Famous Indian Female Pornstars Name List 2024
Obat Aborsi Makassar WA 085226114443 Jual Obat Aborsi Cytotec Asli Di Makassar

Obat Aborsi Makassar WA 085226114443 Jual Obat Aborsi Cytotec Asli Di Makassar
End of Response issues - Code and Rapid Response Workshop

End of Response issues - Code and Rapid Response Workshop
INTERNATIONAL HEALTH AGENCIES BY ANUSHRI SRIVASTAV.pptx

INTERNATIONAL HEALTH AGENCIES BY ANUSHRI SRIVASTAV.pptx
Navigating Conflict in PE Using Strengths-Based Approaches

Navigating Conflict in PE Using Strengths-Based Approaches
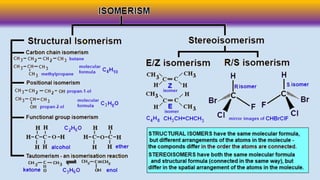
stereomers.pptx
- 2. Chiral Molecules: a) Attached with four different groups The difference between chiral and achiral molecules can be explained on the basis of the plane of symmetry. If all the attached group to the central carbon atom are different then there is no plane of symmetry. Such a molecule is known as a chiral molecule. If all the group attached to the central carbon atom are not different then there exist plane of symmetry. Such molecules are called achiral molecules. It is clear that only molecule having chiral centre will show optical isomerism.
- 4. Optical activity is the ability of a chiral molecule to rotate the plane of plane-polairsed light, measured using a polarimeter. Optical Isomers: Optical isomers are two compounds which contain the same number and kinds of atoms, and bonds (i.e., the connectivity between atoms is the same), and different spatial arrangements of the atoms, but which have non-superimposable mirror images. Each non-superimposable mirror image structure is called an enantiomer.
- 5. Generally defined, stereoisomers are isomers that have the same composition but that differ in the orientation of those parts in space. There are two kinds of stereoisomers: enantiomers and diastereomers
- 6. Enantiomers are chiral molecules that are mirror images of one another. Furthermore, the molecules are non- superimposable on one another. This means that the molecules cannot be placed on top of one another and give the same molecule. Chiral molecules with one or more stereocenters can be enantiomers
- 8. Diastereomers are stereoisomers that are not mirror images of one another and are non-superimposable on one another. Stereoisomers with two or more stereocenters can be diastereomers. It is sometimes difficult to determine whether or not two molecules are diastereomers