Verification of fruendlich adsorption isotherm of coconut shell carbon
•Download as DOCX, PDF•
0 likes•77 views
The graph of log(x/m) v/s Log C is a straight line, where log k is intercept and 1/n is the slope. The graph of adsorption at constant temperature is known as Freundlich adsorption isotherm.
Report
Share
Report
Share
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
How to interpret arterial blood gas ( ABGs ) Chart

How to interpret arterial blood gas ( ABGs ) Chart
Department of biochemistry cell ad molecular biology (1)

Department of biochemistry cell ad molecular biology (1)
Lect. 8. application of nernst distribution law copy - copy

Lect. 8. application of nernst distribution law copy - copy
Similar to Verification of fruendlich adsorption isotherm of coconut shell carbon
Decolorization of Acidic Dye by Charcoal from Coffee Grounds
Decolorization of Acidic Dye by Charcoal from Coffee Grounds

Decolorization of Acidic Dye by Charcoal from Coffee GroundsSchool Vegetable Gardening - Victory Gardens
Similar to Verification of fruendlich adsorption isotherm of coconut shell carbon (20)
Determination of relative strength between h cl and urea hydrochloride for hy...

Determination of relative strength between h cl and urea hydrochloride for hy...
Decolorization of Acidic Dye by Charcoal from Coffee Grounds

Decolorization of Acidic Dye by Charcoal from Coffee Grounds
IIT_Chemistry_manual_B_TEChemistry for engineering

IIT_Chemistry_manual_B_TEChemistry for engineering
Determination_of_Nitrogen_in_the_Wastewater_By_Kjeldahl_Method-1.pdf

Determination_of_Nitrogen_in_the_Wastewater_By_Kjeldahl_Method-1.pdf
IRJET- Kinetic Study of the Reaction of 5-Chlorosalicyaldehyde with M- To...

IRJET- Kinetic Study of the Reaction of 5-Chlorosalicyaldehyde with M- To...
Determination of equivalence conductance, degree of dissociation and dissocia...

Determination of equivalence conductance, degree of dissociation and dissocia...
Determination of equivalence conductance, degree of dissociation and dissocia...

Determination of equivalence conductance, degree of dissociation and dissocia...
More from Mithil Fal Desai
More from Mithil Fal Desai (20)
How long could one survive in a perfectly airtight room.docx

How long could one survive in a perfectly airtight room.docx
Writing Third Year Project Report: Some Handy Guidelines

Writing Third Year Project Report: Some Handy Guidelines
Determine the strength in grams per litre of a given AgNO3 solution being pro...

Determine the strength in grams per litre of a given AgNO3 solution being pro...
Estimation of water of crystallization in mohr’s salt by titrating with stand...

Estimation of water of crystallization in mohr’s salt by titrating with stand...
Recently uploaded
God is a creative God Gen 1:1. All that He created was “good”, could also be translated “beautiful”. God created man in His own image Gen 1:27. Maths helps us discover the beauty that God has created in His world and, in turn, create beautiful designs to serve and enrich the lives of others.
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...christianmathematics
Recently uploaded (20)
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
Micro-Scholarship, What it is, How can it help me.pdf

Micro-Scholarship, What it is, How can it help me.pdf
Mixin Classes in Odoo 17 How to Extend Models Using Mixin Classes

Mixin Classes in Odoo 17 How to Extend Models Using Mixin Classes
On National Teacher Day, meet the 2024-25 Kenan Fellows

On National Teacher Day, meet the 2024-25 Kenan Fellows
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
Measures of Central Tendency: Mean, Median and Mode

Measures of Central Tendency: Mean, Median and Mode
Role Of Transgenic Animal In Target Validation-1.pptx

Role Of Transgenic Animal In Target Validation-1.pptx
Unit-V; Pricing (Pharma Marketing Management).pptx

Unit-V; Pricing (Pharma Marketing Management).pptx
Verification of fruendlich adsorption isotherm of coconut shell carbon
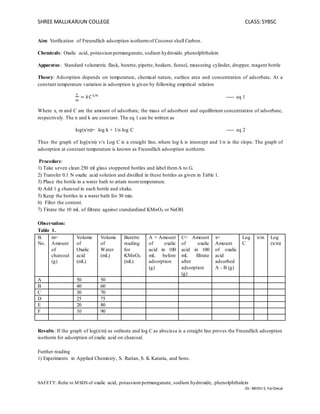
- 1. SHREE MALLIKARJUN COLLEGE CLASS:SYBSC SAFETY: Refer to MSDS of oxalic acid, potassiumpermanganate, sodium hydroxide, phenolphthalein -Dr. Mithil S. FalDesai Aim: Verification of Freundlich adsorption isothermof Coconut shell Carbon. Chemicals: Oxalic acid, potassiumpermanganate, sodium hydroxide, phenolphthalein Apparatus: Standard volumetric flask, burette, pipette, beakers, funnel, measuring cylinder, dropper, reagent bottle Theory: Adsorption depends on temperature, chemical nature, surface area and concentration of adsorbate. At a constant temperature variation in adsorption is given by following empirical relation 𝑥 𝑚 = 𝑘𝐶1/𝑛 ----- eq 1 Where x, m and C are the amount of adsorbate, the mass of adsorbent and equilibrium concentration of adsorbate, respectively. The n and k are constant. The eq 1 can be written as log(x/m)= log k + 1/n log C ----- eq 2 Thus the graph of log(x/m) v/s Log C is a straight line, where log k is intercept and 1/n is the slope. The graph of adsorption at constant temperature is known as Freundlich adsorption isotherm. Procedure: 1) Take seven clean 250 ml glass stoppered bottles and label themA to G. 2) Transfer 0.1 N oxalic acid solution and distilled in these bottles as given in Table 1. 3) Place the bottle in a water bath to attain roomtemperature. 4) Add 1 g charcoal in each bottle and shake. 5) Keep the bottles in a water bath for 30 min. 6) Filter the content. 7) Titrate the 10 mL of filtrate against standardized KMnO4 or NaOH. Observation: Table 1. B. No. m= Amount of charcoal (g) Volume of Oxalic acid (mL) Volume of Water (mL) Burette reading for KMnO4 (mL) A = Amount of oxalic acid in 100 mL before adsorption (g) C= Amount of oxalic acid in 100 mL filtrate after adsorption (g) x= Amount of oxalic acid adsorbed A - B (g) Log C x/m Log (x/m) A 50 50 B 40 60 C 30 70 D 25 75 E 20 80 F 10 90 Results: If the graph of log(x/m) as ordinate and log C as abscissa is a straight line proves the Freundlich adsorption isotherm for adsorption of oxalic acid on charcoal. Further reading 1) Experiments in Applied Chemistry, S. Rattan, S. K Kataria, and Sons.