العضوية مشتقات الهيدروكربونات استاذ رمضان غلاب تالتة ثانوي
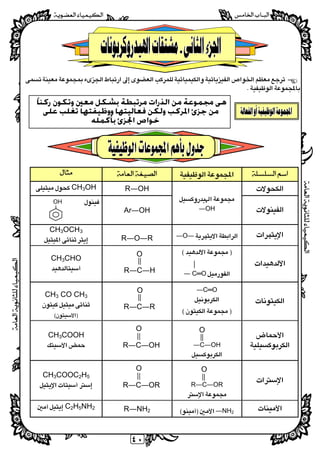
- 1. ٤٠ ﺍﻟﺬﺭﺍﺕ ﻣﻦ ﳎﻤﻮﻋﺔ ﻫﻰﹰﺎﺭﻛﻨ ﻭﺗﻜﻮﻥ ﻣﻌﲔ ﺑﺸﻜﻞ ﻣﺮﺗﺒﻄﺔ ﻣﻦﺟﺰﺉﻭﻭﻇﻴﻔﺘ ﻓﻌﺎﻟﻴﺘﻬﺎ ﻭﻟﻜﻦ ﺍﳌﺮﻛﺐﻬﺎﺗﻐﻠﺐﻋﻠﻰ ﺧﻮﺍﺹﺑﺄﻛﻤﻠﻪ ﺍﳉﺰﺉ R―OHCH3OH OH― Ar―OH ―O― R―O―R CH3OCH3 ― C═O CH3CHO ―C═O CH3 CO CH3 C―OH― CH3COOH R―C―OR CH3COOC2H5 ―NH2 R―NH2 C2H5NH2 R―C―OR O R―C―OH O R―C―H O R―C―R O O OH O
- 2. ٤١ ﺟﺰ ﲢﺘﻮﻯ ﻋﻀﻮﻳﺔ ﻣﺮﻛﺒﺎﺕ ﻫﻰﳎﻤﻮﻋﺔ ﻋﻠﻰ ﺎﻳﺌﺎﺃﻛﺜﺮ ﺃﻭ ﻣﻦﺍﳍﻴﺪﺭﻭﻛﺴﻴﻞ ﳎﻤﻮﻋﺎﺕ OHR─OH OHAr─OH R―OHH―OH Ar―OHH―OH ― C ―OH - H + R - H + Ar H R―OHR―H Ar―OHAr―H - H + OH - H + OH CH3─OH CH2―CH2 C2H4(OH)2 │ │ OH OH CH2―CH─CH2 C3H5(OH)3 ││ │ OHOH OH CH2―(CHOH)4―CH2 C6H8(OH)6 │ │ OH OH R―OHAr―OH CH3OH OH
- 3. ٤٢ ++ """ =" CH3CH2CH2OH CH3 – C – CH2CH3 OH H *C2H5OH R – C – OH H H H * - CH3 – C – OH H H R – C – OH R H CH3 – C – OH CH3 H H * --- R – C – OH R R CH3 – C – OH CH3 CH3 CH3 –CH2– CH – CH3 OH CH– CH2 – OH CH3 CH3 CH3 –CH2– C – OH CH3 CH3 C2H4(OH)2 CH3 –CH2– C – OH CH3 CH3 H I
- 4. ٤٣ OH CH3―CH2―CH2―CH2―CH2―OH- CH3―CH2―CH2―CH―CH3--- CH3―CH2―CH―CH2―CH3 CH3―CH2―CH― CH3 CH3 ―CH ―CH3 – ––– – C12H22O11 + H2O C6H12O6 + C6H12O6 2C2H5OH + 2CO2C6H12O6 – C2H4 CH2═CH2 + HOH CH3CH2OH OH OH H2SO4 OH OH CH3
- 5. ٤٤ CH3CH═CH2 + HOH CH3―C―CH3 - CH3―C―C―CH3+ HOHCH3―C ═ C―CH3 ٢--٢- - - RX + KOH R―OH + KX CH3Cl + KOH(aq) CH3OH + KCl CH3―CH2―Br + K OH(aq) CH3―CH2―OH + KBr CH3―CH―Br + K OH(aq) CH3―CH―OH + KBr - - OH CH3 CH3 OH H2SO4 H2SO4 CH3 CH3 H I H I H I I H
- 6. ٤٥ CH3―C―Cl + K OH(aq) CH3―C―OH + KCl << R─C─Br + KOH R─C─OH + KBr R─C─Br + KOH R─C─OH + KBr R─C─Br + KOH R─C─OH + KBr –––– ٢–ﺒﻴﻭﺘﺎﻨﻭل CH3 CH2─C─CH3 + KOH CH3 CH2─C─CH3 + KBr ٢–ﻣﯿﺜﯿﻞ–٢–ﺑﻨﺘﺎﻧﻮل CH3 CH2 CH2─C─CH3 + KOH CH3 CH2 CH2─C─CH3 + KCl – CH3 CH3 H │ │ OH H │ │ Br │ Cl CH3 │ CH3 │ │ OH CH3 CH3 H H H H ﻣﺎﺋﯿﺔ R H ﻣﺎﺋﯿﺔ R H R R ﻣﺎﺋﯿﺔ R R
- 7. ٤٦ – 2C2H5OH + 2Na 2C2H5ONa + H2 ↑ O─H C2H5O Na + HO H C2H5OH + NaOH R ― C ― O H H │ │ H HCl , HBr , HI H2SO4 أ(ﻋﻨﺪ١٨٠ب ْ(ﻋﻨﺪ١٤٠ْ R─ O H
- 8. ٤٧ – CH3CO OH + H OC2H5 CH3COOC2H5 +H2O :OHH O18 O16 OH ZnCl2 C2H5OH + HCl C2H5Cl + H2O HOH "ﺍﳌﺮﻛﺰ ﺍﻟﻜﱪﻳﺘﻴﻚ ﲝﻤﺾ ﳏﻤﻀﺔ ﻣﻨﻬﻤﺎ ﻛﻞ ﺗﻜﻮﻥ ﺃﻥ ﻋﻠﻰ" – H CH3―C―OH + [O] CH3―C―OH CH3―C═O H2SO4 ZnCl2 H H H OH H ─ C─OH أﻛﺴﺪة أﻛﺴﺪة ﺑﻔﻘﺪ ﯾﺴﺘﻘﺮ ﺟﺰىء–H2O K2Cr2O7 H2SO4
- 9. ٤٨ CH3CHO + [O] CH3COOH +H2 – H CH3―C―OH + [O] CH3―C―OH CH3―C― CH3 – – H ––– ––– : CH3 H CH3 OH K2Cr2O7 H2SO4 أﻛﺴﺪة O ﺑﻔﻘﺪ ﯾﺴﺘﻘﺮ ﺟﺰىء–H2O K2Cr2O7 H2SO4
- 10. ٤٩ –ْ C2H5OH C2H4 + H2O –ْ 2 C2H5OH C2H5 O C2H5 + H2O C2H4 + H2O + [O] H2SO4 180ْC CH2 – CH2 OH OH – ٫ KMnO4 H2SO4 140ْC CH2 – CH2 OH OH
- 11. ٥٠ H―C―C―C―H CH2―O―NO2CH2―OH CH―OH + 3HONO2 CH―O―NO2 + 3H2O CH2―OH CH2―O―NO2 OH – OH – OH H2SO4 –– PEG – – – !!! H OH H OH H OH
- 12. ٥١ – – CH2OH C=O CHOH)3( CH2OH H │ H―C―OH │ C ═ O │ H―C―OH │ H―C―OH │ H―C―OH │ H―C―OH │ H CHO ( CHOH)4 CH2OH CHO │ H―C―OH │ H―C―OH │ H―C―OH │ H―C―OH │ H―C―OH │ H OH OH OH OHOH OH C6H5─OH
- 13. ٥٢ + NaOH + NaCl –O─H –O─HH+ ﻳﺘﻀ ﺫﻟﻚ ﻋﻠﻰ ﻭﺍﻟﺪﻟﻴﻞﻳﻠﻰ ﻓﻴﻤﺎ ﺢ ﺍﻟﻄﺒﻴﻌﻴﺔ ﺍﳊﺎﻟﺔ ﻋﺒﺎﺩ ﻋﻠﻰ ﺍﻟﺘﺄﺛﲑ ﺍﻟﺸﻤﺲ ﺍﳊﺎﻣﻀﻴﺔ ﺍﳋﺎﺻﻴﺔ OH O─HH+ O─H H+ δ- δ+ C2H5─O-H ﺍﻟﻔﻠﺰﺍﺕ ﻣﻊ ﺍﻟﺘﻔﺎﻋﻞ ﻣﺜﻞ ﺍﻟﻨﺸﻄﺔNa C2H5─ONa + H2 ﺍﻟﻘﻠﻮﻳﺎﺕ ﻣﻊ ﺍﻟﺘﻔﺎﻋﻞ ﻣﺜﻞNaOH ﺍﻷﲪﺎﺽ ﻣﻊ ﺍﻟﺘﻔﺎﻋﻞ ﻣﺜﻞHCl OH C2H5─Cl + H2O OH OH C─OH OHCl δ+ δ+ O─H ONa + H2 ONa + H2O
- 14. ٥٣ "" !!! H─C═O a a + b b + ab n :– – : III + 3 HO-NO2 H2SO4 + 3 H2O OH NO2 NO2O2N – H I OH H HH
- 15. ٥٤ ─ C─ OH ─ C── OH – – HCOOH CH3COOH C3H7COOH C15H31COOH O O R─ C─ OH O C─ OH O HCOOH CH3COOH COOH COOH COOH COOH COOH OH COOH
- 16. ٥٥ H─C≡C─H + H2O أﺳﯿﺘﯿﻚ ﺣﻤﺾأﺳﯿﺘﺎﻟﺪھﯿﺪأﺳﺘﯿﻠﯿﻦ O H―O R ― C C ― R O ― H O – – HgSO4 60°C H2SO4 40% CH3─ CH CH3─ C─ OH [O] R―C―O H
- 17. ٥٦ 2 CH3COOH + Mg (CH3COO)2Mg + H2 CH3COOH + NaHCO3 CH3COONa + H2O + CO2 CH3COOH + C2H5OH CH3COOC2H5 + H2O CH3COOH + 2 H2 C2H5OH + H2O CO2 –– 200ْC CuCrO4 – – H2SO4 ﻣﺮﻛﺰ –
- 18. ٥٧ 2 + 3 O2 2 + 2H2O HCl ,CH3COOH , C2H5OH , , + NaOH + H2O + C2H5OH + H2O COOHCH3 V2O5 400 ْC COOH COOH COONa COOH HClﺟﺎف COOC2H5 OH
- 19. ٥٨ ١ HCOOH ١-٢-٣- ٤-٥- ٢ CH3COOH ١-٢-- - ٣ - -٫ ٤ H │ H―C―COOH │ HO―C―COOH │ H―C― COOH │ H - - -pH ٥ CH3 │ HO―C―COOH │ H ٦ ٧ –– – ٨– (─NH2) NH2CH2COOH─NH2 + H CH2COOH ⍺ COOH R─ C ─COOH H I I NH2 ⍺ OH COOH
- 20. ٥٩ ﺍ ﺍﲢﺎﺩ ﻧﻮﺍﺗﺞ ﻫﻰﺍﻟﻜﺤﻮﻻﺕ ﻣﻊ ﺍﻟﻜﺮﺑﻮﻛﺴﻴﻠﻴﺔ ﻷﲪﺎﺽ : CH3COOH + C2H5OH CH3COOC2H5 + H2O ─ OH R – C – OH + H –OR R – C – OR + H2O HCOOCH3 CH3COOC2H5 COOC2H5
- 21. ٦٠ : CH3COOC2H5 + H2O CH3COOH + C2H5OH CH3COOC2H5 + NaOH CH3COONa + C2H5OH C6H5COOC2H5 + NaOH C6H5COONa + C2H5OH CH3─C─OC2H5 + NH3 CH3─C─NH2 + C2H5OH C6H5─C─OC2H5 + NH3 C6H5─C─NH2 + C2H5OH R – C –OR + H2O R – C – OH + R–OH HCl ﻣﺎﺋﯿﺔ ﻣﺎﺋﯿﺔ
- 22. ٦١ : + + 3H2O NaOHKOH O CH3 || | H – C – O – CH2 – CH – CH3 O || CH3 – C – O – (CH2)7 – CH3 O || CH3 – CH2 – C – O – (CH2)4 – CH3 O || CH3 – (CH2)2 – C – O – CH3 O || CH3 – (CH2)2 – C – O – CH2 – CH3 O || CH3 – C – O – CH2 – C6H5 CH2 – OH | CH – OH | CH2 – OH CH2 – O – C – R1 CH – O – C – R2 CH2 – O – C – R3 O O O HO – C – R1 HO – C – R2 HO – C – R3 ∆
- 23. ٦٢ + HO – CH2 – CH2 – OH - - (OH , COOH).. HO – C C – OH - H2O HO – C C – O – CH2 – CH2 – OH n OH O || – C – OH H – O – CH3+ OH O || – C – OCH3 + H2O + HO – C – CH3 || O O H O || – C – OH O– O || – C – OH C – CH3 || O H2O+
