Lecture 8.4b- Polar Molecules
•Download as PPT, PDF•
3 likes•3,450 views
Section 8.4 lecture (part B) for Honors & Prep Chemistry
Report
Share
Report
Share
Recommended
Recommended
Intermolecular Forces: London Dispersion Forces and Dipole-Dipole Attractions

Intermolecular Forces: London Dispersion Forces and Dipole-Dipole AttractionsShaina Mavreen Villaroza
More Related Content
What's hot
Intermolecular Forces: London Dispersion Forces and Dipole-Dipole Attractions

Intermolecular Forces: London Dispersion Forces and Dipole-Dipole AttractionsShaina Mavreen Villaroza
What's hot (20)
Intermolecular Forces: London Dispersion Forces and Dipole-Dipole Attractions

Intermolecular Forces: London Dispersion Forces and Dipole-Dipole Attractions
Lesson 1_Kinetic Molecular Theory of Solids and Liquids.pptx

Lesson 1_Kinetic Molecular Theory of Solids and Liquids.pptx
Viewers also liked
Viewers also liked (20)
Travis County Transportation Plan: Creating Tomorrow's Choice's Today

Travis County Transportation Plan: Creating Tomorrow's Choice's Today
IB Chemistry on Polarity, Hydrogen Bonding and Van Der Waals forces

IB Chemistry on Polarity, Hydrogen Bonding and Van Der Waals forces
Similar to Lecture 8.4b- Polar Molecules
Similar to Lecture 8.4b- Polar Molecules (20)
More from Mary Beth Smith
More from Mary Beth Smith (20)
Chapter 3 and 5 lecture- Ecology & Population Growth

Chapter 3 and 5 lecture- Ecology & Population Growth
Biotechnology Chapter Five Lecture- Proteins (part b)

Biotechnology Chapter Five Lecture- Proteins (part b)
Biotechnology Chapter Five Lecture- Proteins (part a)

Biotechnology Chapter Five Lecture- Proteins (part a)
Recently uploaded
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...?#DUbAI#??##{{(☎️+971_581248768%)**%*]'#abortion pills for sale in dubai@
Recently uploaded (20)
Cloud Frontiers: A Deep Dive into Serverless Spatial Data and FME

Cloud Frontiers: A Deep Dive into Serverless Spatial Data and FME
Apidays New York 2024 - Passkeys: Developing APIs to enable passwordless auth...

Apidays New York 2024 - Passkeys: Developing APIs to enable passwordless auth...
Spring Boot vs Quarkus the ultimate battle - DevoxxUK

Spring Boot vs Quarkus the ultimate battle - DevoxxUK
DEV meet-up UiPath Document Understanding May 7 2024 Amsterdam

DEV meet-up UiPath Document Understanding May 7 2024 Amsterdam
How to Troubleshoot Apps for the Modern Connected Worker

How to Troubleshoot Apps for the Modern Connected Worker
Cloud Frontiers: A Deep Dive into Serverless Spatial Data and FME

Cloud Frontiers: A Deep Dive into Serverless Spatial Data and FME
Polkadot JAM Slides - Token2049 - By Dr. Gavin Wood

Polkadot JAM Slides - Token2049 - By Dr. Gavin Wood
EMPOWERMENT TECHNOLOGY GRADE 11 QUARTER 2 REVIEWER

EMPOWERMENT TECHNOLOGY GRADE 11 QUARTER 2 REVIEWER
Biography Of Angeliki Cooney | Senior Vice President Life Sciences | Albany, ...

Biography Of Angeliki Cooney | Senior Vice President Life Sciences | Albany, ...
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...
Strategies for Landing an Oracle DBA Job as a Fresher

Strategies for Landing an Oracle DBA Job as a Fresher
Axa Assurance Maroc - Insurer Innovation Award 2024

Axa Assurance Maroc - Insurer Innovation Award 2024
Web Form Automation for Bonterra Impact Management (fka Social Solutions Apri...

Web Form Automation for Bonterra Impact Management (fka Social Solutions Apri...
Finding Java's Hidden Performance Traps @ DevoxxUK 2024

Finding Java's Hidden Performance Traps @ DevoxxUK 2024
"I see eyes in my soup": How Delivery Hero implemented the safety system for ...

"I see eyes in my soup": How Delivery Hero implemented the safety system for ...
Why Teams call analytics are critical to your entire business

Why Teams call analytics are critical to your entire business
Lecture 8.4b- Polar Molecules
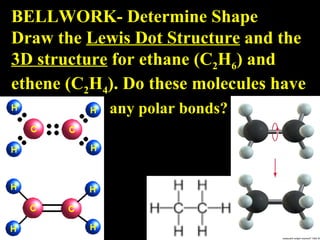
- 1. BELLWORK- Determine Shape Draw the Lewis Dot Structure and the 3D structure for ethane (C 2 H 6 ) and ethene (C 2 H 4 ). Do these molecules have any polar bonds?
- 2. In review Equal sharing of electrons = covalent LOW electronegativity difference Unequal sharing of electrons = polar MEDIUM electronegativity difference Transfer of electrons = ionic HIGH electronegativity difference
- 3. 2 bonding domains Three bonding domains Four bonding domains Three bonding and one lone pair Two bonding and two lone pairs
- 4. Bond polarity and molecule shape determine if a molecule is polar Bond polarity --- When a bond has a partial negative charge on one atom and a partial positive charge on the other atom. Molecule shape--- the arrangement of atoms in three dimensions (3-D)
- 5. A polar molecule has polar bonds and asymmetry
- 6. A polar molecule has polar bonds and asymmetry Polar bonds Non-polar molecule Symmetry- all sides are the same δ - δ - δ - δ - δ +
- 7. A polar molecule has polar bonds and asymmetry Polar bonds Non-polar molecule Symmetry- all sides are the same Polar bonds Polar molecule Asymmetry- has different sides δ - δ - δ - δ - δ - δ + δ + negative side Positive side
- 8. If the electrons are not distributed equally, the molecule is polar. The molecule has a negative end and a positive end.
- 9. A polar molecule has a partially positive side and a partially negative side. The arrow labels the molecular polarity. It shows that electrons are mostly by the oxygen atom O H H
- 10. Polar molecules are affected by electric fields
- 11. Polar molecules have two poles; one is partially positive and one is slightly negative. Positive end Negative end N H H H
- 12. The positive pole ( + ) is attracted to negative ions and the negative poles( - ) in other polar molecules. POLAR MOLECULES INTERACT!!
- 13. + POLAR MOLECULES INTERACT!! The negative pole ( - ) is attracted to positive ions and the positive poles( + ) in other polar molecules.
- 14. Water is a molecule that has two polar O-H bonds.
- 15. The electrons are not distributed evenly, so the water molecule is polar. The negative pole is at the oxygen. O is more electronegative than H, so electrons are pulled toward O. Also, there are two lone pairs around oxygen. negative end positive end
- 16. Na + (aq) A dissolved sodium ion
- 17. Water is very good at dissolving salts because it can surround anions & cations.
- 18. Practice- Using last night’s homework assignment, label the molecular polarity on the four pictures H F