Analysis of the Monoclonal Antibody Adalimumab in Human Blood Collected via Volumetric Adsorptive Microsampling (VAMS) Technology and Utilizing LC-MS/MS Detection
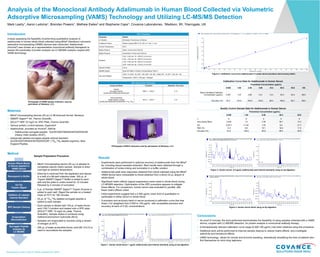
- 1. Presented at July Land O’ Lakes Conference 2019 Analysis of the Monoclonal Antibody Adalimumab in Human Blood Collected via Volumetric Adsorptive Microsampling (VAMS) Technology and Utilizing LC-MS/MS Detection Mark Leahy1, Aaron Ledvina1, Brendan Powers1, Mathew Ewles2 and Stephanie Cape1; Covance Laboratories, 1Madison, WI, 2Harrogate, UK Introduction A study assessing the feasibility of performing quantitative analysis of adalimumab in human whole blood collected using Mitra® (Neoteryx) volumetric adsorptive microsampling (VAMS) devices was conducted. Adalimumab (Humira®) was chosen as a representative monoclonal antibody therapeutic to assess the practicality of protein analysis via LC-MS/MS analysis coupled with VAMS technology. Materials ▶ Mitra® microsampling devices (20 µL) in 96-Autorack format, Neoteryx ▶ SMART Digest™ Kit, Thermo Scientific ▶ SOLA™ HRP 10 mg/2 mL SPE Plate, Thermo Scientific ▶ Various protein Lo-bind labware, Eppendorf ▶ Adalimumab, provided as Humira®, AbbVie ▶ Adalimumab surrogate peptide: GLEWVSAITWNSGHIDYADSVEGR (Heavy chain position 44-67) ▶ Isotopically labeled surrogate peptide internal standard, GLEWVSAITWNSGHIDYADSVEGR^ (13C6,15N4-labeled arginine), New England Peptide Results ▶ Experiments were performed to optimize recovery of adalimumab from the Mitra® tip, including impact-assisted extraction. Best results were obtained through a combination of vortex-mixing and sonication in a buffer solution. ▶ Adalimumab peak area responses obtained from blood collected using the Mitra® VAMS device were comparable to those obtained from a direct 20 µL aliquot of whole blood. ▶ Significant matrix effects (signal suppression) were noted in whole blood during LC-MS/MS detection. Optimization of the SPE procedure appears to mitigate these effects. For comparison, human serum was evaluated in parallel, with fewer matrix effects noted. ▶ Initial experiments suggest that a 0.500 µg/mL lower limit of quantitation is achievable in either serum or whole blood. ▶ A precision and accuracy batch in serum produced a calibration curve that was linear (1/x2 weighted) from 0.500 to 100 µg/mL, with acceptable precision and accuracy at each of 5 QC concentrations. HPLC Parameters Parameter Details LC System Shimadzu, Prominence, 30 Series Analytical Column Waters, Acquity BEH C18, 100 x 2.1 mm, 1.7 µm Column Temperature 45°C Mobile Phase A Water: Formic Acid (100:0.5) Mobile Phase B Acetonitrile: Formic Acid (100:0.5) Gradient 0.00 – 6.00 min, 15→35% B, 0.4 mL/min 6.00 – 7.00 min, 35→90% B, 0.4 mL/min 7.00 – 8.50 min, 90→90% B, 0.4 mL/min 8.50 – 8.60 min, 90→15% B, 0.4 mL/min Injection Details 4-6 µL MS/MS System Sciex API 6500 in Positive TurboIonSpray® (ESI+) Gas and Voltages CAD = 8, CUR = 30, GS1 = 50, GS2 = 50, ISV = 4500, EP = 10, DP = 130, CE = 36 Temperature = 650°C, CAD gas = Nitrogen Mass Spectrometer Parameters Compound Name Transition Retention Time (min) Analyte Surrogate Peptide GLEWVSAITWNSGHIDYADSVEGR 886.5 → 1040.0 5.12 Internal standard Stable labeled peptide GLEWVSAITWNSGHIDYADSVEGR^ (13C6,15N4) 891.5 → 1044.4 5.12 Photograph of VAMS sample collection, used by permission of Neoteryx, LLC. Photograph of Mitra® Autorack used by permission of Neoteryx, LLC Human Whole Blood or Serum Applied to VAMS Device Resuspend in Buffer Addition of Labeled Internal Standard SPE Sample Cleanup Evaporation/ Reconstitution Surrogate Peptide Analysis by LC-MS/MS On-Tip Trypsin Digest ▶ Mitra® microsampling device (20 µL) is allowed to completely adsorb matrix sample. Sample is dried overnight at ambient temperature. ▶ Dried tip is removed from the applicator and placed in a well of a 96-well collection plate. 300 µL of Trypsin SMART Digest™ Buffer is added to each well and the plate is vortex-mixed for 10 minutes followed by 5 minutes of sonication. ▶ 5 µL of Soluble SMART Digest™ Trypsin Enzyme is added to each well. Digest the sample on a heated mixer at 70°C for 180 minutes. ▶ 25 µL of 13C6,15N4-labeled surrogate peptide is added to each sample. ▶ Each sample is diluted with 100 µL of water:formic acid (100:1) solution and loaded onto a SPE plate (SOLA™ HRP, 10 mg/2 mL plate, Thermo Scientific). Sample elution is achieved using methanol:ammonium hydroxide (95:5). ▶ Samples are evaporated to dryness using a stream of nitrogen at 40°C. ▶ 200 µL of water:acetonitrile:formic acid (85:15:0.5) is used to reconstitute the samples. Sample Preparation Procedure Method Figure 1. Human whole blood: 1 µg/mL adalimumab (and internal standard) using on-tip digestion. Figure 2. Calibration curve from adalimumab in human serum precision and accuracy batch. Figure 3. Human serum: 0.5 µg/mL adalimumab (and internal standard) using on-tip digestion. Figure 4. Human serum blank using on-tip digestion. Conclusions ▶ As proof of concept, the work performed demonstrates the feasibility of using samples collected with a VAMS device, coupled with LC-MS/MS detection, for protein analysis in monoclonal antibody therapy. ▶ A therapeutically relevant calibration curve range (0.500-100 µg/mL) has been obtained using this procedure. ▶ Additional work will be performed to improve sample cleanup to reduce matrix effects, and investigate selectivity and hematocrit effects. ▶ VAMS technology allows for remote and at-home sampling, dramatically simplifying the lives of patients who find themselves on strict drug regimens. Quality Control Sample Data for Adalimumab in Human Serum Theoretical Concentration (µg/mL) 0.500 1.50 8.00 40.0 80.0 n 6 6 6 6 6 Intra-Assay Mean 0.459 1.5 7.58 39.7 74.3 SD 0.0571 0.132 1.45 6.6 8.17 RSD (%) 12.4 8.8 19.1 16.6 11.0 Accuracy (%) 91.8 100.0 94.8 99.3 92.9 Calibration Curve Data for Adalimumab in Human Serum Theoretical Concentration (µg/mL) 0.500 1.00 2.50 5.00 15.0 50.0 85.0 100 Back-Calculated Calibrator Concentration (µg/mL) 0.487 1.03 2.60 5.21 13.5 53.0 83.4 96.9 Accuracy (%) 97.4 103.0 104.0 104.2 90.0 106.0 98.1 96.9
