More Related Content
Similar to molecular geometry.ppt
Similar to molecular geometry.ppt (20)
AQA A-Level Chemistry New Spec: Shapes of molecules

AQA A-Level Chemistry New Spec: Shapes of molecules
CHEMICAL BONDING II: Molecular geometry and Hybridization of Atomic orbitals

CHEMICAL BONDING II: Molecular geometry and Hybridization of Atomic orbitals
STEROCHEMISTRY AND BONDING IN MAIN GROUP COMPOUNDS 

STEROCHEMISTRY AND BONDING IN MAIN GROUP COMPOUNDS
More from carlmanaay
More from carlmanaay (20)
Recently uploaded
young call girls in Janakpuri🔝 9953056974 🔝 escort Service

young call girls in Janakpuri🔝 9953056974 🔝 escort Service9953056974 Low Rate Call Girls In Saket, Delhi NCR
Sexy Call Girls Patel Nagar New Delhi +918448380779 Call Girls Service in Del...

Sexy Call Girls Patel Nagar New Delhi +918448380779 Call Girls Service in Del...Stunning ➥8448380779▻ Call Girls In Hauz Khas Delhi NCR
FULL ENJOY Call Girls In kashmiri gate (Delhi) Call Us 9953056974

FULL ENJOY Call Girls In kashmiri gate (Delhi) Call Us 99530569749953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
Call In girls Connaught Place (DELHI)⇛9711147426🔝Delhi NCR

Call In girls Connaught Place (DELHI)⇛9711147426🔝Delhi NCR
9873940964 High Profile Call Girls Delhi |Defence Colony ( MAYA CHOPRA ) DE...

9873940964 High Profile Call Girls Delhi |Defence Colony ( MAYA CHOPRA ) DE...
young call girls in Janakpuri🔝 9953056974 🔝 escort Service

young call girls in Janakpuri🔝 9953056974 🔝 escort Service
Call Girls South Delhi Delhi reach out to us at ☎ 9711199012

Call Girls South Delhi Delhi reach out to us at ☎ 9711199012
EARTH DAY Slide show EARTHDAY.ORG is unwavering in our commitment to end plas...

EARTH DAY Slide show EARTHDAY.ORG is unwavering in our commitment to end plas...
VIP Kolkata Call Girl Kalighat 👉 8250192130 Available With Room

VIP Kolkata Call Girl Kalighat 👉 8250192130 Available With Room
Call {Girls Delhi} Very Low rateVaishali 9711199012 DownLoad PDF

Call {Girls Delhi} Very Low rateVaishali 9711199012 DownLoad PDF
Call Girls Sarovar Portico Naraina Hotel, New Delhi 9873777170

Call Girls Sarovar Portico Naraina Hotel, New Delhi 9873777170
Spiders by Slidesgo - an introduction to arachnids

Spiders by Slidesgo - an introduction to arachnids
Delivering nature-based solution outcomes by addressing policy, institutiona...

Delivering nature-based solution outcomes by addressing policy, institutiona...
Private Escorts Aliganj \ 9548273370 Indian Call Girls Service Lucknow ₹,9527

Private Escorts Aliganj \ 9548273370 Indian Call Girls Service Lucknow ₹,9527
Sexy Call Girls Patel Nagar New Delhi +918448380779 Call Girls Service in Del...

Sexy Call Girls Patel Nagar New Delhi +918448380779 Call Girls Service in Del...
VIP Call Girls Service Bandlaguda Hyderabad Call +91-8250192130

VIP Call Girls Service Bandlaguda Hyderabad Call +91-8250192130
Air pollution soli pollution water pollution noise pollution land pollution

Air pollution soli pollution water pollution noise pollution land pollution
FULL ENJOY Call Girls In kashmiri gate (Delhi) Call Us 9953056974

FULL ENJOY Call Girls In kashmiri gate (Delhi) Call Us 9953056974
molecular geometry.ppt
- 2. VSEPR theory assumes that the shape of a molecule is determined by the repulsion of electron pairs. Molecular Shape
- 3. VSEPR Theory Based on Electron Dot (Lewis structures) Theory predicts shapes of compounds abbreviated VSEPR VSEPR (pronounced “vesper”) stands for Valence Shell Electron Pair Repulsion VSEPR predicts shapes based on electron pairs repelling (in bonds or by themselves) Electrons around central nucleus repel each other. So, structures have atoms maximally spread out
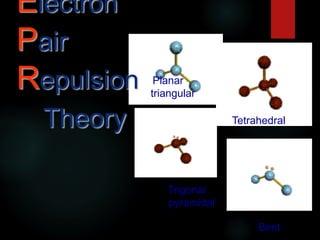
- 4. VSEPR overview Each shape has a name (you will have to know these) Names of Shapes: tetrahedral trigonal pyramidal Bent Linear trigonal planar
- 5. Triangular Planar Tetrahedral Trigonal pyramidal Linear Bent or V Models
- 6. C H H H H methane, CH4 Bonds are all evenly spaced electrons 109.5° C H H H H Tetrahedral
- 7. N H H H C H H H H Less repulsion between the bonding pairs of electrons .. ammonia NH3 .. .. .. Trigonal Pyramidal
- 8. C H H H H N H H H O H H .. .. .. 109.5° (109.5°) 109.5° (107°) 109.5° (104.5°) water, H2O
- 9. C H H H H N H H H O H H .. .. ..
- 10. H C H H H N H H H O H H Bent or V 2 unshared pairs of e’s at top of O repel bonds and force them to bend
- 11. Molecule Lewis Structure Number of electron pairs CH4 NH3 SHAPE Tetrahedral Trigonal Pyramidal 4 4 (3 shared 1 lone pair)
- 12. Molecule Lewis Structure Number of electron pairs H2O CO2 SHAPE Bent or V 4 (2 shared 2 lone pairs) 2 Linear
- 13. Molecule Lewis Structure Number of electron pairs BeCl2 BF3 SHAPE 2 3 Linear Trigonal Planar
Editor's Notes
- 3
- 4
- 14
- 15