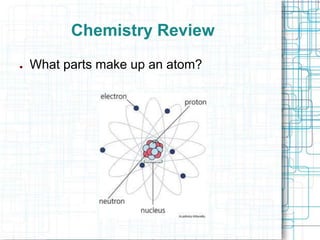
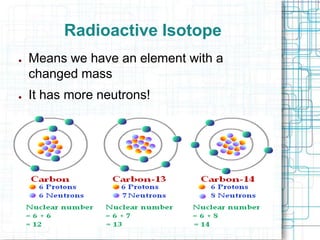
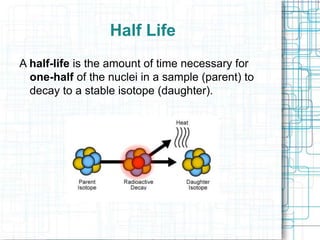
This document provides information about absolute age dating and radioactive dating techniques. It defines key terms like absolute age dating, radioactive isotopes, parent material, half-life, and daughter material. Examples are given of radioactive elements like uranium, radon, and radium that are used in radioactive dating. Methods like radiocarbon dating are also summarized.