Column chromatography
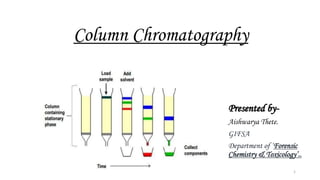
- 1. Column Chromatography Presented by- Aishwarya Thete. GIFSA Department of ‘Forensic Chemistry & Toxicology’.. 1
- 2. Outline: Introduction Basic term Principle Experimental setup adsorbent Solvents Working Factors affecting column efficiency 2
- 4. Introduction Also known as adsorption chromatography. Developed by- D.T. Day(1900) & M.S. Tswett (1906) Experiment on separation of plant pigment Solid - liquid technique Stationary phase -solid & Mobile phase - liquid. Generally used for separation of lipid molecule. 2 types of Column Chromatography are generally used: a) Adsorption chromatography: Procedure which separates lipid mixtures mainly according to their relative polarities. Ex: mix of organic comp. b) ion exchange chromatography: Procedure which is based on the importance of ionic groups present in some lipid molecules. Ex: mix of proteins, amino acid 4
- 5. Principle Selective Adsorption/differential adsorption. The component - -which has greater adsorbing powder is adsorbed in the upper part of the column. - The next component is adsorbed in the lower portion of the column which has lesser adsorbing power than the first component. - This process is continued & as a result, the materials are partially separated & adsorbed in the various parts of the column. 5
- 6. Adsorbent • Uniform size particle, spherical shape • Good adsorbent, good filtration properties • Chemically inert • Neutral surface except in case of ion exchanger. • Good mechanical stability so that their will not be formation of dust particle. 6
- 7. Silica gel Activated Charcoal Calcium Carbonate (Magnesium Carbonate) 7
- 8. Selection of solvent * Functions to perform: a) act as developer. b) introduces the mixture to the columns. c) act as eluents *Solvent selection: a) Solubility of sample. b) Boiling point b/w 45-85˚ C. c) Pet ether having-most widely used B.P.<80 ˚ C. d) Mobile phase can be single solvent or mixture of solvent. It depends on eluting power. 8
- 9. Pet ether Cyclohexane TCM 9
- 10. 10
- 11. Column preparation & procedure 1. • Keep the supporting material(glass wool/cotton plug). 2 • Clamp the column vertically. 3 • Add the adsorbent material by making slurry in a suitable medium to the 2/3rd of the column. 4 • If adsorbent material is solid, pack the glass tube uniformly. After adding each portion press it with flattened glass rod. 5 • Cover the surface again with supporting material. 6 • Add the sample to b separated & cover the surface with glass wool. 7 • Add the eluent. 11
- 13. Dissolve the sample in suitable solvent Preparation of column Selection of eluent Introduce the sample Collection of eluent STEPS Video 1 13
- 14. Process 14
- 15. Factors Affecting Column Efficiency 1. Nature of solvent -Low viscosity -Rate of flow α 1 Viscosity 2. Pore diameter - <20A˚ 3. Temperature of the column: - Generally we use room temp. - Samples which are difficult to soluble, we use high temperature - 15
- 16. 4. Partical size of column packing: - Usually it ranges from 100-200 mesh. - Column efficiency= 1 Particle size 5. Dimensions of column: - Ratio of length: diameter > 20:1 - Ideal dimension is 40:1 Peak resolution= RtA-RtB/ WA+WB 16
- 17. Applications 17 Separatio n of Proteins Amino acids Carbohydrates Peptides Mixture of compounds Geometrical isomers Alkaloids
- 18. 18 Other applications Determination of primary & sec. glycosides in digitalis leaf. Isolation of active constituents Synthesizing or isolating novel compounds Isolation of metabolites from biological fluids.