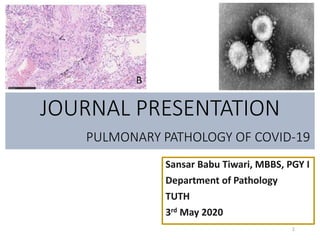
Pulmonary pathology of corona virus
- 1. JOURNAL PRESENTATION PULMONARY PATHOLOGY OF COVID-19 Sansar Babu Tiwari, MBBS, PGY I Department of Pathology TUTH 3rd May 2020 1
- 2. Pulmonary post-mortem findings in a large series of COVID-19 cases from Northern Italy • Aurelio Sonzogni, MD et. al. • Pathology Unit, L. Sacco Hospital, Milan, Italy • Department of Pathology- Papa Giovanni XXIII Hospital - Bergamo, Italy
- 3. INTRODUCTION: • Italy has been the first country in Europe to be reached by the epidemic and Lombardy was devastated in just one month. • L. Sacco Hospital, Milan and Papa Giovanni XXIII, Bergamo have been the first hospitals in this region to manage the epidemic crisis. • The clinical spectrum of SARS-CoV-2 disease (COVID-19) is reported to include mild asymptomatic infection, mild upper respiratory disease with fever and cough, and severe pneumonia that can lead to ARDS in 15% of the hospitalized cases.
- 4. INTRODUCTION: • They described the first available large series of lung histopathological findings in patients died from COVID-19 in Northern Italy, with the aim to report the main microscopic pulmonary lesions in SARS-CoV-2 infection and severe respiratory failure.
- 5. METHODS: • Histological analysis of post-mortem lung tissues from 38 cases who died for COVID-19 between February and March in two referral centres for the management of the COVID-19 outbreak in Northern Italy, Luigi Sacco Hospital, Milan, and Papa Giovanni XXIII, Bergamo (patient permission given by the Ethics Committees of the two hospitals to use personal and sensitive data for scientific research related to the disease was collected).
- 6. METHODS: • The autopsies were performed in Airborne Infection Isolation Autopsy Rooms and the personnel used the correct Personal Protection Equipment (PPE), according to “Engineering control and PPE recommendations for autopsies”. • A medium of 7 tissue blocks were taken from each lung (range 5-9), selecting the most representative areas at macroscopic examination. Tissues were fixed in 10% buffered formalin for >48 hours. Three-μm paraffin sections were stained by hematoxylin-eosin. Immunohistochemistry reactions were performed on selected cases (CD45, CD68, CD61, TTF1, p40, Ki67, Masson Trichome) to better characterize inflammatory infiltrate, epithelial cells and fibrosis.
- 7. METHODS: • Histological evaluation was performed blindly by two pathologists from each hospital, with expertise in the field. Histological features of the cellular and interstitial damage were described and graded by using a semi-quantitative scale. • Additional samples from selected cases were fixed in glutaraldehyde for electron microscopy and examined by EM-109 ZEISS and CCD-Megaview G2 (I-TEM imaging platform software).
- 8. RESULTS: • Patients were 33 males and 5 females, average age of 69 years (range 32-86); the time that the patients spent in the Subintensive/Intensive Care Unit ranged from 1 to 23 days (6.87 days). • Regarding past comorbidities, data were available in 31/38 patients: 9 diabetes, 18 hypertension, 4 past malignancies, 11 cardiovascular disorders, 3 mild chronic obstructive pulmonary disorders.
- 9. RESULTS: • At the time of hospitalization, all the patients had throat swab sample positive for SARS-CoV-2 infection and had clinical and radiological features of interstitial pneumonia. D-dimer was available in 26/38 patients, with high value in all of them (>10 x the upper reference limit). • Patients died after a median time of 16.27 days (range 5-31) from the onset of symptoms.
- 10. RESULTS: • Macroscopic examination of the lungs revealed heavy, congested and oedematous organs, with spotty involvement. • At histological examination, the features of Diffuse Alveolar Disease (DAD) were found, corresponding to those observable in the exudative and early/intermediate proliferative phases of the disease. • Both phases often overlapped in the different areas of the lungs, with pluri-focal pattern of distribution. The fibrotic phase was rarely observed, possibly due to the short duration of the disease. Moreover, five patients also had bacterial (4) and fungal (1) abscesses.
- 11. RESULTS: Hyaline membrane Microthrombus Megakaryocyte entrapment Type II pneumocyte hyperplasia
- 12. RESULTS: • Capillary congestion, interstitial edema, dilated alveolar ducts, hyaline membranes composed of serum proteins and condensed fibrin, loss of pneumocytes were the histological patterns of the exudative phase mostly observed in all the cases. • Platelet-fibrin thrombi in small arterial vessels (<1mm in diameter) were found in 33 cases. • Moreover, type II pneumocyte hyperplasia showing reactive atypia, myofibroblast proliferation, alveolar granulation tissue and obliterating fibrosis were present in half of the patients but were focal.
- 14. RESULTS: • Microcystic honeycombing and mural fibrosis were occasionally present. • Inflammatory component was represented by a few CD45 positive lymphocytes located in the interstitial space; a large number of CD68 positive macrophages were mainly localized in the alveolar lumens. • Immunohistochemistry with anti CD61 antibody identified increased number of megakaryocytes in lung capillaries
- 15. RESULTS: CD68 macrophages CD45 Interstitial lymphocytes
- 16. RESULTS: • Ultrastructural examination revealed viral particles, with morphology typical of the family of Coronaviridae and localized along plasmalemmal membranes and within cytoplasmic vacuoles of pneumocytes. • Virions had an average diameter of 82nm and viral projection about 13nm in length.
- 17. DISCUSSIONS: • The peculiar histopathological findings were atypical pneumocytes (reactive atypia) and diffuse thrombosis of the peripheral small vessels. • In their study, fibrin thrombi of small arterial vessels (diameter < 1mm) were observed in 33 /38 patients, half of them with >25% of tissue involvement and associated with high levels of D- dimer in blood. • These findings might explain the severe hypoxemia which characterizes the clinical feature of ARDS in SARS-CoV-2 patients.
- 18. DISCUSSIONS: • Their data strongly support the hypothesis proposed by recent clinical studies, that COVID-19 is complicated or anyway strictly related to coagulopathy and thrombosis. • Moreover detection of D-dimer values >1 μg/ml have been associated with fatal outcome of COVID- 19. • For these reasons, the use of anticoagulants has been recently suggested as potentially beneficial in patients with severe COVID-19, albeit its efficacy and safety have not been demonstrated.
- 19. DISCUSSIONS: • Finally, the search for viral particles was carried out in a subset of patients and highlighted the presence of rare virions in the cytoplasm of pneumocytes. Despite the low number of cases, these findings may suggest that the virus remains in lung tissue for many days, even if in small quantities, possibly being the trigger of the mechanism that leads to and feeds lung damage.
- 20. Histopathology and genetic susceptibility in COVID-19 pneumonia. • von der Thüsen, MD, PhD • Menno van der Eerden, MD PhD • Dept. of Pathology and Dept. of Pulmonology, Erasmus MC, Rotterdam, The Netherlands
- 21. INFLUENZA VIRUS: • The respiratory tract histology of influenza reflects its cellular tropism, as influenza virus replicates in respiratory epithelial cells throughout the respiratory tree, with non-fatal infections predominantly involving the upper respiratory tract and trachea, but fatal cases of influenza usually result from pneumonia. • Changes in the smaller airways include necrosis and complete loss of the epithelial layer (both ciliated and goblet cells), often resulting in the formation of hyaline membranes at these sites. Neutrophilic inflammation may be present in the lumen, and extend into surrounding alveoli.
- 22. INFLUENZA VIRUS: • In addition, a more chronic lymphoplasma- histiocytic infiltrate is often seen in influenza- infected airways. The parenchyma shows evidence of acute injury with interstitial congestion, edema, and inflammation (predominantly neutrophilic with some eosinophils), as well as desquamated alveolar epithelial cells, intra-alveolar edema, intra- alveolar hemorrhage, fibrin, hyaline membrane formation, and sometimes necrosis of the alveolar septa (necrotizing alveolitis), the latter possibly resulting from frequently observed vascular changes with capillary congestion and thrombosis.
- 23. COVID-19: • They have recently also observed similar changes in COVID-19 autopsy cases, with evidence of extensive microvascular damage and thrombotic occlusion as the foremost pattern of injury, resulting in intra-alveolar fibrinous exudates akin to acute fibrinous and organizing pneumonia (AFOP).
- 24. COVID-19 : • This spectrum of abnormalities is highly reminiscent of changes seen in the context of the chronic lung allograft rejection (CLAD) of the restrictive phenotype (restrictive allograft syndrome (RAS)), which is presumed to occur secondary to antibody-mediated endothelial damage and complement activation of humoral rejection episodes. • Such vascular changes are likely to play a central role in the pathogenesis of COVID-19, and may in part be attributable to dysregulation of the endothelial ACE2 receptor with ensuing bradykinin-dependent local lung edema, as well as a highly pro-thrombotic state, which can manifest itself in the lung as well as in extra- pulmonary tissues.
- 25. COVID-19 : • Direct infection of endothelial cells may play a central role in this process. More chronic changes included intra-alveolar organization with fibroblastic proliferation and diffuse fibrotic thickening of alveolar walls, consisting of proliferating interstitial fibroblasts. • In RAS, we have found this to eventually result in chronic fibrotic patterns of intra-alveolar fibroelastosis and non-specific interstitial pneumonia (NSIP). • Time will tell if a similar progression to fibrosis will take place in a subset of survivors of severe COVID- 19, and whether this is indeed secondary to vascular damage, primarily related to the initial epithelial infection, or a combination of these response patterns.
- 26. OTHERS CORONA VIRUS: • Apart from the prominent microvascular changes, the pathological features of COVID- 19 thereby appear to resemble those seen in SARS and Middle Eastern respiratory syndrome (MERS) coronavirus infection. • Early SARS-CoV infections were found to be typified by acute diffuse alveolar damage, and late stages by a combination of diffuse alveolar damage and acute fibrinous and organizing pneumonia.
- 27. OTHERS CORONA VIRUS: • MERS-CoV infections also showed exudative diffuse alveolar damage with hyaline membranes, pulmonary edema, type II pneumocyte hyperplasia, (lymphocytic) interstitial pneumonia, and multinucleated syncytial cells.
- 28. FUTURE GUIDANCE: • Poor oxygenation requiring ventilation and oxygen administration in influenza as well as coronavirus infection is likely to be in part directly related to decreased diffusion capacity due to parenchymal destruction as well as increased diffusion distances due to 1) intra-alveolar aggregates of edema, fibrin, hyaline membranes, cells and organization 2) widening of alveolar septa due to edema and inflammation, 3) vascular changes with capillary congestion and thrombosis and 4) eventually, intrabronchiolar, intra-alveolar and interstitial fibrosis.
- 29. FUTURE GUIDANCE: • While regeneration and reversal of the first 3 phenomena is theoretically possible, and as in influenza may be related to the regenerative capacity of the host, it may occur at lower rates in SARS-CoV-2 infection as compared to other viral pneumonias. • Intra-alveolar and interstitial fibrosis, however, are most likely irreversible, and may have contributed to the remarkably high number of patients who cannot be weaned off ventilation in the current COVID-19 outbreak.
- 30. FUTURE GUIDANCE: • Also, in survivors we may eventually see a significant number of cases with irreversible fibrotic lung damage and limited pulmonary function, which has previously been termed COLD (‘Corona Obstructive Lung Disease’). • To which extent this term truly reflects the underlying physiology remains to be seen, as the interstitial fibrotic changes which have thus far been observed are likely to also lead to a restrictive lung defect as well as permanently decreased diffusion capacity, and are thus more akin to other types of (idiopathic) interstitial lung fibrosis, such as forms of the abovementioned intra-alveolar fibroelastosis and fibrotic NSIP, and could therefore perhaps be more accurately termed Corona-Associated Lung Disease (‘CALD’). This will require multi-center studies which correlate histopathological findings with clinical and radiological data.
- 31. WHY COVID-19 AFFECTS MALE: • Interestingly, as an X-linked phenotype, the effectiveness of interaction-booster and interaction-inhibitor variants of ACE2 can be more definite in males than females, and could contribute towards a higher mortality rate in males, accounting for up to ∼70% of death caused by SARS-CoV2, SARS-CoV or MERS-CoV. • ACE inhibitors or ARB increases the ACE2 receptor thereby increasing the viral entry. • However it would to premature to say that ACE2 inhibitors should be stopped.
- 33. • In the lung tissue of patients who died of respiratory failure due to COVID19 pneumonia, there can be evidence of epithelial infection with • cytopathic effects of pneumocytes (A, box); denudation of bronchiolar epithelium (B), evidence of diffuse alveolar damage (DAD) with hyaline membrane formation with organization (C). • Observed vascular changes include extensive bilateral and diffuse (micro)vascular damage and its sequelae, with arterial thrombosis with organization (D), microvascular fibrinoid change with hyaline thrombi (E, fibrin-Lendrum (MSB) stain, arrows) and edema (**), and extensive intra- alveolar fibrinous aggregates (F) with an acute fibrinous and organizing pneumonia (AFOP) pattern (F, *). COVID-19 :
- 34. • Fibrotic changes vary in appearance and include organizing pneumonia with progression to fibrosis (G), intra-alveolar fibro-elastosis (Elastic-van Gieson, H) and fibrotic non-specific interstitial pneumonia (F-NSIP; I). • There is often relatively sparse interstitial chronic inflammation with an acute interstitial pneumonia pattern (AIP; J), foci of lymphocytic vasculitis (K) and acute inflammation, especially in relation to areas of necrosis and possible secondary infection (L). COVID-19 :
- 35. Thank You!!
