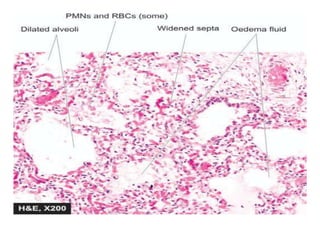
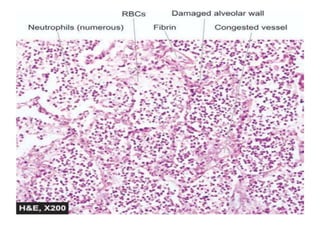
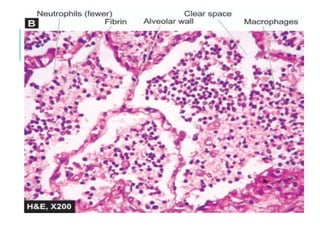
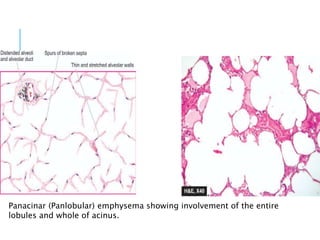
This document discusses pneumonia and emphysema. It provides details on pneumonia, including the pathogenesis, etiology, classification, and features of lobar pneumonia. Pneumonia is defined as acute lung inflammation distal to the terminal bronchioles. It is commonly caused by bacteria, viruses, or other factors. Lobar pneumonia specifically involves inflammation of an entire lung lobe. It is usually caused by Streptococcus pneumoniae and presents as distinct pathological phases from congestion to resolution. Complications can include organization of exudate, pleural effusions, empyema, or lung abscesses.