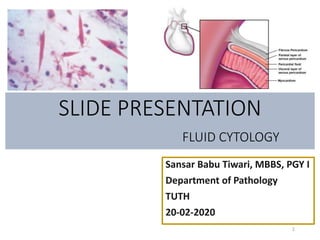
Pericardial fluid eosinophilia
- 1. SLIDE PRESENTATION FLUID CYTOLOGY Sansar Babu Tiwari, MBBS, PGY I Department of Pathology TUTH 20-02-2020 1
- 2. Slide Details • One Giemsa stained and another Pap stained smear of pericardial fluid • 7 Years old male child
- 3. Description: • Effusion smears from the pericardial fluid show mostly eosinophils with lymphocytes, scattered neutrophils and macrophages along with few reactive mesothelial cells in a proteinaceous background. • Abundant extracellular and few phagocytosed Charcot Leyden crystals are also seen. • Parasites are not seen in the examined smears. • Atypical cells are not seen.
- 4. Description: • PBS findings: • HB: 12.7 gm/dl • WBC: 10300 • N 46 L 30 M 04 E 20 • Platelet: 297000
- 9. DIAGNOSIS: DIAGNOSIS: EOSINOPHIL RICH EFFUSION. Differentials: 1. Parasitic infections: • Filariasis • Hydatid Disease 2. Allergic or Hypersensitivity: • Drug induced (Ceftriaxone) • Food allergy 3. Autoimmune Disorders: • Eosinophilic Granulomatosis with Polyangitis (EGPA) 4. Clonal proliferation: • Chronic Eosinophilic leukemia • T-cell Lymphoma 5. Idiopathic
- 15. BODY CAVITIES: Histology • Although mesothelial cells are derived from mesoderm, they demonstrate many morphological and biological features of epithelial cells.
- 17. BODY CAVITIES: Cytology • Mesothelial cells round-up and appear polyhedral after exfoliation due to surface tension of the surrounding fluid. • 1.5 to 2 times the size of the neutrophils. • When the differential is performed, all nucleated cells are counted, including mesothelial cells and malignant cells
- 18. BODY CAVITIES: Cytology • With pericardial fluid, a WBC count of greater than 1000 cells/µl is suggestive of pericarditis. • Pleural fluids: • Neutrophils predominate in 90% of acute inflammation. • Lymphocytes in TB, neoplasms and systemic diseases. • Peritoneal fluids: • Neutrophils predominate in 25% • Lymphocytes in TB, neoplasms and lymphatic obstruction.
- 19. BODY CAVITIES: Cytology • Pericardial fluids: • Differential is seldom performed as it has little diagnostic value. • Overall the cell types are: • Neutrophils • Eosinophils • Lymphocytes • Monocytes • Macrophages • Plasma cells • Mesothelial cells • Malignant cells
- 23. BODY CAVITIES: • Lined by a thin layer of connective tissue composed of flat mesothelial layer. • The cavity fluid is created and maintained through plasma ultrafiltration in the parietal membrane and absorption by the visceral membrane. • Serous fluid have a composition similar to that of serum.
- 24. BODY CAVITIES: • Normally serous fluids do not contain blood or fibrinogen • But a traumatic puncture procedure, hemorrhagic effusion, or an active bleed (e.g., from a ruptured blood vessel) can result in serous fluid that appears bloody and clots spontaneously. • Therefore, to prevent clot formation and entrapment of cells and micro-organisms, EDTA is preferred.
- 25. BODY CAVITIES: • Refrigeration ( 4 to 8°c) adversely affects the viability of microorganisms and should not be used for serous fluid specimens. • However, serous fluid samples intended for cytology examination can be refrigerated at 4°c when storage is necessary.
- 26. BODY CAVITIES:
- 27. BODY CAVITIES:
- 29. BODY CAVITIES: • Mesothelial cells can vary in appearance, such as appearing singly or in clumps, can be multi-nucleated, and can show reactive or degenerative changes, mesothelial cells can be difficult to differentiate from malignant cells and macrophages.
- 30. BODY CAVITIES:
- 31. BODY CAVITIES:
- 32. BODY CAVITIES:
- 33. BODY CAVITIES: Mesothelial markers • Positive markers: • Calretinin • D2-40 (podoplanin) • CK 5/6 • WT1 • Negative markers: • Ber-EP4 • mCEA • MOC-31 • BG-8 • B72.3
- 34. BODY CAVITIES: Mesothelial markers
- 35. BODY CAVITIES: LYMPHOCYTE RICH EFFUSION
- 36. Hyper-eosinophilia: • HE in a peripheral blood is defined as an absolute eosinophil count (AEC) > 1500/µL) on two examinations separated in time by at least one month and/or pathological confirmation of tissue HE. • Tissue HE is defined by: • On bone marrow, eosinophil percentage that exceeds 20% of all nucleated cells and/or • Tissue infiltration that is extensive in the opinion of pathologist and/or • Marked deposition of eosinophil granule proteins in tissue (demonstrated by IF)
- 37. Hyper-eosinophilic Syndrome: • HES is defined by the association of HE with eosinophil mediated organ damage and/or dysfuntion, provided other potential causes for the damage have been excluded. • Restrictive cardiac disease occurring in the setting of parasitic infections. [Loefflers endocarditis] • In contrast Persistent HE and thoracic pain secondary to coronary artery occlusion shouldn't be diagnosed as HES.
- 38. Hyper-eosinophilic Syndrome: •Primary HES (Neoplastic) •Secondary (Reactive) •Parasitic infestations •Solid tumors •T cell lymphoma •Idiopathic HES
- 39. HES: Pathophysiology • Mechanism of eosinophil overproduction: • Clonal eosinophilic proliferation as a result of primary molecular defect involving hematopoietic stem cells FIP1LI-PDGFRA PDGFRB FGFR1 PCM1-JAK2 • Overproduction of eosinophilic cytokines such as IL-5 Lymphocytic variant HES (L-HES): CD3- CD4+ phenotype of T cell
- 40. Organ-restricted HES • Blood eosinophilia > 1500/µl in the setting of single organ involvement. • Eosinophilic gastrointestinal diseases • Chronic eosinophilic pneumonia • Well’s syndrome
- 41. HES: Clinical features A retrospective multicenter study on 188 patients: • Dermatological (rash) 37 % • Pulmonary (cough and breathlessness) 25 % • GI 14% • Cardiac 5% • Neurologic 4%
- 43. HES: Pathophysiology • Cardiac Diseases: • Eosinophilic myocarditis is a major cause of morbidity and mortality in HES • PDGFRA associated HES has more predilection to heart • The severity of cardiac injury does not correlate with the degree of peripheral eosinophilia. • In EGPA: cardiac involvement of the myocardium and pericardium is due to small vessel vasculitis that impairs cardiac function without the development of endomyocardial thrombi and fibrosis.
- 44. HES: Pathophysiology • Cardiac Diseases: • Three stages: • Acute necrotic stage: Endocardial damage, myocardial infiltration with eosinophils and lymphocytes, myocardial necrosis (Usually clinically silent). Trop I can be used as a indicator of damage in HES and EGPA. ECHO may be normal. Endomyocardial biopsy can be done with MBP-1 immunostain even if eosinophils are not seen in the sections examined. • Thrombus formation: Eosinophil peroxidase forms hypothiocyanous acid which inturn induces tissue factor expression. Embolic stroke can be the presentation. • Fibrotic stage: Fibro-inflammatory remodeling can lead to rupture of chordae tendinae or fusion of valves to the endocardial surface. Dyspnea, chest pain, MR, TR, cardiomegaly, restrictive cardiomyopathy can occur.
- 47. Pericardial effusion: Causes • Normal fluid volume is 10-50 ml. • Causes: • Infection • Malignancy • Trauma • Uremia • Hypothyroidism • Autoimmune disorders Exudates Transudates
- 49. Pericardial effusion: Eosinophilia • > 10% eosinophil is considered pleural and pericardial fluid eosinophilia. • Pericardial fluid eosinophilia is a rare entity and correlates with disease severity and mortality. • Causes: • Trauma (Haemothorax and pneumothorax) • Allergic reactions • Parasitic infections
- 52. B
- 53. Eosinophilic Effusion: Case Series of 12 cases • 2005-2018 (13 years) • Retrospective study • Rashid Hospital, UAE (Dubai) • 12 patients who demonstrated eosinophilic fluids • Constituted • 0.5% of 2305 total serous body cavity effusion fluids. • 0.7% of 1800 nonmalignant effusions. • 5 pleural, 5 peritoneal and 2 pericardial. • 4 ( >95%), 2 (> 50%) and 6 (<50%). • 1 case without peripheral blood eosinophilia. • 2 cases showed Charcot-Leyden crystals (Galectin-10).
- 54. Eosinophilic Effusion: Case Series of 12 cases • The higher the fluid eosinophilia the less likely the cause to be malignancy or tuberculosis. • Pleural eosinophilia is frequently associated with traumatic pleural effusions making the assumption that introduced air and blood act as a stimulant for eosinophilic pleuritic. • This is also unlikely in pericardial and peritoneal EEs since most are spontaneous. • The significant presence of lymphocytosis in eosinophilic fluids may suggest a role played by T helper lymphocytes and their interleukins in inducing eosinophilia.
- 55. Eosinophilic Effusion: Case Series of 12 cases • Eosinophilic pericardial effusions may indicate a life-threatening complication of eosinophilic pericarditis secondary to eosinophilic myocarditis due to hypereosinophilic syndrome.
- 57. Pericardial Eosinophilia: Case Report • 16 Years-old girl • Chest pain, palpitations, dyspnea, dizziness, nausea, low-grade fever, vomiting and fatigue for 2 days • An allergy to seafood, similar episode 1 month earlier • No history of asthma, drugs intake, skin rash or diarrhea • Initial impression: Food poisoning, lung infection, tuberculosis or gastritis
- 58. Pericardial Eosinophilia: Case Report • Chest X-ray showed marked pericardial effusion with clear lungs • USG showed mild to moderate pleural effusion and ascites • WBC: 14200 (Eosinophils: 5600 i.e. 40%) • PBS: Mild reactive eosinophilia without blast cells • LFT, RFT, TFT, ESR and CRP normal
- 59. Pericardial Eosinophilia: Case Report • Serological tests for viruses, mycoplasma, brucella, toxoplasma, parasites and autoantibodies for autoimmune diseases were negative. • Mantoux test negative • Stool and urine test for parasites, ova, eosinophils and crystals are negative
- 60. Pericardial Eosinophilia: Case Report • Developed cardiac tamponade and heart failure • Pericardiocentesis was performed and a 250 ml of cloudy bloody fluid was aspirated • Cytological examination of the pericardial fluid revealed a mixed inflammatory infiltrate and florid reactive mesothelial hyperplasia • Predominantly eosinophils with small lymphocytes, macrophages, and neutrophils. • Diamond shaped crystals were also seen.
- 61. Pericardial Eosinophilia: Case Report • Pleural fluid showed reactive mesothelial cells, small lymphocytes and macrophages without eosinophils and neutrophils. • The clinical diagnosis of eosinophilic myopericarditis of the restrictive cardiomyopathy type in a setting of hyper- eosininophilic syndrome. • Churg-Strauss syndrome was excluded because the patient had no history of asthma, no evidence of clinical manifestations of vasculitis, pulmonary infiltrate and paranasal sinus polyps.
- 62. Pericardial Eosinophilia: Case Report • The patient improved after pericardial drainage and systemic steroid therapy. The patient was asymptomatic and the pericardial effusion and peripheral eosinophilia improved after 6 weeks follow up. • D/D: • Parasitic infestations • Drugs • HES • Churg-Strauss syndrome • Restrictive cardiomyopathy.
- 63. TAKE HOME MESSAGE 1. Serous lining is developed from mesoderm but resembles that of epithelial lining. 2. Obstruction of drainage of serous fluid lead to lymphocyte rich effusion. 3. Pericardial fluid eosinophilia is a rare entity. 4. Charcot-leyden crystals are formed by combinations of galectin-10 proteins from eosinophils. 5. Cardiac involvement in HES usually occurs in FIP1LI-PDGFRA1. 6. Microfilariae and hooklets can be seen.
- 64. Thankyou!!!!
