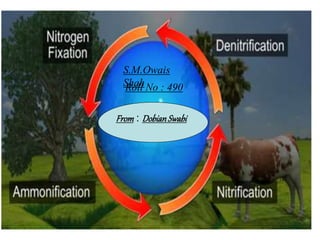
Nitrogen
- 1. From : DobianSwabi S.M.Owais ShahRoll No : 490
- 3. • Nitrogen (N) is an essential component of DNA, RNA, and proteins, The building blocks of life. • All organisms require nitrogen to live and grow. Nitrogen
- 4. The majority (78%) of the Earth’s atmosphere is N2.
- 7. Nitrogen Fixation (N2 --> NH3 or NH4 +) HOW? Biological or Environmental or Humans BIOLOGICAL: Bacteria (called Nitrogen- fixing bacteria) These bacteria form symbiotic relationships with host plants. The bacteria live in nodules found in the roots of the legume family of plants (e.g. beans, peas, and clover) Nitrogen Fixation (N2 --> NH3 or NH4 +) HOW? Biological or Environmental or Humans BIOLOGICAL: Bacteria (called Nitrogen- fixing bacteria) These bacteria form symbiotic relationships with host plants. The bacteria live in nodules found in the roots of the legume family of plants (e.g. beans, peas, and clover)
- 8. Nitrogen Fixation (N2 --> NH3 or NH4 +) ENVIRONMENTAL High-energy natural events which break the bond N2 Examples: lightning forest fires hot lava flows
- 9. Nitrogen Fixation N2 --> NH3 or NH4 + How? HUMAN IMPACT • Burning fossil fuels, • using synthetic nitrogen fertilizers, • and cultivation of legumes all fix nitrogen.
- 10. Nitrogen Mineralization also called Ammonification Organic N --> NH4 + • Decay of dead things, manure, etc. • Done by decomposers (bacteria, fungi, etc.) • During this process, a significant amount of the nitrogen contained within the dead organism is converted to ammonium (NH4 +).
- 11. Nitrification NH3 or NH4 + --> NO2 - --> NO3 - (Nitrifying) Bacteria add oxygen to nitrogen. Two steps: • STEP 1: Bacteria take NH3 or NH4 + & make NO2 - = nitrite • Step 2: • Bacteria take in NO2 - • & make NO3 - = nitrate
- 14. • Yellow arrows indicate human sources of nitrogen to the environment. Red arrows indicate bacterial transformations of nitrogen. Blue arrows indicate physical forces acting on nitrogen. And green arrows indicate natural, non- microbial processes affecting the form and fate of nitrogen.
- 15. • References Burford, J.R., and J.M. Bremner. 1975. Relationships between the nitrification capacities of soils and total, water-soluble and readily decomposable soil organic matter. Soil Biochem. 7:389-394. • Marschner, H. 1995. Mineral Nutrition in Higher Plants. Academic Press, London. • Tisdale, S.L., W.L. Nelson, J.D. Beaton, and J.L. Havlin. 1993. Soil Fertility and Fertilizers. MacMillan Publishing Co., New York, N.Y.
