Gene therapy: a cure for disease
- 1. Gene therapy
- 2. Introduction In medicine, gene therapy (also called human gene transfer) is the therapeutic delivery of nucleic acid into a patient's cells as a drug to treat disease.[ The first attempt at modifying human DNA was performed in 1980 by Martin Cline, but the first successful nuclear gene transfer in humans, approved by the National Institutes of Health, was performed in May 1989.[2] The first therapeutic use of gene transfer as well as the first direct insertion of human DNA into the nuclear genome was performed by French Anderson in a trial starting in September 1990. Between 1989 and February 2016, over 2,300 clinical trials had been conducted, more than half of them in phase I.
- 4. Background Gene therapy was conceptualized in 1972, by authors who urged caution before commencing human gene therapy studies. After extensive research on animals throughout the 1980s and a 1989 bacterial gene tagging trial on humans, the first gene therapy widely accepted as a success was demonstrated in a trial that started on 14 September 1990, when Ashi DeSilva was treated for ADA-SCID.
- 5. Somatic Germline Cell types In somatic cell gene therapy (SCGT), the therapeutic genes are transferred into any cell other than a gamete, germ cell, gametocyte, or undifferentiated stem cell. Any such modifications affect the individual patient only, and are not inherited by offspring. Somatic gene therapy represents mainstream basic and clinical research, in which therapeutic DNA (either integrated in the genome or as an external episome or plasmid) is used to treat disease. In germline gene therapy (GGT), germ cells (sperm or egg cells) are modified by the introduction of functional genes into their genomes. Modifying a germ cell causes all the organism's cells to contain the modified gene. The change is therefore heritable and passed on to later generations. Australia, Canada, Germany, Israel, Switzerland, and the Netherlands prohibit GGT for application in human beings, for technical and ethical reasons, including insufficient knowledge about possible risks to future generations and higher risks versus SCGT. The US has no federal controls specifically addressing human genetic modification (beyond FDA regulations for therapies in general).
- 6. Speculative uses Fertility- Gene Therapy techniques have the potential to provide alternative treatments for those with infertility. Recently, successful experimentation on mice has proven that fertility can be restored by using the gene therapy method, CRISPR. Spermatogenical stem cells from another organism were transplanted into the testes of an infertile male mouse. The stem cells re-established spermatogenesis and fertility.
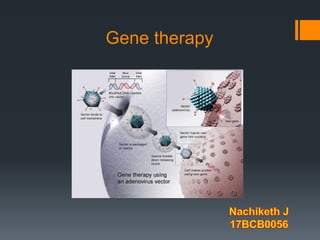
- 7. How does gene therapy work? A gene that is inserted directly into a cell usually does not function. Instead, a carrier called a vector is genetically engineered to deliver the gene. Certain viruses are often used as vectors because they can deliver the new gene by infecting the cell. The viruses are modified so they can't cause disease when used in people. Some types of virus, such as retroviruses, integrate their genetic material (including the new gene) into a chromosome in the human cell. Other viruses, such as adenoviruses, introduce their DNA into the nucleus of the cell, but the DNA is not integrated into a chromosome
- 8. 2017 Was the Year of Gene- Therapy Breakthroughs Decades in the making, gene therapy—the idea of modifying a person’s DNA to treat disease—represents a major shift in medicine. Instead of just treating symptoms like the vast majority of drugs on the market, gene therapy aims to correct the underlying genetic cause of a disease. Doctors and scientists hope these treatments will be a one-shot cure. Last year, we wrote that 2016 was gene therapy’s most promising year. But 2017 proved to be even bigger.
- 9. Cancer killers This year the FDA approved two pioneering treatments, Kymriah and Yescarta, that use a patient’s own immune cells to fight rare types of cancer. Called CAR-T therapies, these “living drugs” are made by extracting T cells from patients and genetically engineering them to go after and destroy cancer cells. The cells are then infused back into the body. So far, these therapies are being tested only in a handful of lethal cancers as a last resort when more traditional treatments, like chemotherapy, don’t work. Kymriah treats a bone marrow cancer that affects children and young adults, and Yescarta treats a type of lymphoma. Some patients have had remarkable recoveries and remain in remission months or years later.
- 10. Sickle-cell cure Researchers announced that a teenage boy in France had been cured of sickle-cell disease after receiving an experimental gene therapy developed by Bluebird Bio. Caused by a single genetic mutation, sickle-cell is an inherited blood disorder that affects 100,000 people in the U.S. and millions around the world. Scientists removed stem cells from the boy’s bone marrow and modified them in the lab by introducing copies of a gene to prevent his red blood cells from becoming “sickled.” When the treated cells were infused back into his body, they began to make normal blood cells.
- 11. Speculative uses Gene doping- Athletes might adopt gene therapy technologies to improve their performance. Gene doping is not known to occur, but multiple gene therapies may have such effects. Kayser et al. argue that gene doping could level the playing field if all athletes receive equal access. Critics claim that any therapeutic intervention for non- therapeutic/enhancement purposes compromises the ethical foundations of medicine and sports.
- 12. Human genetic engineering Genetic engineering could be used to cure diseases, but also to change physical appearance, metabolism, and even improve physical capabilities and mental faculties such as memory and intelligence. Ethical claims about germline engineering include beliefs that every foetus has a right to remain genetically unmodified, that parents hold the right to genetically modify their offspring, and that every child has the right to be born free of preventable diseases.] For parents, genetic engineering could be seen as another child enhancement technique to add to diet, exercise, education, training, cosmetics, and plastic surgery. Another theorist claims that moral concerns limit but do not prohibit germline engineering
- 13. The cost of gene therapy Many genetic disorders that can be targeted with gene therapy are extremely rare. Gene therapy therefore often requires an individual, case-by- case approach. This may be effective, but may also be very expensive. Making sure the new gene doesn’t disrupt the function of other genes ideally, a new gene introduced by gene therapy will integrate itself into the genome of the patient and continue working for the rest of their lives. There is a risk that the new gene will insert itself into the path of another gene, disrupting its activity.
- 14. Regulations Regulations covering genetic modification are part of general guidelines about human-involved biomedical research. There are no international treaties which are legally binding in this area, but there are recommendations for national laws from various bodies. The Helsinki Declaration (Ethical Principles for Medical Research Involving Human Subjects) was amended by the World Medical Association's General Assembly in 2008. This document provides principles physicians and researchers must consider when involving humans as research subjects. The Statement on Gene Therapy Research initiated by the Human Genome Organization (HUGO) in 2001 provides a legal baseline for all countries. HUGO’s document emphasizes human freedom and adherence to human rights, and offers recommendations for somatic gene therapy, including the importance of recognizing public concerns about such research