Ring opening polymerization using N-heterocyclic olefins
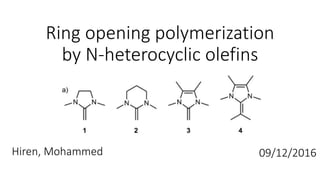
- 1. Ring opening polymerization by N-heterocyclic olefins Hiren, Mohammed 09/12/2016
- 2. Paper 1 – N-heterocyclic olefins as organic catalysts for polymerization: preparation of well defined poly(propylene oxide) Paper 2 – Highly polarized alkenes as organocatalysts for the polymerization of lactones and trimethylene carbonate
- 3. Essential points • Advantages of using organic catalysts • Differences in the catalysts • Mechanisms • HNMR
- 4. • N-heterocyclic olefins • Used alongside benzyl alcohol initiator to ring open propylene oxide, lactones and trimethylene carbonate. • Over the 2 papers there are 5 catalysts • Anionic ring opening polymerization
- 5. Paper 1
- 6. Catalysts 1-4 + monomer Propylene oxide
- 8. Catalyst 3 • Less control over the reaction • Produced a primary carbanion • Impurities in the polymer produced • 2 proposed mechanisms
- 10. CATALYST 4 • More control over the reaction • It forms a tertiary carbanion which favors the anionic polymerization over the zwitterionic polymerization. • Abstracts protons from the initiator better than catalyst 3, as it’s a stronger base.
- 12. Changing the concentration of initiator
- 13. HNMR Green = aromatic Red = methylene unit BnOH
- 14. Advantages of 4 over 3 • Stronger base • More stable, able to predict the outcome • Produces less impurities in polymer
- 15. Paper 2
- 16. N-heterocyclic olefins -More active than NHCs in ROP. -Exocyclic carbon partially anionic. -Ring size + substitutions affect the activity. -The stability of the carbanion. - S. Naumann, A. W. Thomas, and A. P. Dove, ACS Macro Lett. 2016, 5
- 17. The monomers used: L- lactide Delta- valerlactone Trimethyl carbonate(TMC) ω- pentadecalactone (PDL) ε-caprolactone
- 18. S. Naumann, A. W. Thomas, and A. P. Dove, ACS Macro Letters. 2016, 5
- 19. S. Naumann, A. W. Thomas, and A. P. Dove, ACS Macro Letters. 2016, 5 The deactivation of the catalyst occurred. Substitutions avoid the deprotonation.
- 20. S. Naumann, A. W. Thomas, and A. P. Dove, ACS Macro Letters. 2016, 5 High Conversion (No BnOH). The ratio increases the activity. No improvement in control. Broad Mwt distribution. Low catalyst loading.
- 22. Advantages of NHO’s • Less toxic than organometallic compounds. • They produce a high conversion with and without initiators. • Easy to edit the structure in order to change the activity. • ability to operate under alternative polymerization mechanisms.
