Hyperconjugation by konain z.m.
•Download as PPTX, PDF•
1 like•932 views
slides on hyperconjugation by konain
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (20)
Nucleophilic substitution sn1 sn2 nucleophile halogenoalkane in organic chemi...

Nucleophilic substitution sn1 sn2 nucleophile halogenoalkane in organic chemi...
Diastreoslectivity,chemoslectivity&;regioslectivity crams rule felkin anh m...

Diastreoslectivity,chemoslectivity&;regioslectivity crams rule felkin anh m...
Mechanism of the reaction between hydrogen and bromine

Mechanism of the reaction between hydrogen and bromine
Electronic effects inductive,mesomeric,electromeric

Electronic effects inductive,mesomeric,electromeric
Similar to Hyperconjugation by konain z.m.
Similar to Hyperconjugation by konain z.m. (20)
organic chemistry lecture module - Alkyl halides.pdf

organic chemistry lecture module - Alkyl halides.pdf
2020_Reaction_of_Carbocations_Carbenes_and_Radicals_ALL.pdf

2020_Reaction_of_Carbocations_Carbenes_and_Radicals_ALL.pdf
Aromatic Electrophilic Substitution Reactions (1).pdf

Aromatic Electrophilic Substitution Reactions (1).pdf
Recently uploaded
Rohini Sector 26 Call Girls Delhi 9999965857 @Sabina Saikh No Advance

Rohini Sector 26 Call Girls Delhi 9999965857 @Sabina Saikh No AdvanceCall Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
VIP Call Girls Pune Madhuri 8617697112 Independent Escort Service Pune

VIP Call Girls Pune Madhuri 8617697112 Independent Escort Service PuneCall girls in Ahmedabad High profile
Recently uploaded (20)
'Future Evolution of the Internet' delivered by Geoff Huston at Everything Op...

'Future Evolution of the Internet' delivered by Geoff Huston at Everything Op...
Challengers I Told Ya ShirtChallengers I Told Ya Shirt

Challengers I Told Ya ShirtChallengers I Told Ya Shirt
VIP 7001035870 Find & Meet Hyderabad Call Girls LB Nagar high-profile Call Girl

VIP 7001035870 Find & Meet Hyderabad Call Girls LB Nagar high-profile Call Girl
Delhi Call Girls Rohini 9711199171 ☎✔👌✔ Whatsapp Hard And Sexy Vip Call

Delhi Call Girls Rohini 9711199171 ☎✔👌✔ Whatsapp Hard And Sexy Vip Call
GDG Cloud Southlake 32: Kyle Hettinger: Demystifying the Dark Web

GDG Cloud Southlake 32: Kyle Hettinger: Demystifying the Dark Web
Low Rate Young Call Girls in Sector 63 Mamura Noida ✔️☆9289244007✔️☆ Female E...

Low Rate Young Call Girls in Sector 63 Mamura Noida ✔️☆9289244007✔️☆ Female E...
VIP 7001035870 Find & Meet Hyderabad Call Girls Dilsukhnagar high-profile Cal...

VIP 7001035870 Find & Meet Hyderabad Call Girls Dilsukhnagar high-profile Cal...
VIP Kolkata Call Girl Kestopur 👉 8250192130 Available With Room

VIP Kolkata Call Girl Kestopur 👉 8250192130 Available With Room
Call Girls In Mumbai Central Mumbai ❤️ 9920874524 👈 Cash on Delivery

Call Girls In Mumbai Central Mumbai ❤️ 9920874524 👈 Cash on Delivery
Call Girls Dubai Prolapsed O525547819 Call Girls In Dubai Princes$

Call Girls Dubai Prolapsed O525547819 Call Girls In Dubai Princes$
Chennai Call Girls Alwarpet Phone 🍆 8250192130 👅 celebrity escorts service

Chennai Call Girls Alwarpet Phone 🍆 8250192130 👅 celebrity escorts service
VIP Kolkata Call Girls Salt Lake 8250192130 Available With Room

VIP Kolkata Call Girls Salt Lake 8250192130 Available With Room
Call Girls In Defence Colony Delhi 💯Call Us 🔝8264348440🔝

Call Girls In Defence Colony Delhi 💯Call Us 🔝8264348440🔝
Rohini Sector 26 Call Girls Delhi 9999965857 @Sabina Saikh No Advance

Rohini Sector 26 Call Girls Delhi 9999965857 @Sabina Saikh No Advance
AWS Community DAY Albertini-Ellan Cloud Security (1).pptx

AWS Community DAY Albertini-Ellan Cloud Security (1).pptx
Russian Call Girls in Kolkata Samaira 🤌 8250192130 🚀 Vip Call Girls Kolkata

Russian Call Girls in Kolkata Samaira 🤌 8250192130 🚀 Vip Call Girls Kolkata
VIP Kolkata Call Girl Dum Dum 👉 8250192130 Available With Room

VIP Kolkata Call Girl Dum Dum 👉 8250192130 Available With Room
VIP Call Girls Pune Madhuri 8617697112 Independent Escort Service Pune

VIP Call Girls Pune Madhuri 8617697112 Independent Escort Service Pune
Networking in the Penumbra presented by Geoff Huston at NZNOG

Networking in the Penumbra presented by Geoff Huston at NZNOG
Hyperconjugation by konain z.m.
- 1. 6/22/2020 1
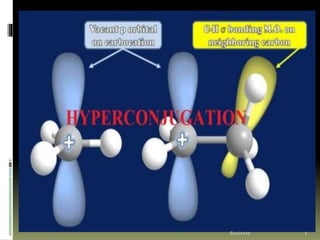
- 2. Hyper-conjugation Resonance involves delocalization of π electrons, leaving the σ bond untouched. However in some cases, a σ bond and an adjacent π bond may get involved in resonance. Such a delocalization is called as Hyperconjugation. It is a stabilizing effect of the molecule. 6/22/2020 2
- 3. Hyper-conjugation Hyperconjugation occur in following types of molecules such as; Carbocations free radical alkenes and alkynes. 6/22/2020 3
- 4. Hyperconjugation in carbocations: Hyperconjugation occur in carbocation for example in butyl carbocation. Butyl carbocation has 9 alpha hydrogen and hence 9 Hyperconjugation structures can be written. 6/22/2020 4
- 5. Hyperconjugation in free radicals: Hyper conjugation in free radicals for example in ethyl free radical. A radical is explained where there is an overlap between the p orbital occupied by the odd electron and σ orbital of the alkyl group (Hyperconjugation). 6/22/2020 5
- 6. Hyperconjugation in alkenes: Hyperconjugation in alkenes for example in propene. Hyper conjugation arise due to partial overlap of sp3 sigma bond orbital and the empty p- orbital of an adjacent carbon atom. Here one of the carbon-hydrogen bonds of methyl group can lie in the plane of pi-bond orbital, hence partial overlap with pi-bond orbital. 6/22/2020 6
- 7. Hyperconjugation in alkynes: Hyperconjugation in alkynes for example in propyne. Higher substituted alkynes are more stable than their lower substituted (terminal) isomers. The reason for this behavior is the Hyperconjugation of the π orbitals with the sp3 hybrid orbitals of the alkyl groups. 6/22/2020 7
- 8. 6/22/2020 8