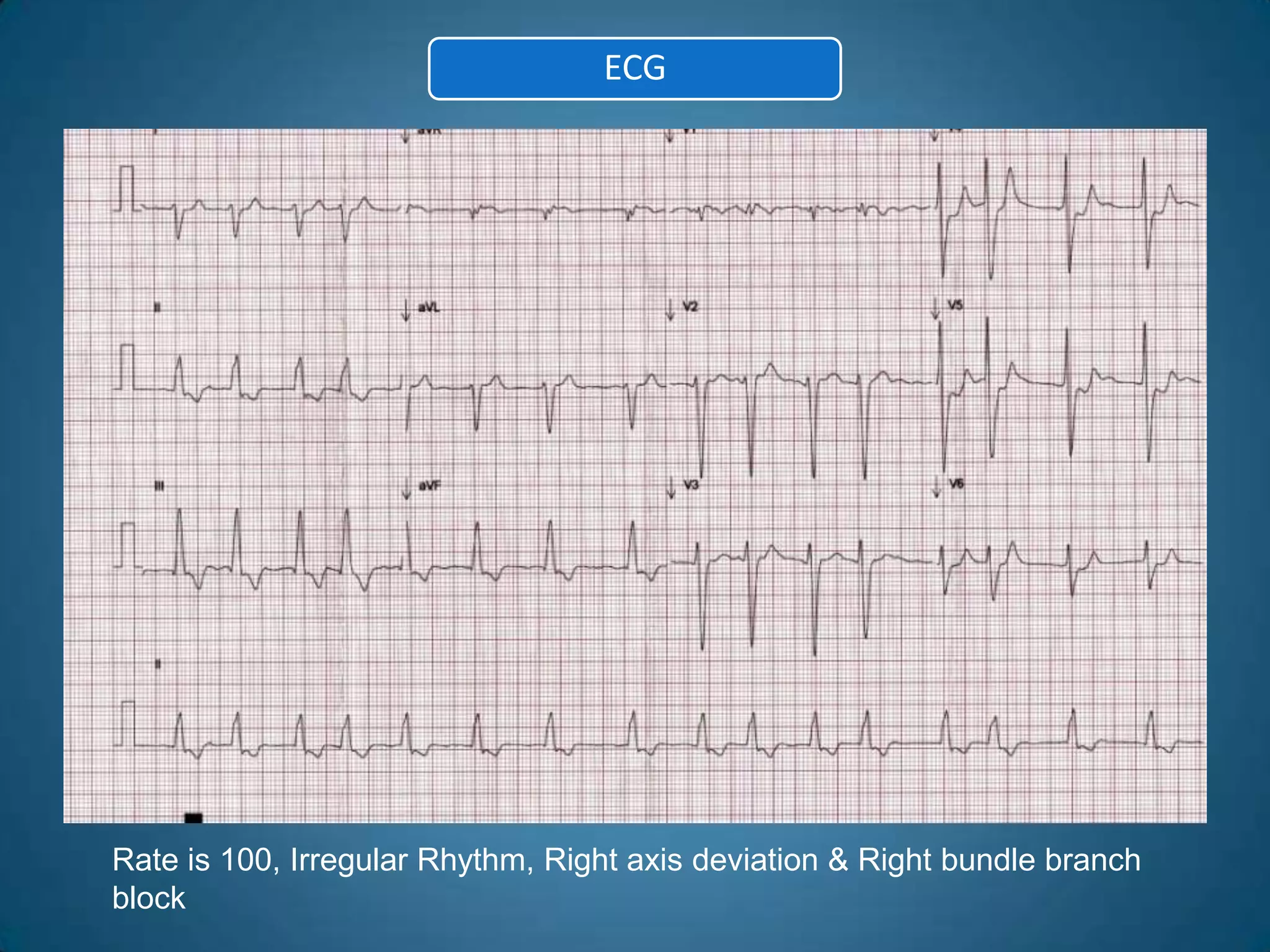
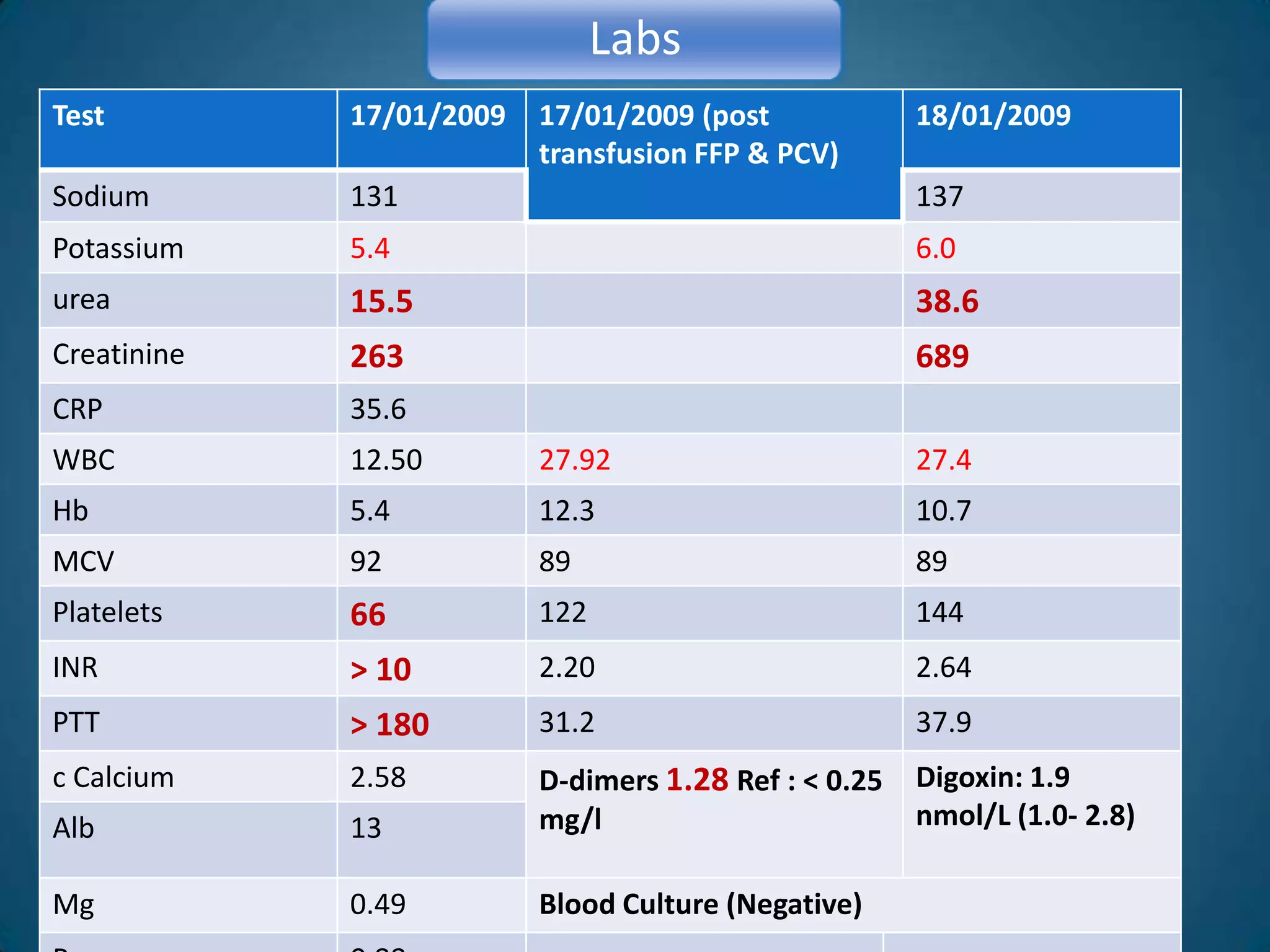
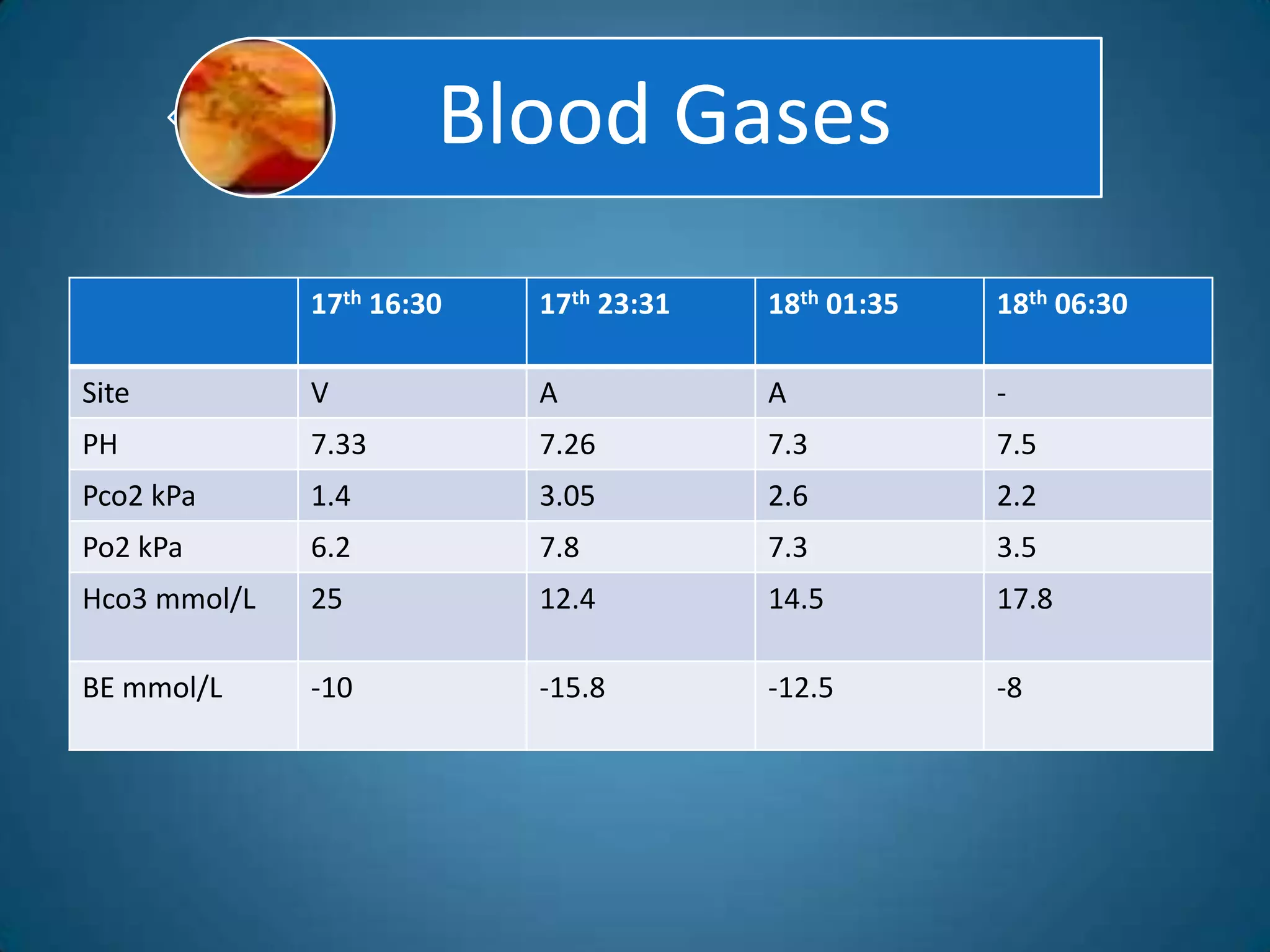
A 51-year-old woman with a history of valvular heart surgery in 2001 presented with symptoms of dizziness, weakness, loss of appetite, nausea, confusion and irritability. She had been taking warfarin 10 mg daily along with other medications. Examination found bradycardia, atrial fibrillation, and signs of internal bleeding. Laboratory tests showed severe warfarin toxicity with an INR over 10. She was diagnosed with warfarin toxicity and internal bleeding. Management included fluid resuscitation, blood transfusions, and ventilation support. However, she deteriorated and died the next day.















![Anticoagulation (Prosthetic heart valves)
The goal of anticoagulant therapy is to administer the lowest possible dose of
anticoagulant to prevent clot formation or expansion. The required degree of
anticoagulation continues to evolve as studies provide more information about the
efficacy and safety of lower doses*.
INR Duration
‡--Depending on the type of mechanical valve (i.e., caged ball or caged disk) and the valve
position (mitral), some patients may benefit from INRs in the upper end of the range.
* Stein PD, Alpert JS, Copeland J, Dalen JE, Goldman S, Turpie AG. Antithrombotic therapy in patients with
mechanical and biologic prosthetic heart valves. Chest 1995;108(4 Suppl):371S-9S [Published erratum in
Chest 1996;109:592].](https://image.slidesharecdn.com/warfrinmonitering-090616041729-phpapp01/75/Warfrin-Monitering-24-2048.jpg)


![Risk factor for Hemorrhagic Complications of
Anticoagulation
• The most common complication of warfarin therapy is bleeding, which
occurs in 6 to 39 percent of recipients annually **
Age greater than 65 years¹
Age greater than 75 years with
concomitant atrial fibrillation (intracranial hemorrhage)²
History of gastrointestinal bleeding³
Comorbid disease states*
Hypertension³
Cerebrovascular disease**
Serious heart disease
Renal insufficiency
¹ Landefeld CS, Rosenblatt MW, Goldman L. Major bleeding in outpatients treated with warfarin: incidence and
prediction by factors known at the start of outpatient therapy. Am J Med 1989;87:144-52.
² Albers GW. Atrial fibrillation and stroke: three new studies, three remaining questions. Arch Intern Med1994;154:1443-8 & Risk
factors for stroke and efficacy of antithrombotic therapy in atrial fibrillation: analysis of pooled data from five randomized
controlled trials. Arch Intern Med 1994;154:1449-57 [Published erratum in Arch Intern Med1994;10:2254].
³ Routledge PA, Chapman DH, Davies DM, Rawlins MD. Pharmacokinetics and pharmacodynamics of warfarin
at steady state. Br J Clin Pharmacol 1979; 8:243-7.
* Fihn SD, McDonell M, Martin D, Henikoff J, Vermes D, Kent D, et al. Risk factors for complications of
chronic anticoagulation: a multicenter study. Ann Intern Med 1993;118:511-20.
** Hirsh J, Dalen JE, Deykin D, Poller L, Bussey H. Oral anticoagulants: mechanism of action, clinical
effectiveness, and optimal therapeutic range. Chest 1995;108(4 Suppl):231S-46S.](https://image.slidesharecdn.com/warfrinmonitering-090616041729-phpapp01/75/Warfrin-Monitering-27-2048.jpg)