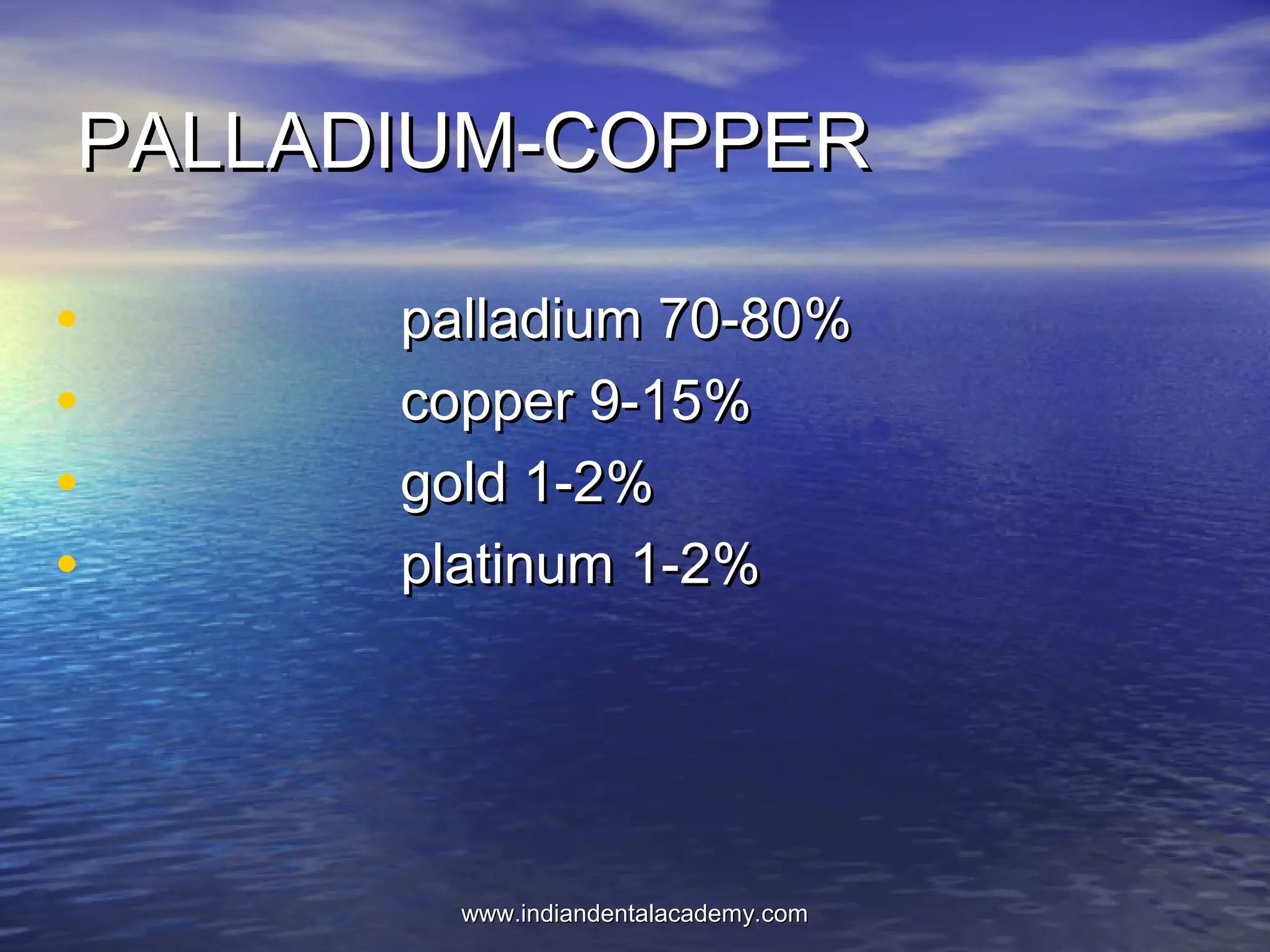
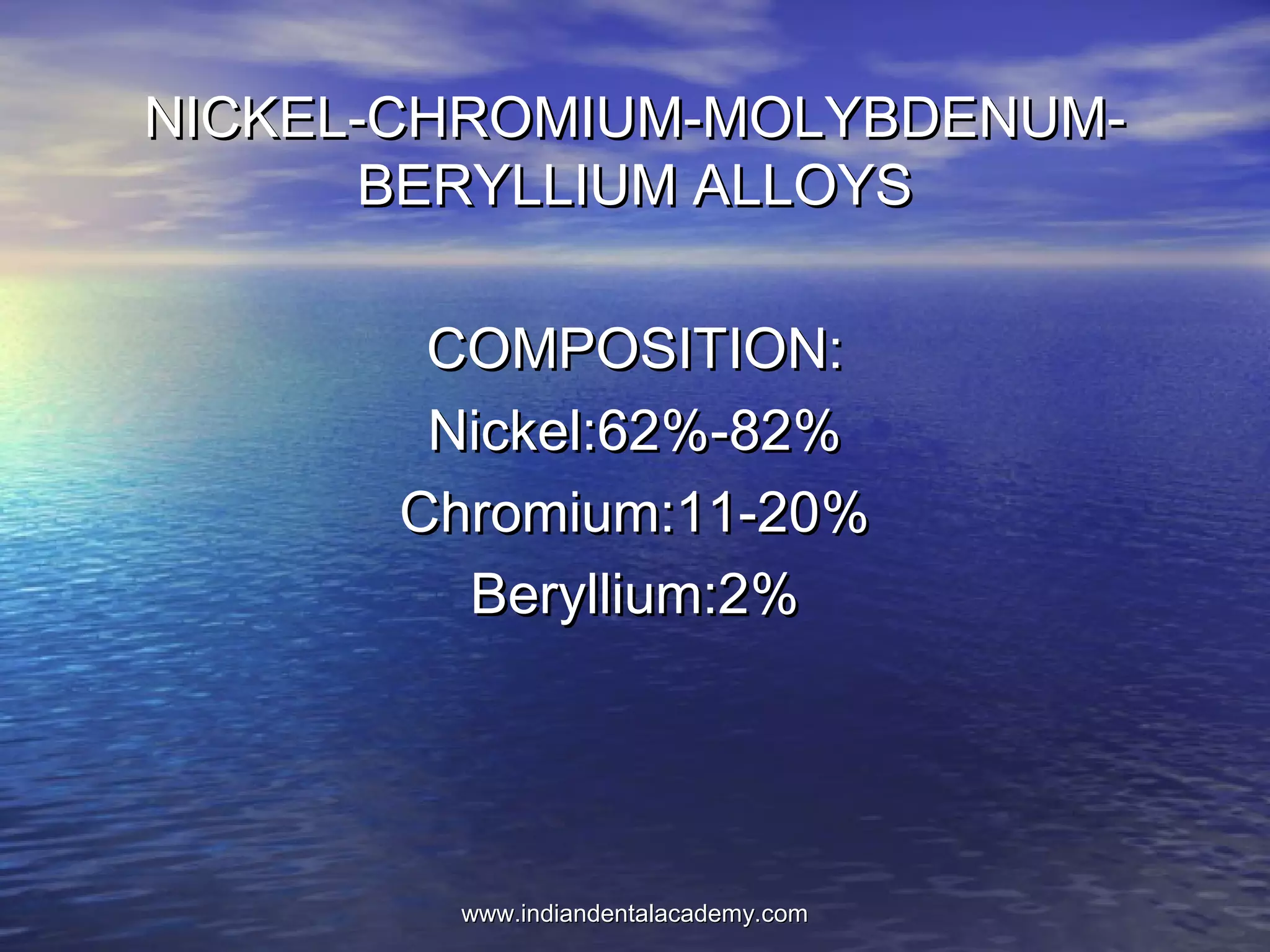
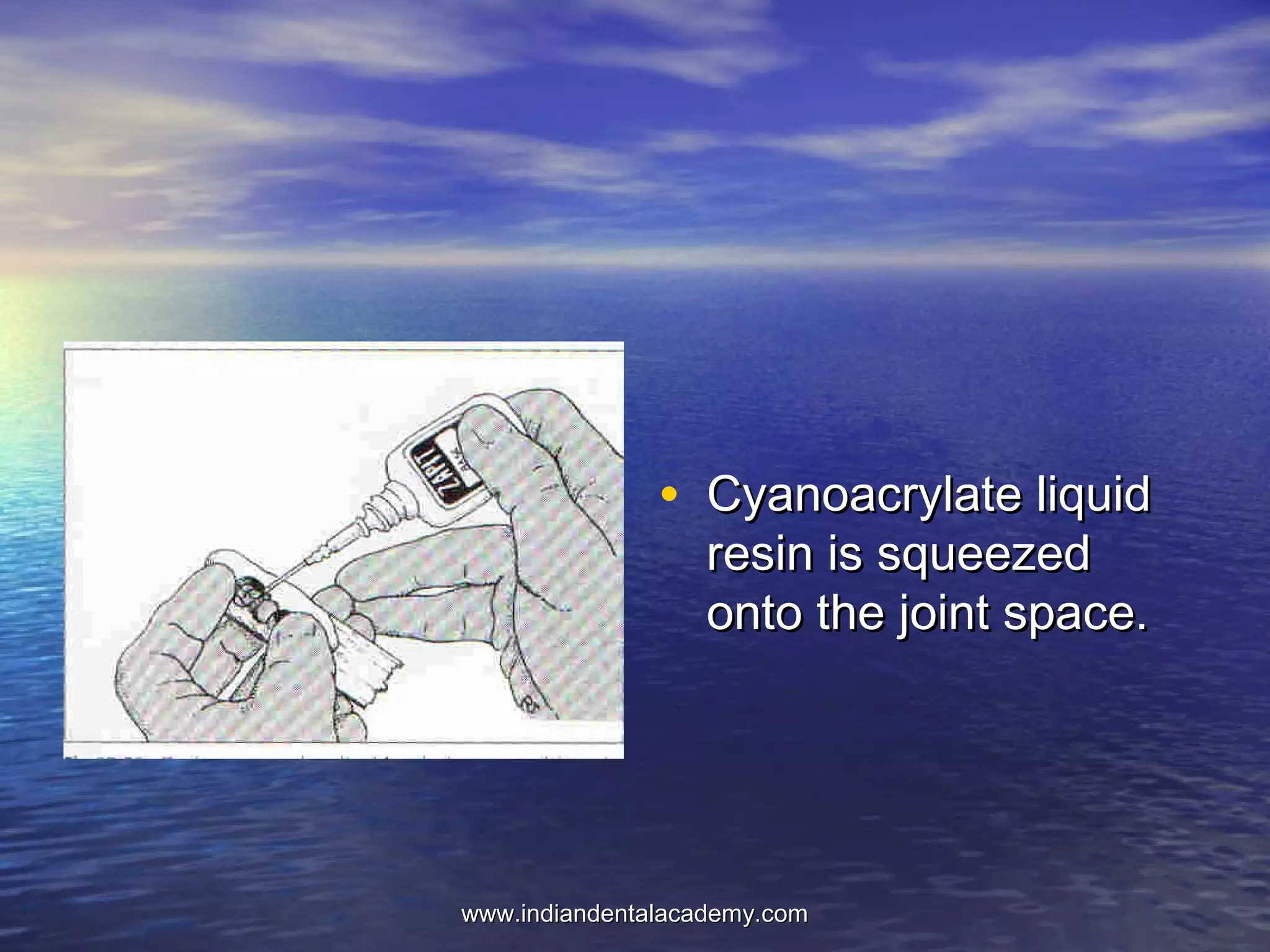
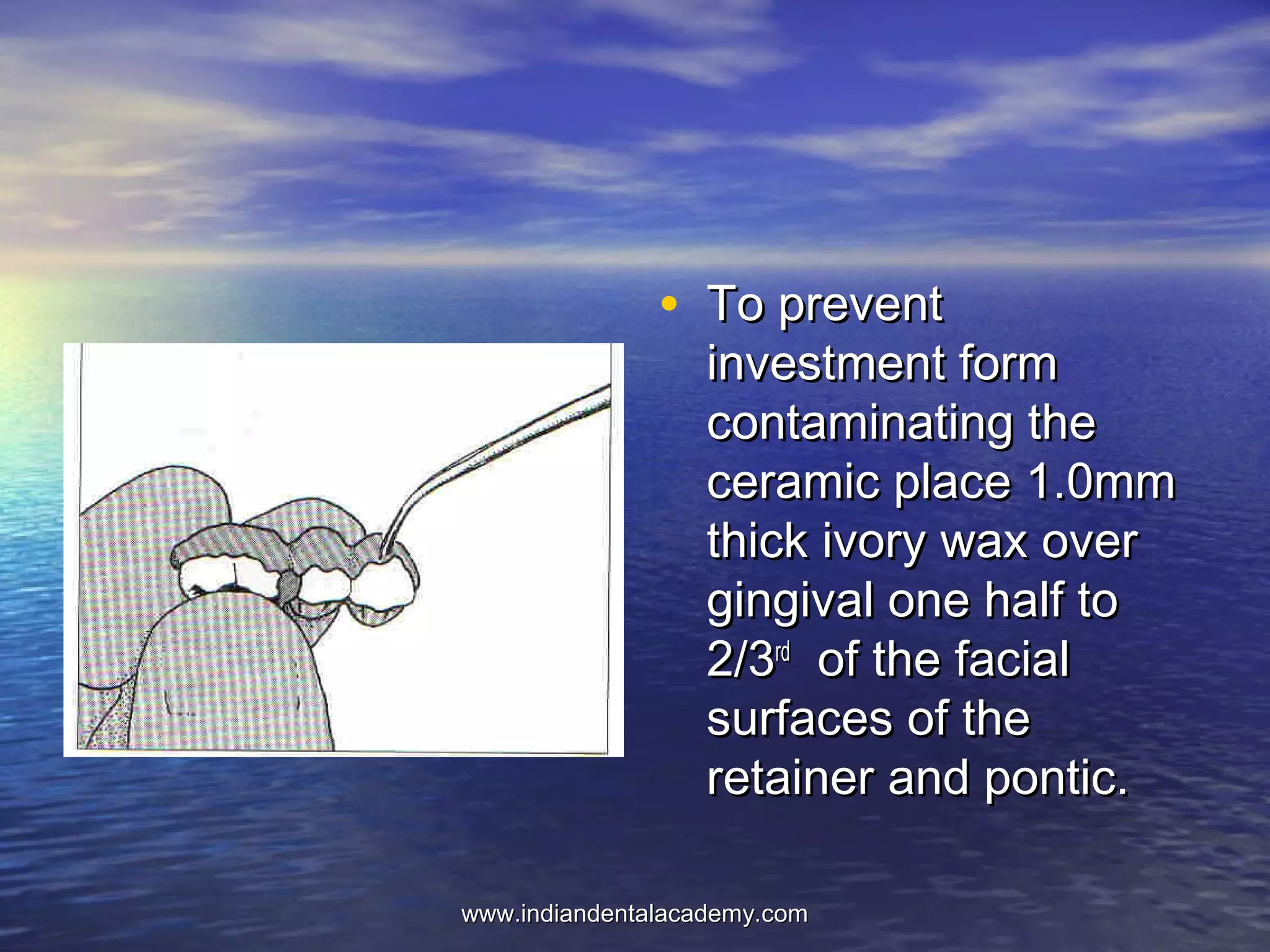
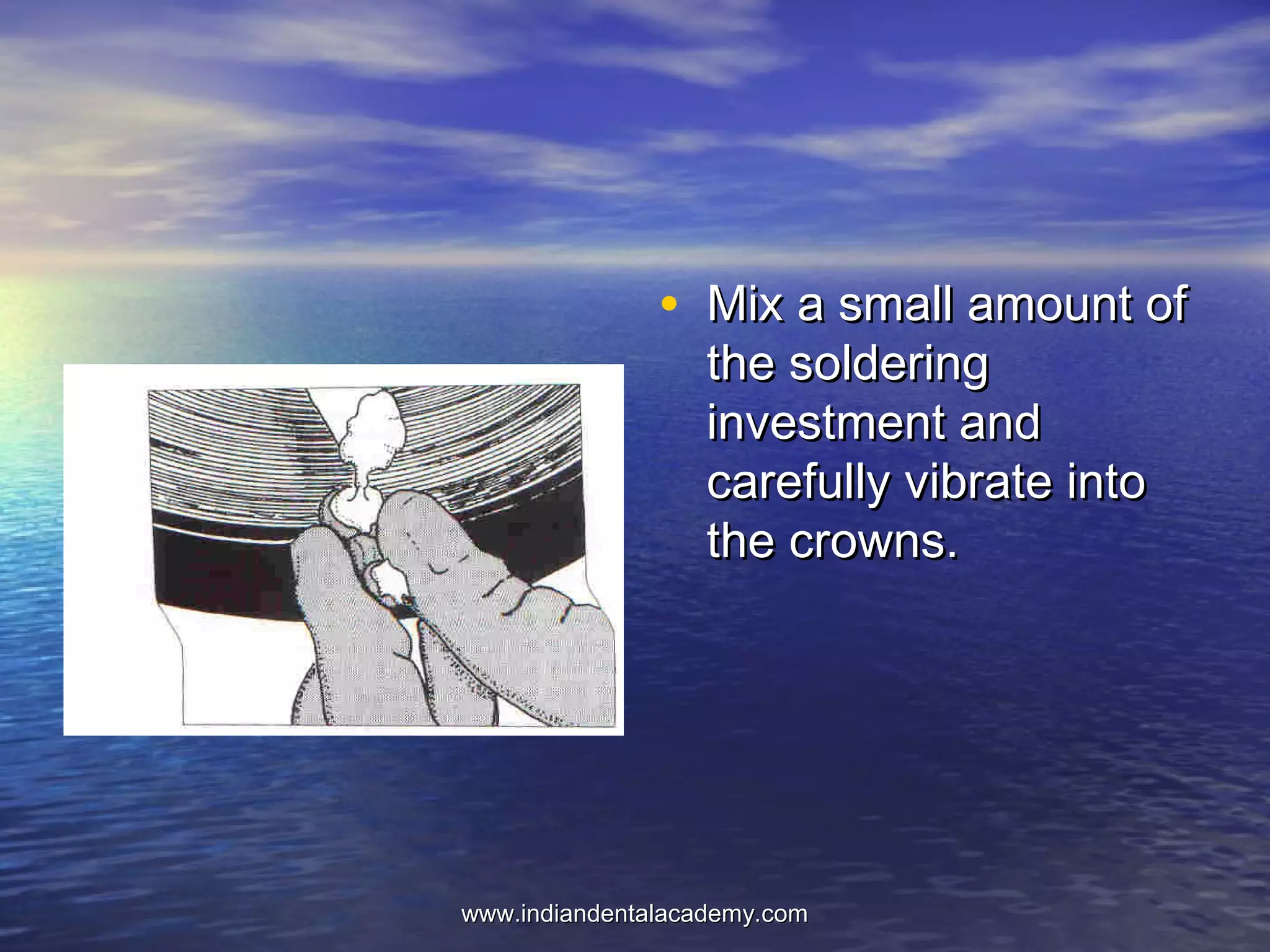
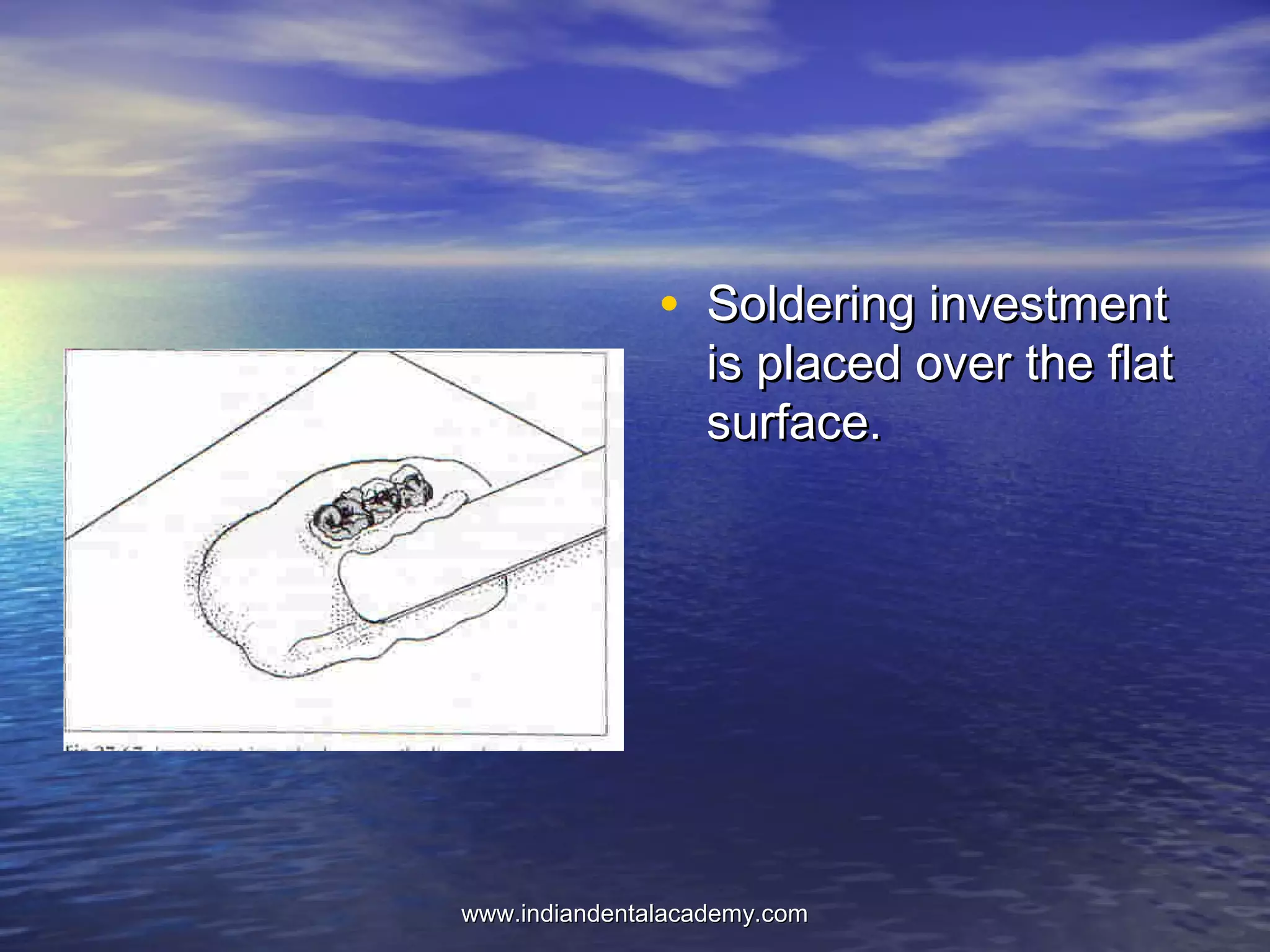
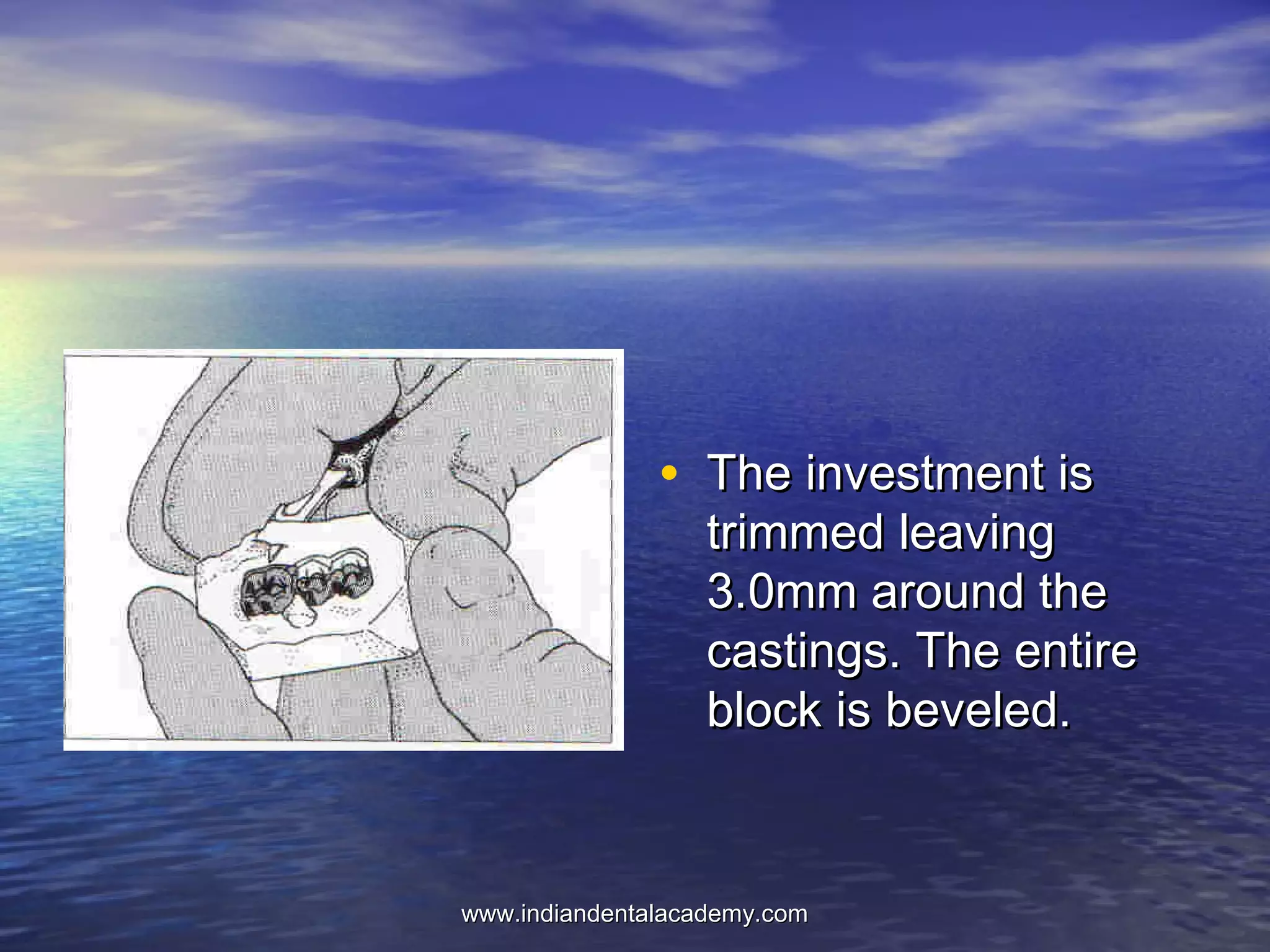
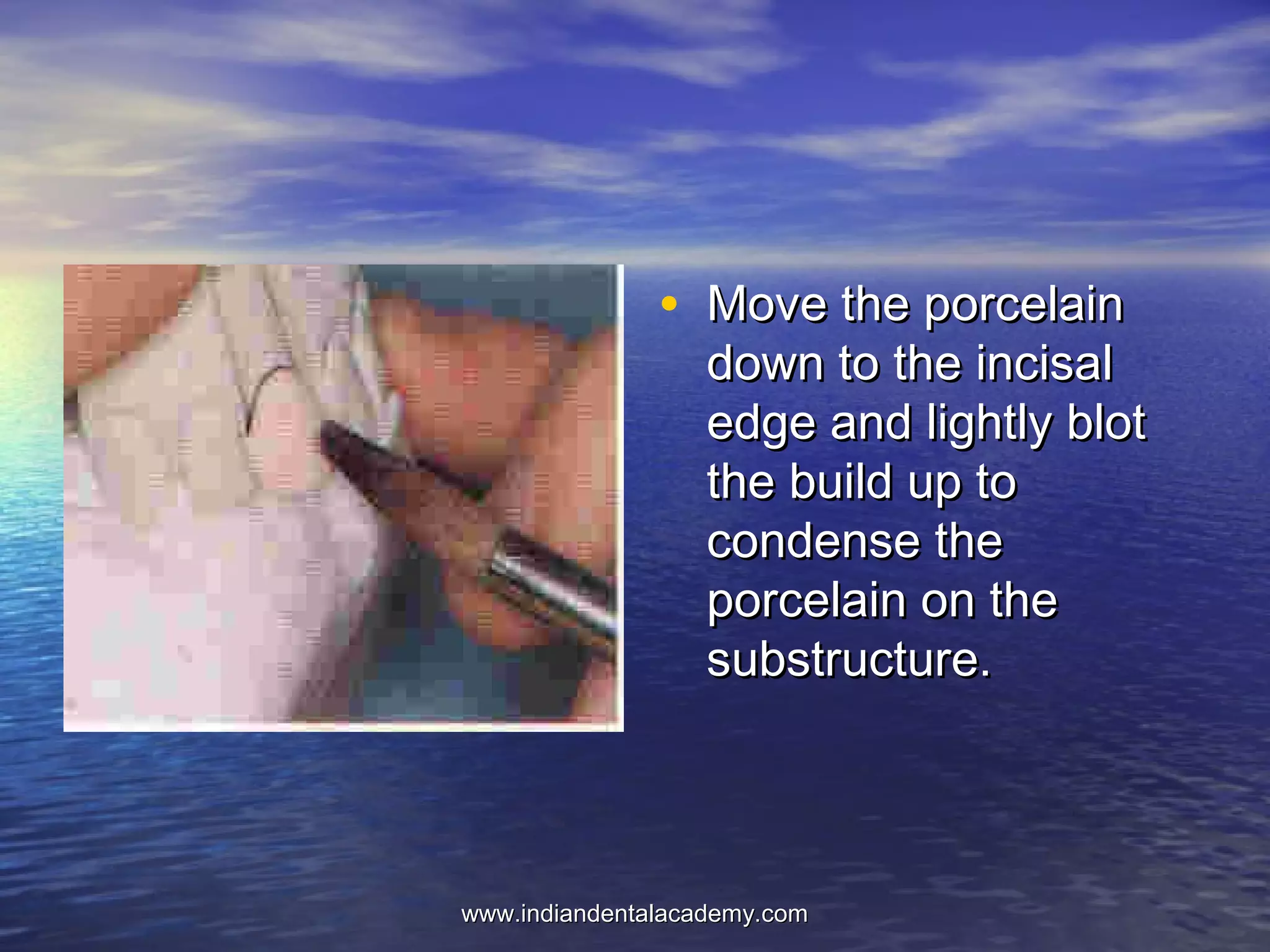
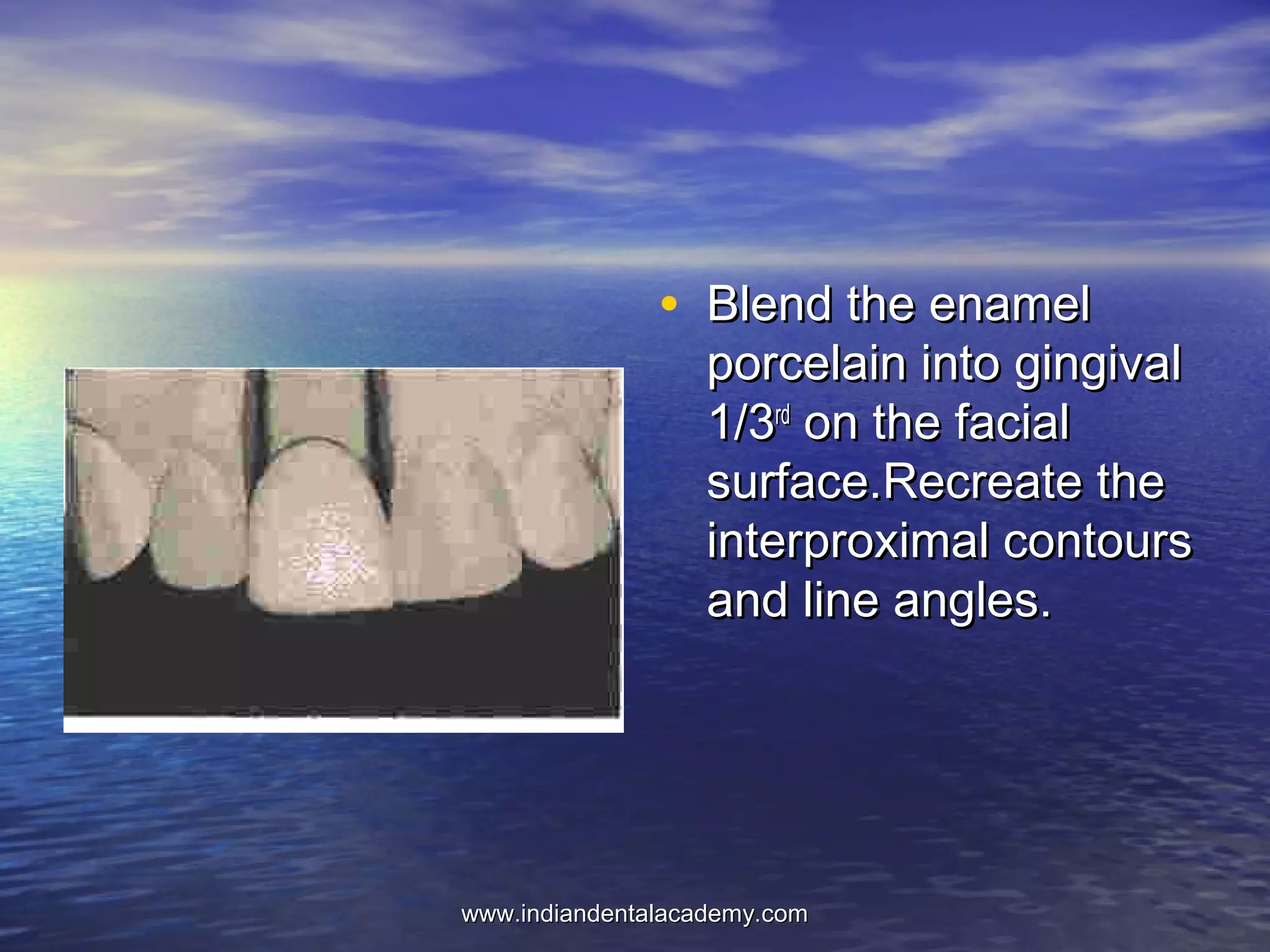
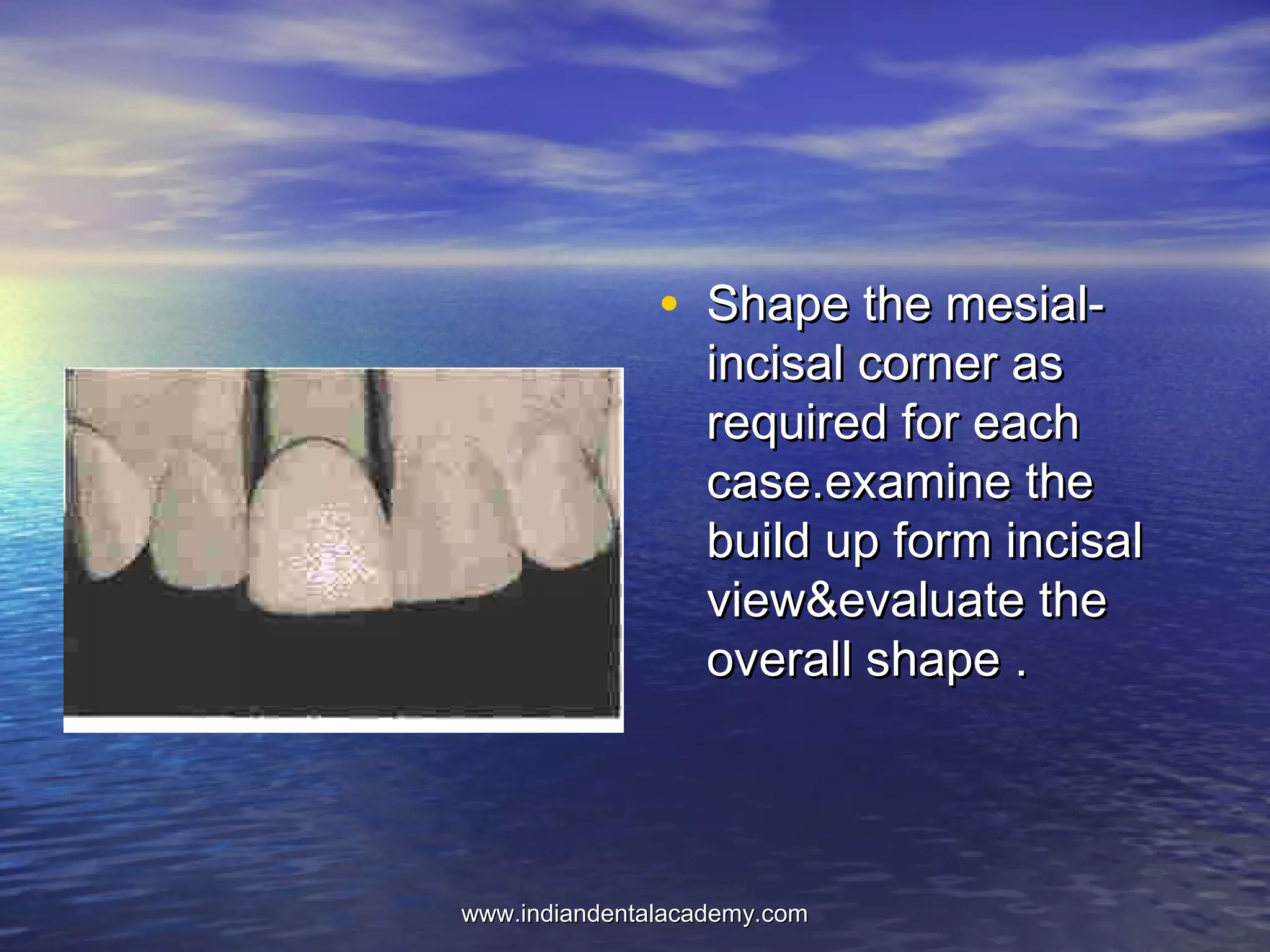
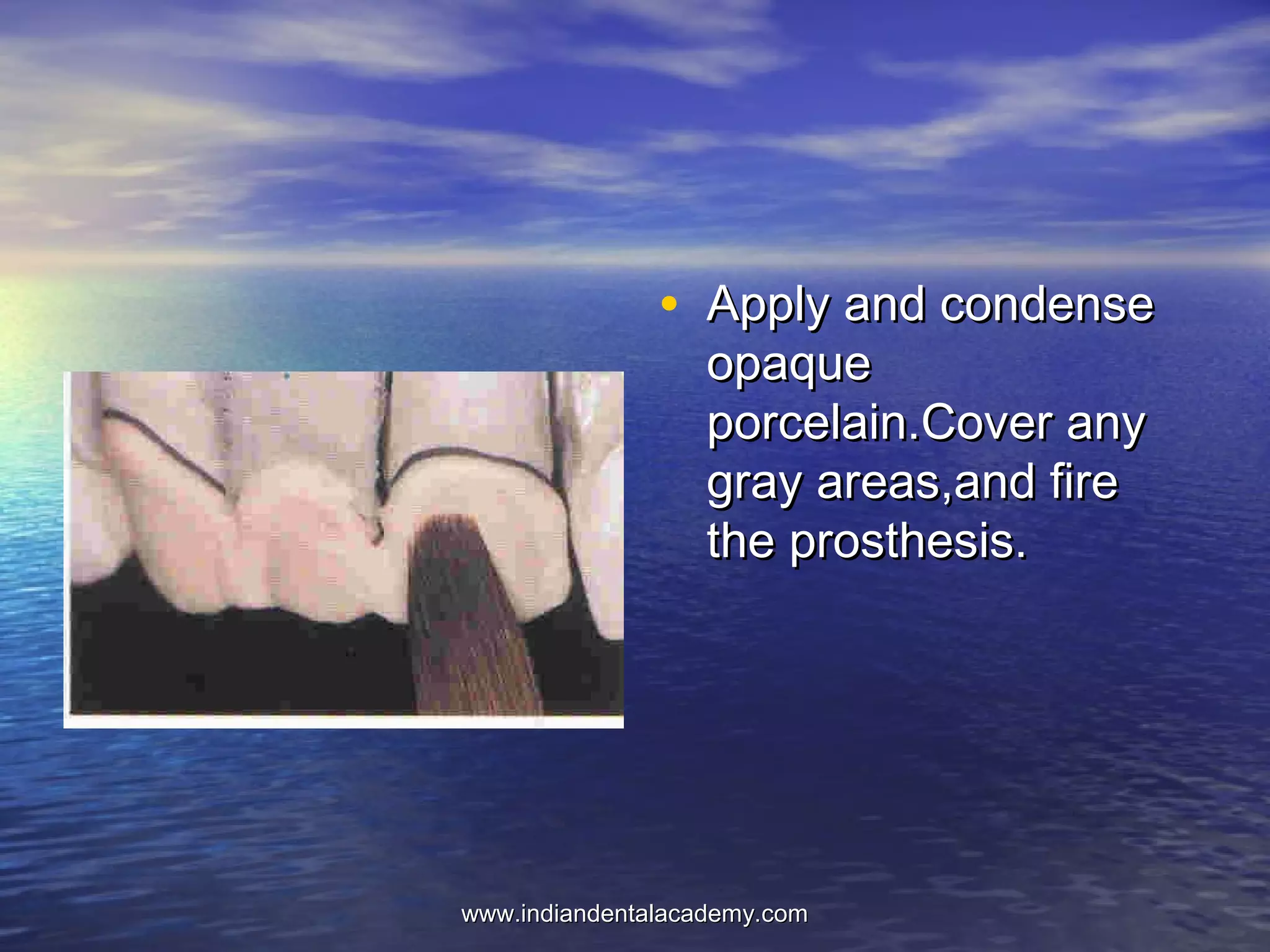
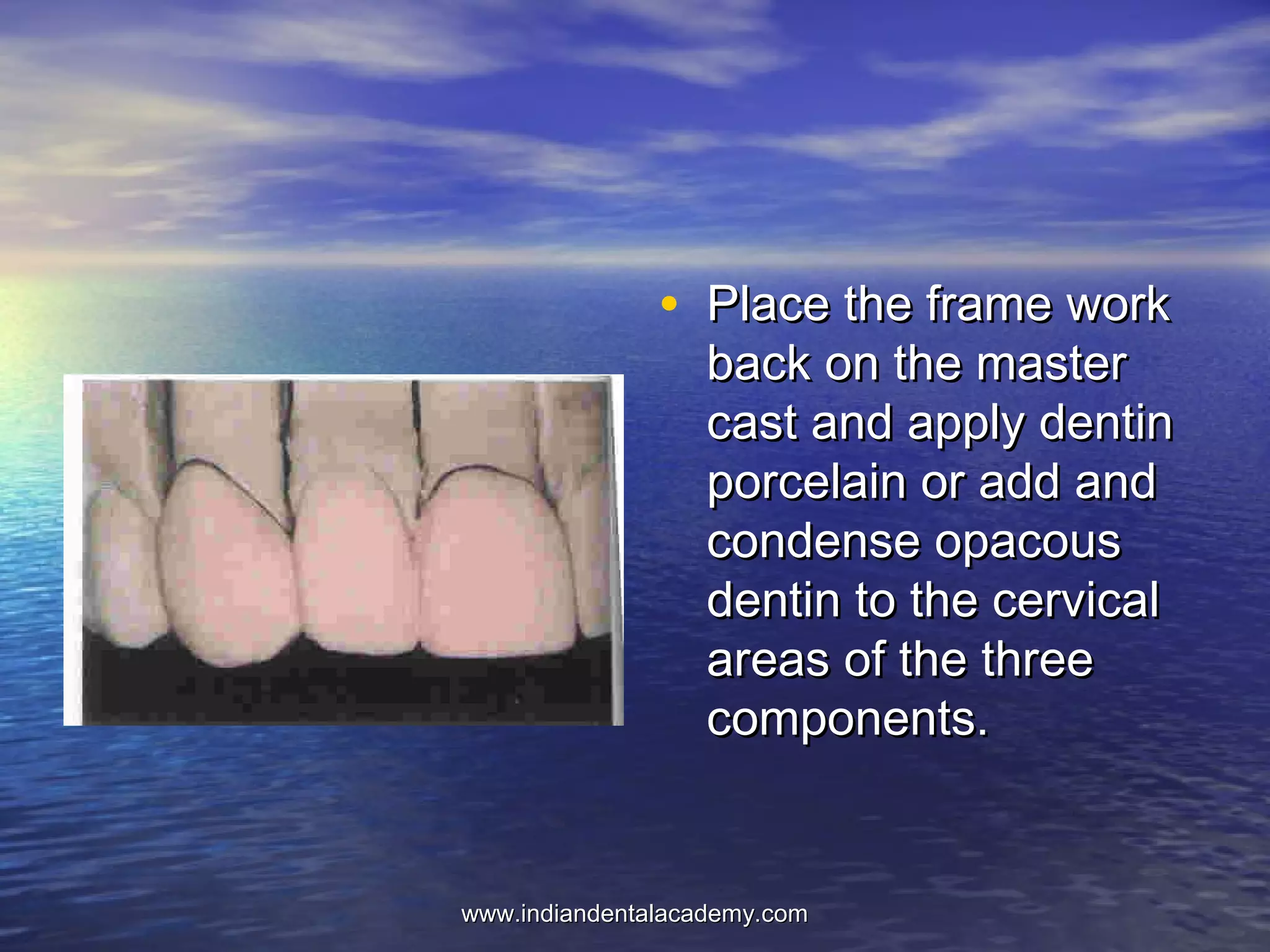
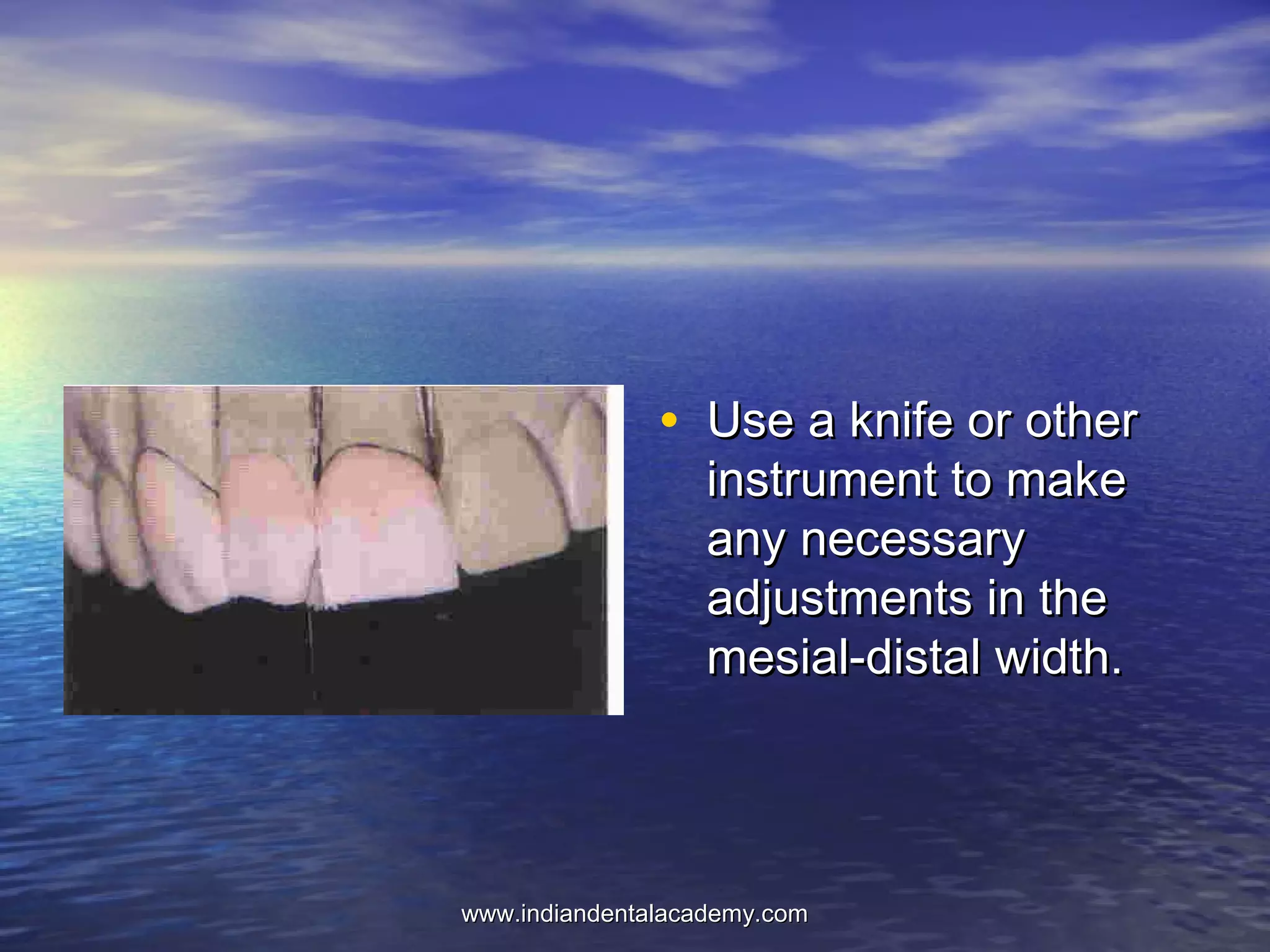
The document provides an extensive overview of dental casting alloys, including their classification, requirements, and advantages and disadvantages of different alloy systems. It categorizes the alloys based on function, color and composition, and outlines the American Dental Association (ADA) classification system for cast alloys. Additionally, it details the composition and properties of various alloy systems such as gold-platinum-palladium, palladium-silver, and others, emphasizing their use in dental applications.


































![• Cobalt is used for alternative of the nickelCobalt is used for alternative of the nickel
based alloys, but the cobalt based alloysbased alloys, but the cobalt based alloys
are difficult to process.]are difficult to process.]
• Cobalt is added to in palladium alloys toCobalt is added to in palladium alloys to
increase the CTE,& acts as a strengthener.increase the CTE,& acts as a strengthener.
www.indiandentalacademy.comwww.indiandentalacademy.com](https://image.slidesharecdn.com/castingalloys2-140807010737-phpapp02/75/Casting-alloys-dentistry-website-35-2048.jpg)