Isothermal Isobaric Isochoric Adiabatic Processes.pptx
•Download as PPTX, PDF•
0 likes•559 views
Thermodynamics unit for BTEC
Report
Share
Report
Share
More Related Content
What's hot
What's hot (20)
Similar to Isothermal Isobaric Isochoric Adiabatic Processes.pptx
Similar to Isothermal Isobaric Isochoric Adiabatic Processes.pptx (20)
Work done by constant volume and pressure using PV diagram

Work done by constant volume and pressure using PV diagram
Recently uploaded
Call Girls in Nagpur Suman Call 7001035870 Meet With Nagpur Escorts

Call Girls in Nagpur Suman Call 7001035870 Meet With Nagpur EscortsCall Girls in Nagpur High Profile
College Call Girls Nashik Nehal 7001305949 Independent Escort Service Nashik

College Call Girls Nashik Nehal 7001305949 Independent Escort Service NashikCall Girls in Nagpur High Profile
Recently uploaded (20)
Software Development Life Cycle By Team Orange (Dept. of Pharmacy)

Software Development Life Cycle By Team Orange (Dept. of Pharmacy)
Processing & Properties of Floor and Wall Tiles.pptx

Processing & Properties of Floor and Wall Tiles.pptx
(PRIYA) Rajgurunagar Call Girls Just Call 7001035870 [ Cash on Delivery ] Pun...![(PRIYA) Rajgurunagar Call Girls Just Call 7001035870 [ Cash on Delivery ] Pun...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![(PRIYA) Rajgurunagar Call Girls Just Call 7001035870 [ Cash on Delivery ] Pun...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
(PRIYA) Rajgurunagar Call Girls Just Call 7001035870 [ Cash on Delivery ] Pun...
VIP Call Girls Service Kondapur Hyderabad Call +91-8250192130

VIP Call Girls Service Kondapur Hyderabad Call +91-8250192130
(MEERA) Dapodi Call Girls Just Call 7001035870 [ Cash on Delivery ] Pune Escorts![(MEERA) Dapodi Call Girls Just Call 7001035870 [ Cash on Delivery ] Pune Escorts](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![(MEERA) Dapodi Call Girls Just Call 7001035870 [ Cash on Delivery ] Pune Escorts](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
(MEERA) Dapodi Call Girls Just Call 7001035870 [ Cash on Delivery ] Pune Escorts
Introduction to IEEE STANDARDS and its different types.pptx

Introduction to IEEE STANDARDS and its different types.pptx
IMPLICATIONS OF THE ABOVE HOLISTIC UNDERSTANDING OF HARMONY ON PROFESSIONAL E...

IMPLICATIONS OF THE ABOVE HOLISTIC UNDERSTANDING OF HARMONY ON PROFESSIONAL E...
Call Girls Delhi {Jodhpur} 9711199012 high profile service

Call Girls Delhi {Jodhpur} 9711199012 high profile service
Coefficient of Thermal Expansion and their Importance.pptx

Coefficient of Thermal Expansion and their Importance.pptx
VIP Call Girls Service Hitech City Hyderabad Call +91-8250192130

VIP Call Girls Service Hitech City Hyderabad Call +91-8250192130
Call Girls in Nagpur Suman Call 7001035870 Meet With Nagpur Escorts

Call Girls in Nagpur Suman Call 7001035870 Meet With Nagpur Escorts
College Call Girls Nashik Nehal 7001305949 Independent Escort Service Nashik

College Call Girls Nashik Nehal 7001305949 Independent Escort Service Nashik
What are the advantages and disadvantages of membrane structures.pptx

What are the advantages and disadvantages of membrane structures.pptx
ZXCTN 5804 / ZTE PTN / ZTE POTN / ZTE 5804 PTN / ZTE POTN 5804 ( 100/200 GE Z...

ZXCTN 5804 / ZTE PTN / ZTE POTN / ZTE 5804 PTN / ZTE POTN 5804 ( 100/200 GE Z...
HARDNESS, FRACTURE TOUGHNESS AND STRENGTH OF CERAMICS

HARDNESS, FRACTURE TOUGHNESS AND STRENGTH OF CERAMICS
Isothermal Isobaric Isochoric Adiabatic Processes.pptx
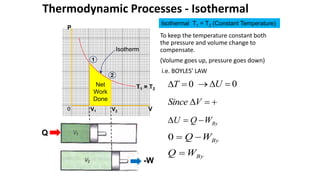
- 1. Thermodynamic Processes - Isothermal To keep the temperature constant both the pressure and volume change to compensate. (Volume goes up, pressure goes down) i.e. BOYLES’ LAW P V 0 2 1 Isotherm Isothermal T1 = T2 (Constant Temperature) V1 V2 T1 = T2 Q -W 0 T 0 U V Since By W Q U By W Q 0 By W Q Net Work Done
- 2. Thermodynamic Processes - Isobaric Heat is added to the gas which increases the Internal Energy (U). Work is done by the gas as it changes in volume. The path of an isobaric process is a horizontal line called an isobar. 0 2 1 V1 V2 Isobar T1 T2 T2>T1 Q -W Isobaric P1 = P2 (Constant Pressure) By W Q U P V Net Work Done
- 3. Thermodynamic Processes – Isovolumetric (Isochoric) 0 2 1 V1= V2 Isomet T1 T2 T2>T1 P V Isovolumetric V1 = V2 (Constant Volume) Q W = 0 0 V Since By W Q U 0 Q U Q U
- 4. Thermodynamic Processes - Adiabatic ADIABATIC- GREEK (adiabatos- "impassable") In other words, NO HEAT can leave or enter the system, it is fully insulated. 0 V1 V2 P V Adiabatic = Nothing is Constant T1 T2 T1>T2 2 1 Adiabat Q = 0 -W 0 Q Since By W Q U By W U 0 By W U
- 5. In Summary 0 P V (b) (a) (c) (d) Label Process Important Points Gas Law a Isothermal Constant T, ΔU = Q , Q = WBy Boyles Law b Isovolumetric (isochoric) Constant V, W = 0, ΔU = Q Charles Law c Isobaric Constant P, ΔU = Q - WBy Pressure Law d Adiabatic Nothing is Constant, Q = 0, ΔU = -WBy Combined Gas Law Isobaric Isothermal Isochoric Adiabatic