1. Significant figures indicate the precision or uncertainty of a measurement. Exact numbers are obtained by counting or definition while measured numbers use measuring devices and have uncertainty.
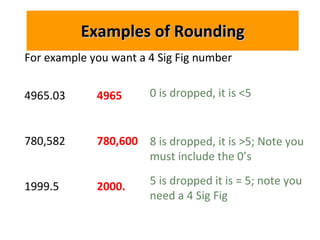
2. There are rules for determining which digits in a measured number are significant based on their position relative to the decimal point. Numbers should be rounded appropriately based on the number of significant figures.
3. Calculations with measured numbers must follow rules to avoid reporting more significant figures than the original measurements support, such as limiting digits in addition/subtraction and figures in multiplication/division.