S.T. Moe publications on opioid receptor ligands
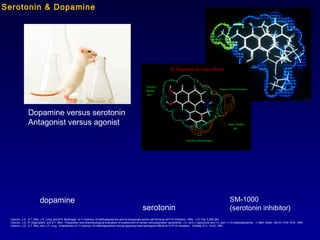
- 1. Cannon, J.G., S.T. Moe, J.P. Long, and R.K. Bhatnagar. S-11-hydroxy-10-methylaporphine and its biologically active salt forms as 5HT1A inhibitors; 1993. U.S. Pat. 5,258,384. Cannon, J.G., R. Raghupathi, and S.T. Moe. Preparation and pharmacological evaluation of enantiomers of certain nonoxygenated aporphines: (+)- and (-)-aporphine and (+)- and (-)-10-methylaporphine. J. Med. Chem. 36(10) 1316-1318, 1993. Cannon, J.G., S.T. Moe, and J.P. Long. Enantiomers of 11-hydroxy-10-methylaporphine having opposing pharmacological effects at 5-HT1A receptors. Chirality 3(1), 19-23, 1991. Dopamine versus serotonin Antagonist versus agonist dopamine serotonin SM-1000 (serotonin inhibitor) Serotonin & Dopamine
- 2. Portoghese, P.S., S. Ohkawa, and S.T. Moe. Preparation of spiroindane opiate analogs; 1994. U. S. Pat. 5,298,622 Kshirsagar, T.A., S.T. Moe, and P.S. Portoghese. Stereospecific synthesis of pseudocodeine: [2,3]-Sigmatropic rearrangement using selenium intermediates. J. Org. Chem. 63(5), 1704-1705, 1998. Portoghese, P.S., S. Ohkawa, S.T. Moe, and A.E. Takemori. Synthesis and delta-opioid receptor antagonist activity of naltrindole analogues with a regioisomeric indole moiety. J. Med. Chem. 37(12), 1886-1888, 1994. Portoghese, P.S., M. Sultana, S.T. Moe, and A.E. Takemori. Synthesis of naltrexone-derived δ-opioid antagonists. Role of conformation of the δ address moiety. J. Med. Chem. 37(5), 579-585, 1994. Portoghese, P.S., S.T. Moe, and A.E. Takemori. A selective delta-1 opioid receptor agonist derived from oxymorphone. Evidence for separate recognition sites for delta-1 opioid receptor agonists and antagonists. J. Med. Chem. 36(17), 2572-2574, 1993. Kong, H., K. Raynor, K. Yasuda, S.T. Moe, P.S. Portoghese, G.I. Bell, and T. Reisine. A single residue, aspartic acid 95, in the δ-opioid receptor specifies selective high-affinity agonist binding. J. Biol. Chem. 268(31), 23055-23058, 1993. morphine enkephalin Opioid Antagonists
- 3. SensiparSensipar®® (Cinacalcet)(Cinacalcet) • Sensipar (Cinacalcet)Sensipar (Cinacalcet) • NPS1506 (Delucemine)NPS1506 (Delucemine) • Ronacaleret (Glaxo)Ronacaleret (Glaxo) • NPS1776 (Abbott)NPS1776 (Abbott) 50 uM lead The Merck Index, 14th Edition (online) Monograph number: 02282 Antihyperparathyroid drug Hyperparathyroidism
- 4. Factors Influencing Pharmaceutical Product Design Natural Product Drug Models (Phytomedicinals) Absorption, Distribution, Metabolism, Excretion Safety, Toxicology Neurotransmitter-Like Compounds Pharmaceutical Perspective, (“Drug-like” Scaffolds) Soft Drug Approach Weak Drug Approach Commercially Successful Drug Products, (Templates) Pharmaceutical Relevance (Marketability) ?Pharmacology Synthetic Accessibility Drug Design
- 5. • Lidocaine synthesisLidocaine synthesis • USP Purity/UV AnalysisUSP Purity/UV Analysis • OctaethylporphoryrinOctaethylporphoryrin • 7-step synthesis7-step synthesis • Natural product purificationNatural product purification • Botonical identificationBotonical identification • IbuprophenIbuprophen • Qualitative identificationQualitative identification Local Anesthetics and Pain
- 6. Step 1: Finding a ring system NMDA antagonism (IC50) in rat cerebellar granular cells (RCGCs) NH2 O (2600 nM) NH2 S (676 nM) NH2 (69 nM) NH2 O (1780 nM) NH2 (1500 nM) N NH2 (609 nM) NMDA Inhibitors
- 7. Drug Discovery CycleDrug Discovery Cycle DATA MINING Drug Discovery Cycle ANALOG DESIGN
- 8. Design of Conformationally Restricted Analogs NH2 F F NH2 F F NH2 F F NH2 F F NH2 F F X-Ray Crystal Analysis from: S.T. Moe, J. Clardy, et al., Bioorg. Med. Chem. Lett. 1999, 9, 1915-1920. Structural Rigidification
- 9. 1506 reduces infarct volume by approximately 50% in animal models of stroke NPS 1506 - Novel NMDA Antagonist Control NPS 1506 Ischemic Stroke
- 10. 2000 Parke-Davis/Pfizer2000 Parke-Davis/Pfizer • Medicinal ChemistryMedicinal Chemistry • Physical-ChemicalPhysical-Chemical PropertiesProperties • ADMEADME • LeadershipLeadership • TeamworkTeamwork Drug-Like Property Analysis
- 11. 300x Antibacterial Potency300x Antibacterial Potency • D-Ala-D-Ala LigaseD-Ala-D-Ala Ligase • Cell wall synthesisCell wall synthesis • Structure-based DesignStructure-based Design • Parallel SynthesisParallel Synthesis • 5-Approach Strategy5-Approach Strategy 30 uM 0.1 uM D-Ala Ligase Inhibitors
- 12. Complementary Approaches to Analog Design Active-site Residue Targeting (Crystallography). Identify residues within reach of ligand and design analogs with complementary functional groups within range of residue Virtual-Library Docking (Modeling). Dock virtual library molecules into active-site models and score. SAR-Driven Substituent Selection (Biochemistry). Use biochemical data as a guide to select “active” functional groups. Traditional, Systematic Analoging (Synthetic Chemistry). Directed at specific questions, e.g., polarity, electronic effects, logP Diversity Analysis (Computational Chemistry). Select a diverse set of functional groups to explore chemical space, Drug Design Strategies
- 13. Parallel Synthesis • Analogs selected using the 5-approaches • 80 analogs per week • Solution phase • 5-chemists in-house • 6-step synthesis • R3-Variation on step 1 !!! D-Ala Ligase Crystal Structures
- 15. Michaelis Complex Model FXIa • Michaelis complex (green) moved from the acylated structure (grey) but the primary interactions remain intact • Phenylurea in the prime sites (left) • Guanidino in the S1 pocket (center) • Acid still H-binding to the waters • Lactam C=O close to Oγ-Ser195 and pointing toward the “oxyanion hole” Factor XIa Crystal Structure
- 16. Silvaggi N.R., et al., Chem. Biol. 14(5):533-42, 2007. 600 analogs via Solid phase synthesis Moe, S.T., et al., Bioorg. Med. Chem. (manuscript submitted Jan. 2009). Botulism Antitoxins
Editor's Notes
- Outline of strategy was use when presented with the dilemma of what to synthesize. Strategy is to use 5 complementary approaches, simultaneously. Read slide. Note: we are currently evaluating whether divesity analysis or similarity analysis would be more suitable.