Wolff kishner reduction.pdf
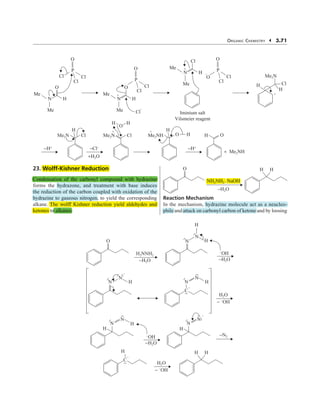
- 1. ORGANIC CHEMISTRY 3.71 Cl Cl Cl P O H N Me Me O Cl Cl Cl P O H + + + − N Me Me Cl H −H+ −H+ −Cl− O Cl H H Cl Me2N Me2N +H2O Cl Iminium salt Vilsmeier reagent P O O H + + − N Me Me Cl O H H Me2NH Cl H H Me2N O + Me2NH H O 23. Wolff-Kishner Reduction Condensation of the carbonyl compound with hydrazine forms the hydrazone, and treatment with base induces the reduction of the carbon coupled with oxidation of the hydrazine to gaseous nitrogen, to yield the corresponding alkane. The wolff Kishner reduction yield aldehydes and ketones to alkanes. O NH2NH2 .NaOH −H2O H H Reaction Mechanism In the mechanism, hydrazine molecule act as a neucleo- phile and attack on carbonyl carbon of ketone and by loosing O H2NNH2 −H2O − OH −H2O H N N H − − H N N − − − − OH −H2O H H2O − − OH H2O − − OH H N N − − − − − C H − − C H −N2 N N − − H − N N − − − H H Wolff-Kishner Reduction The wolff Kishner reduction yield aldehydes and ketones o alkanes. Condensation of the carbonyl compound with hydrazine NH2NH2 .NaOH hydrazone, and treatment with base induces the reduction of the carbon coupled with oxidation of the hydrazine to gaseous nitrogen, hydrazine molecule act as a neucleo- phile a d attack on carbonyl carbon of ketone a
- 2. 3.72 CHAPTER 2 Reaction Mechanism: Nucleophilic Addition then Elimination In the mechanism the first step is an addition of the phos- phorous ylide to the carbonyl icarbon of Aldehyde/ketone lead to the zwitterionic intermediate betaine. Than the extra electron on oxygen atom attack on electrodeficient phosphorous atom to form a four-membered cyclic intermediate, an oxaphosphetane. The four membered ring clave (due to ring strain) to form stable alkene and stable tri phenyl phosphene. of water molecule convert in to imine function group. In sub- sequent steps by loosing of two mole water and nitrogen mol- ecule via carbanion intermediate it will generate alkane. 24. Wittig Reaction It is a reaction of an aldehyde or ketone with a triphenyl phosphonium ylide (often called a Wittig reagent) to give an alkene and triphenylphosphine oxide (Ph3 PO). The reaction also known as Wittig Olefination. Wittig reactions are most commonly used to couple aldehydes and ketones to singly substituted phosphine ylides. With simple ylides this results in almost exclusively the Z-alkene product. Ylides: It may be defined as the group 15 and 16 of periodic table having positive charge directly attached to carbon con- taining negative charge (extra unshared pair of electron) due to pY-dY bonding is known as ylides. + + P+ C− O R2 R1 R3 C C R2 R1 R4 R3 C Aldehyde/ Ketone Phosphorous Ylide Alkene Triphenyl Phosphene O Ph3P Ph3 R4 R1 R1 R1 R1 H H H H H H Betain Like Intermediate H O 1 2 7 6 5 3 4 O R2 R2 R2 R2 PH3P PH3P PH3P + Ph3PO + − − O − + + R1 H H R2 PH3P O 25. Wurtz Reaction In this reaction, two alkyl halide molecules are coupled in presence of sodium metal (Na) in anhydrous ether or Tetrahydrofuran to form a new carbon carbon bond and thus by giving a symmetrical alkane. The Wurtz reaction must be performed under anhydrous conditions because the alkyl free radical formed. R–X + 2Na + R–X Dry Ether R–R + 2NaX Reaction Mechanism R X + 2Na R− Na+ + NaX R− Na+ + R R + NaX R X f water molecule convert in to imine function group. In sub- sequent steps by loosing of two mole water and nitrogen mol- ecule via carbanion intermediate it will generate alkane.