4Fe+3O2= 2Fe2O3n(Fe2O3) =16.7160=0.104375moleare u sure that.pdf
•
0 likes•2 views
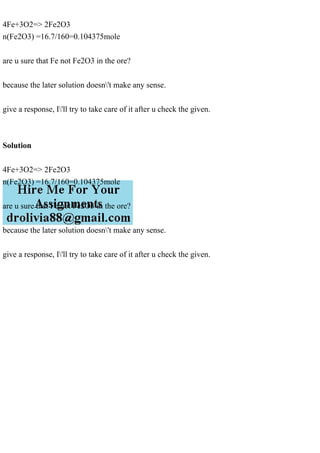
4Fe+3O2=> 2Fe2O3 n(Fe2O3) =16.7/160=0.104375mole are u sure that Fe not Fe2O3 in the ore? because the later solution doesn\'t make any sense. give a response, I\'ll try to take care of it after u check the given. Solution 4Fe+3O2=> 2Fe2O3 n(Fe2O3) =16.7/160=0.104375mole are u sure that Fe not Fe2O3 in the ore? because the later solution doesn\'t make any sense. give a response, I\'ll try to take care of it after u check the given..
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
More Related Content
More from sooryasalini
More from sooryasalini (20)
WHITE COLLAR CRIMEWhite-collar crime is nonviolent crime commited.pdf

WHITE COLLAR CRIMEWhite-collar crime is nonviolent crime commited.pdf
Web 3.0 is just how, why and at what time.Web serviceis a software.pdf

Web 3.0 is just how, why and at what time.Web serviceis a software.pdf
There are a few different reagent could be used to perform this tran.pdf

There are a few different reagent could be used to perform this tran.pdf
now a days security is very important to organization and physical s.pdf

now a days security is very important to organization and physical s.pdf
5.Write the VHDL code for the state machine.library ieee;use i.pdf

5.Write the VHDL code for the state machine.library ieee;use i.pdf
pH = pKa + log([A-][HA])(base) HONH2 + H2O (acid) HONH3+ + OH-.pdf![pH = pKa + log([A-][HA])(base) HONH2 + H2O (acid) HONH3+ + OH-.pdf](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![pH = pKa + log([A-][HA])(base) HONH2 + H2O (acid) HONH3+ + OH-.pdf](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
pH = pKa + log([A-][HA])(base) HONH2 + H2O (acid) HONH3+ + OH-.pdf
Solution Relation between carpels and locules Carpels is the p.pdf

Solution Relation between carpels and locules Carpels is the p.pdf
Solution.Providing legal advice to the president of the company co.pdf

Solution.Providing legal advice to the president of the company co.pdf
S and Se are 6th group elementsSize increase the leaving nature in.pdf

S and Se are 6th group elementsSize increase the leaving nature in.pdf
Please refer this solution. This is working file for IntegersHeade.pdf

Please refer this solution. This is working file for IntegersHeade.pdf
Simple DefinationsIntertidal zone is the area that is above wate.pdf

Simple DefinationsIntertidal zone is the area that is above wate.pdf
Production.javapublic class Production { Declaring instance.pdf

Production.javapublic class Production { Declaring instance.pdf
Proteobacteria are major phylum of gram negative bacteria. Major gro.pdf

Proteobacteria are major phylum of gram negative bacteria. Major gro.pdf
Na and K are highly reactive metals and form ionic compounds.Also hy.pdf

Na and K are highly reactive metals and form ionic compounds.Also hy.pdf
Network Communication Flow of information among the group of memb.pdf

Network Communication Flow of information among the group of memb.pdf
Muscle contractions occur through the sliding of actin and myosin fi.pdf

Muscle contractions occur through the sliding of actin and myosin fi.pdf
Lion being a carnivore animal has a big brain as compared to other a.pdf

Lion being a carnivore animal has a big brain as compared to other a.pdf
It rained this morning so the construction crew stayed indoors while.pdf

It rained this morning so the construction crew stayed indoors while.pdf
It has been observed that diseases are evolutionarily related .The h.pdf

It has been observed that diseases are evolutionarily related .The h.pdf
Java program to remove duplicatesimport java.util.;class Bag.pdf

Java program to remove duplicatesimport java.util.;class Bag.pdf
Recently uploaded
https://app.box.com/s/xplac2t6bphx6pe1mofyfj0fvrjx1f3i24 ĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH SỞ GIÁO DỤC HẢI DƯ...

24 ĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH SỞ GIÁO DỤC HẢI DƯ...Nguyen Thanh Tu Collection
Recently uploaded (20)
Personalisation of Education by AI and Big Data - Lourdes Guàrdia

Personalisation of Education by AI and Big Data - Lourdes Guàrdia
When Quality Assurance Meets Innovation in Higher Education - Report launch w...

When Quality Assurance Meets Innovation in Higher Education - Report launch w...
Observing-Correct-Grammar-in-Making-Definitions.pptx

Observing-Correct-Grammar-in-Making-Definitions.pptx
Transparency, Recognition and the role of eSealing - Ildiko Mazar and Koen No...

Transparency, Recognition and the role of eSealing - Ildiko Mazar and Koen No...
Andreas Schleicher presents at the launch of What does child empowerment mean...

Andreas Schleicher presents at the launch of What does child empowerment mean...
Basic Civil Engineering notes on Transportation Engineering & Modes of Transport

Basic Civil Engineering notes on Transportation Engineering & Modes of Transport
Spring gala 2024 photo slideshow - Celebrating School-Community Partnerships

Spring gala 2024 photo slideshow - Celebrating School-Community Partnerships
Trauma-Informed Leadership - Five Practical Principles

Trauma-Informed Leadership - Five Practical Principles
Sternal Fractures & Dislocations - EMGuidewire Radiology Reading Room

Sternal Fractures & Dislocations - EMGuidewire Radiology Reading Room
Including Mental Health Support in Project Delivery, 14 May.pdf

Including Mental Health Support in Project Delivery, 14 May.pdf
24 ĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH SỞ GIÁO DỤC HẢI DƯ...

24 ĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH SỞ GIÁO DỤC HẢI DƯ...
4Fe+3O2= 2Fe2O3n(Fe2O3) =16.7160=0.104375moleare u sure that.pdf
- 1. 4Fe+3O2=> 2Fe2O3 n(Fe2O3) =16.7/160=0.104375mole are u sure that Fe not Fe2O3 in the ore? because the later solution doesn't make any sense. give a response, I'll try to take care of it after u check the given. Solution 4Fe+3O2=> 2Fe2O3 n(Fe2O3) =16.7/160=0.104375mole are u sure that Fe not Fe2O3 in the ore? because the later solution doesn't make any sense. give a response, I'll try to take care of it after u check the given.