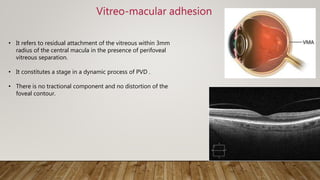
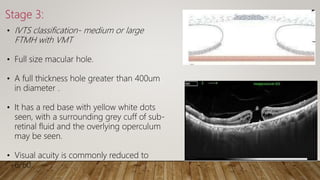
The document provides a detailed analysis of vitreo-macular interface disorders, covering functional anatomy, posterior vitreous detachment, epiretinal membranes, vitreomacular adhesion, traction, and macular holes. It explores the pathophysiology, symptoms, and treatment options for various conditions, emphasizing their etiology and classification. A comprehensive bibliography is included for further reference on the subject.



![Functional Anatomy of Vitreous
• Vitreous is a jelly like substance of 4cc, weighing 4g present in the
posterior segment of the eye behind the lens, embryologically derived
from the mesenchymal cells.
• Vitreous consists of –
-Water (99%)
- Collagen fibrils (Type 2 Collagen)
- Glycosaminoglycans [Hyaluronic acid > Chondroitin sulphate]
• Parts of the vitreous:
1)Hyaloid membrane – Anterior and Posterior Hyaloid
membrane
2) Cortical vitreous- It is present along the periphery and
contains Hyalocytes, that secrete Hyaluronic acid and is at highest
concentration at near the vitreous base and the posterior pole.
3) Medullary vitreous- It forms the majority of the vitreous body
it is similar to cortical vitreous but contains less fibrillar structure and is
essentially cell-free.](https://image.slidesharecdn.com/vitreo-macularinterfacedisorder-200819175532/85/Vitreo-macular-interface-disorder-4-320.jpg)