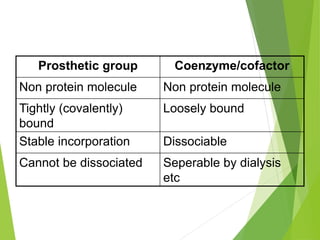
Enzymes are biomolecules that catalyze chemical reactions and increase their rates. They have high catalytic power, increasing reaction rates by factors of 108 to 1020. Enzymes also exhibit specificity, only catalyzing certain reactions and acting on specific substrates. Regulation of enzyme activity ensures metabolic reactions proceed at appropriate rates. Enzymes are classified based on the type of reaction they catalyze and given four-digit codes describing their functions. They have complex three-dimensional structures essential for catalytic activity and often require cofactors to function properly.