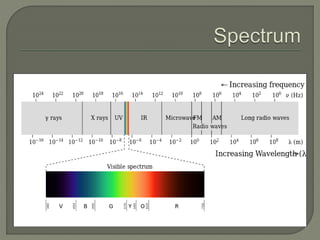
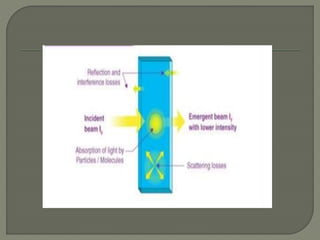
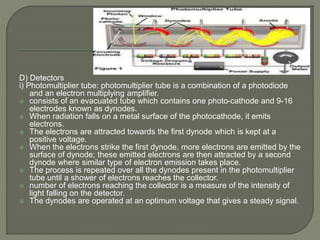
The document provides an extensive overview of UV-Vis spectroscopy, discussing its principles, instrumentation, and applications in analyzing the interaction of electromagnetic radiation with matter. It details electronic transitions, the Beer-Lambert Law, and various factors such as chromophores, auxochromes, and solvent effects that influence absorbance and spectral shifts. Additionally, it describes the operation of different types of lamps and detectors used in spectrometers.