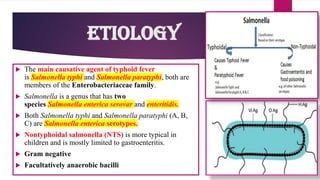
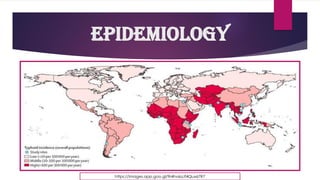
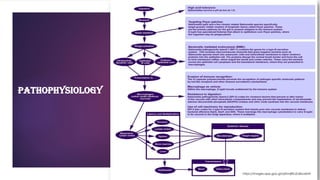
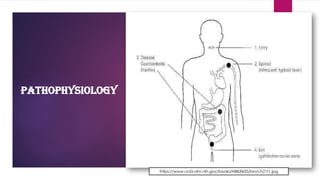
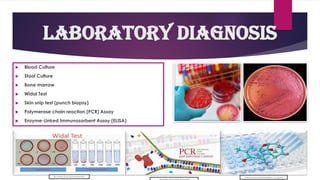
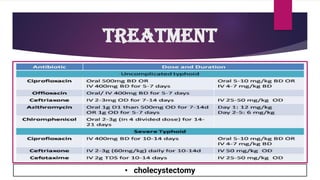
The document provides an overview of typhoid fever, primarily caused by Salmonella typhi, including its history, etiology, transmission, pathophysiology, clinical manifestations, and existing treatments and vaccinations. Typhoid fever remains a significant global public health issue, especially in developing nations, with millions of cases and deaths reported annually. Despite medical advancements, the incidence continues to rise, necessitating effective preventative measures and treatment strategies.