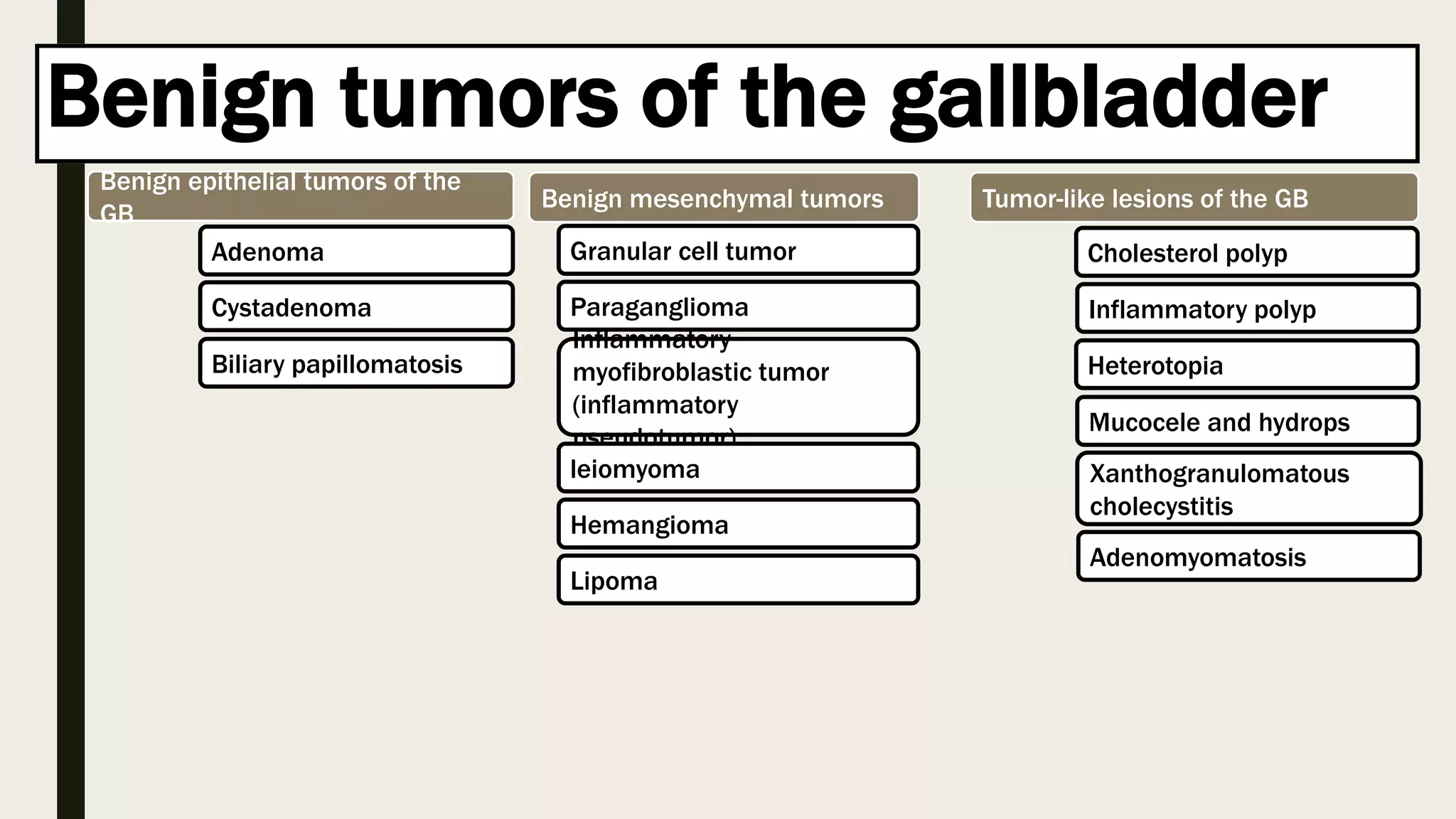
Benign and malignant tumors of the gallbladder are described. Benign tumors include cholesterol polyps, adenomyomatosis, and adenomas. Risk factors for gallbladder cancer are discussed, including gallstones, porcelain gallbladder, gallbladder polyps larger than 1 cm, and anomalous union of the pancreaticobiliary ductal system. Gallbladder cancer is often diagnosed at an advanced stage and has a poor prognosis despite surgical resection being the only potentially curative treatment.



![Gallbladder adenomas (GBA)
■ GBA are found in <0.2- 0.5% of cholecystectomy specimens.[1]
■ They are more common in women (70%) than men, and occur in adults (range 17-
79 years, mean 58 years). Rare cases have also been reported in children.[2]
■ GBA have also been reported in a/with PeutzeJegher’s [3] and Gardener’s
syndromes.[4]
■ Unless they are located in the gallbladder neck or are large enough to obstruct bile
flow, they remain clinically asymptomatic and are discovered incidentally after
cholecystectomy.
1. Albores-Saavedra J, Henson DE. Tumors of the gallbladder and extrahepatic bile ducts. Washington, D.C.: Armed Forces Institute of Pathology under the auspices of Universities Associated for
Research and Education in Pathology, 1986. p. 208.
2. Stringer MD, Ceylan H, Ward K, Wyatt JI. Gallbladder polyps in children e classification and management. J Pediatr Surg 2003; 38: 1680e4.
3. Wada K, Tanaka M, Yamaguchi K. Carcinoma and polyps of the gallbladder associated with PeutzeJeghers syndrome. Dig Dis Sci 1987; 32: 943e6.
4. Tantachamrun T, Borvonsombat S, Theetranont C. Gardner’s syndrome associated with adenomatous polyp of gall bladder: report of a case. J Med Assoc Thai 1979; 62: 441e7.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-5-2048.jpg)
![Gallbladder adenomas (GBA)
■ Adenomas may occur in any region of the GB, but MC occur in the body, followed by
the fundus and neck of the GB.
■ They are normally well-delineated lesions and may be pedunculated or sessile.[1]
■ They are typically solitary and <2 cm in size but they can be larger. Sometimes
(10%) the lesions are multiple, and rarely can be numerous enough to fill the entire
GB.[2]
Pyloric gland type adenomas
Intestinal-type adenomas
Biliary-type adenomas
1. Sato H, Mizushima M, Ito J, Doi K. Sessile adenoma of the gallbladder. Reappraisal of its importance as a precancerous lesion. Arch Pathol Lab Med 1985; 109: 65e9.
2. Albores-Saavedra J, Henson DE. Tumors of the gallbladder, extrahepatic bile ducts, and ampulla of vater. Washington, DC: Armed Forces Institute of Pathology under the auspices of Universities
Associated for Research and Education in Pathology, 2000. 365 p.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-6-2048.jpg)




![Gallbladder Carcinoma
■ Gallbladder cancer is the MC malignant tumour of the biliary tract worldwide [1]. It is
also the most aggressive cancer of the biliary tract with the shortest median survival
from the time of diagnosis [2].
■ GBC is usually considered not amenable to medical or radiation therapy, and
surgical resection is the only potentially curative treatment. Unfortunately, only a
minority of pts (10 %) are surgical candidates at the time of DX.
■ The prognosis of GBC is dismal, with 5-year survival rates of 0% to 10% and a
median survival of <6 months.
1. C. H. E. Lai and W. Y. Lau, “Gallbladder cancer—a comprehensive review,” Surgeon, vol. 6, no. 2, pp. 101–110, 2008.
2. A. X. Zhu, T. S. Hong, A. F. Hezel, and D. A. Kooby, “Current management of gallbladder carcinoma,” The Oncologist, vol. 15, no. 2, pp. 168–181, 2010.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-11-2048.jpg)
![Epidemiology
■ The distribution of GBC is geographically heterogeneous, with the highest incidence
rates (up to 21.5 per 100,000 population) observed in India.
■ Incidence rates are also high in South America, Asia, and certain Eastern European
countries such as Poland.[1]
■ GBC is rare in Western European countries and the US, where the National Cancer
Institute reported an incidence rate of 1.2 per 100,000 in 2007.
■ The average age at DX is 65 years, and globally, there is a female predisposition to
GBC.[1]
1. Rani G, Franceschi S, La Vecchia C. Gallbladder cancer worldwide: Geographical distribution and risk factors. Int J Cancer 2006; 118:1591-602.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-12-2048.jpg)
![RFs for GBC: gall stone
■ The cause of GBC is not well understood but is thought to be multifactorial.
■ Several RFs for GBC have been described. The primary RF for GBC is cholelithiasis.
■ Gallstones are found in 65% to 90% of pts with GBC.
■ Autopsy-based studies from Chile have suggested a 7-X ↑ risk of GBC in pts with
cholelithiasis, whereas epidemiologic studies in the US have observed only a
marginally significant 3-X↑ risk of GBC in men with cholelithiasis. [1,2]
1. Maringhini A, Moreau J, Melton LJ, et al. Gallstones,gallbladder cancer, and other gastrointestinal malignancies. An epidemiologic study in Rochester, Minnesota.
Ann Intern Med 1987; 107:30-5.
2. Nervi F, Duarte I, Gomez G, et al. Frequency of gallbladder cancer in Chile, a high-risk area. Int J Cancer 1988; 41:657-60.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-13-2048.jpg)
![RFs for GBC: gall stone
■ GBC actually develops in only 1% to 3% of pts with cholelithiasis. Therefore, a
prophylactic cholecystectomy in an asymptomatic pt with gallstones to prevent GBC
cannot be recommended.
■ A positive correlation between the risk of GBC and the size and number of gallstones
has been reported but likely reflects the duration of cholelithiasis.[1]
■ No differences in the risk of GBC have been observed with types of gallstones.
(SLEISENGER)/Cholesterol stones are the most common type a/with GBC.(YAMADA)
1. Miyazaki M, Takada T, Miyakawa S, et al. Risk factors for biliary tract and ampullary carcinomas and prophylactic surgery for these factors. J Hepatobiliary Pancreat
Surg 2008; 15:15-24.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-14-2048.jpg)
![RFs for GBC: Porcelain GB
■ Porcelain GB (extensive calcification of the GB wall) is a classic, although
controversial, RF for GBC.[1]
■ Although an ↑ risk of GBC has been reported in pts with a porcelain GB, the risk may
be limited to pts with selective mucosal calcification (types II and III porcelain GB)
rather than those with diffuse mucosal calcification (type I).[2]
1. Miyazaki M, Takada T, Miyakawa S, et al. Risk factors for biliary tract and ampullary carcinomas and prophylactic surgery for these factors. J Hepatobiliary
Pancreat Surg 2008; 15:15-24.
2. Stephen A, Berger D. Carcinoma in the porcelain gallbladder: A relationship revisited. Surgery 2001; 129:699-703.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-15-2048.jpg)
![GB POLYP
■ Adenomatous polyps of the GB constitute another RF for GBC.
■ The risk correlates positively with the size, type, and growth rate of the polyps.
■ Patients with polyps >1 cm in size, sessile, and a/with gallstones, exhibit a rapid
increase in size, demonstrate arterial flow on Doppler US, or are symptomatic are at
↑ risk of malignant transformation and warrant prophylactic cholecystectomy.[1,2]
1. Miyazaki M, Takada T, Miyakawa S, et al. Risk factors for biliary tract and ampullary carcinomas and prophylactic surgery for these factors. J Hepatobiliary
Pancreat Surg 2008; 15:15-24.
2. Myers R, Shaffer E, Beck P. Gallbladder polyps: Epidemiology, natural history and management. Can J Gastroenterol 2002; 16:187-94.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-16-2048.jpg)
![RFs for GBC: Anomalous union of the pancreaticobiliary
ductal system (AUPBD)
■ AUPBD has been a/with the development of GBC.
■ In this congenital defect, the pancreatic and bile ducts unite outside the duodenal wall in a long
common channel. The anomaly is found incidentally in 1.5% to 2% of pts who undergo ERCP and
leads to cholestasis and reflux of pancreatic secretions into the GB, with resulting chronic
inflammation of the mucosa.
1. Miyazaki M, Takada T, Miyakawa S, et al. Risk factors for biliary tract and ampullary carcinomas and prophylactic surgery for these factors. J Hepatobiliary Pancreat
Surg 2008; 15:15-24.
• Approx 10% of pts with GBC have coexisting AUPBD, and
GBC develops in 15% to 40% of those with AUPBD.
• On the basis of the significantly ↑ risk of GBC, several
Japanese hepatobiliary oncology associations have
recommended prophylactic cholecystectomy in pts with
AUPBD.[1]
Pic: Kimura et al. Association of Gallbladder Carcinoma and Anomalous Pancreaticobiliary Ductal
Union. GASTROENTEROLOGY 1985:89:1258-65](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-17-2048.jpg)
![RFs for GBC: PSC
■ PSC has been a/with GBC, and studies have reported that adenoca of the GB
develops in up to 20% of pts with PSC. [1,2]
■ Therefore, pts with PSC and a GB mass of any size should undergo cholecystectomy
or be monitored closely for GBC.
1. Buckles D, Lindor K, Larusso N, et al. In primary sclerosing cholangitis, gallbladder polyps are frequently malignant. Am J Gastroenterol 2002; 97:1138-42.
2. Lewis J, Talwalkar J, Rosen C, et al. Prevalence and risk factors for gallbladder neoplasia in patients with primary sclerosing cholangitis: Evidence for a
metaplasiadysplasia-carcinoma sequence. Am J Surg Pathol 2007; 31:907-13.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-18-2048.jpg)
![RFs for GBC: Adenomyomatosis of GB
■ A large Japanese study showed an ↑ incidence of GBC in pts 60 years of age or older
with segmental adenomyomatosis of the GB.[1]
■ In general, however, adenomyomatosis is viewed as a benign condition.
1. Nabatame N, Shirai Y, Nishimura A, et al. High risk of gallbladder carcinoma in elderly patients with segmental adenomyomatosis of the gallbladder. J Exp Clin
Cancer Res 2004; 23:593-8.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-19-2048.jpg)
![RFs for GBC: Others
■ Other conditions a/ with GBC include IBD, intrahepatic biliary dysplasia, and CCA. [1]
■ Chronic carriers of Salmonella Typhi or Paratyphi have been shown to be at ↑ risk for
the development of GBC.[2]
■ Other bacteria such as Escherichia coli and Hp have also been a/w GBC, but the
data are not conclusive.
■ First-degree relatives of pts with GBC have a relative risk of 13.9 for developing this
malignancy.[3]
1. Lewis J, Talwalkar J, Rosen C, et al. Prevalence and risk factors for gallbladder neoplasia in patients with primary sclerosing cholangitis: Evidence for a
metaplasiadysplasia-carcinoma sequence. Am J Surg Pathol 2007; 31:907-13.
2. Shukla V, Singh H, Pandey M, et al. Carcinoma of the gallbladder—Is it a sequel of typhoid? Dig Dis Sci 2000; 45:900-3.
3. Fernandez E, La Vecchia C, D’Avanzo B, et al. Family history and the risk of liver, gallbladder, and pancreatic cancer. Cancer Epidemiol Biomarkers Prev 1994;
3:209-12.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-20-2048.jpg)
![RFs for GBC: Others
■ Carcinogens, including methylcholanthrene, O-aminoazotoluene, and nitrosamines,
have been identified in animal models of GBC.
■ Other potential carcinogens include mustard oil, products of free radical oxidation,
and secondary bile acids.[1]
■ Obesity has been suggested to be a RF for GBC, especially in women,[2] but the
independence of obesity from cholelithiasis as a RF has not been shown.
1. Misra S, Chaturvedi A, Misra N, et al. Carcinoma of the gallbladder. Lancet Oncol 2003; 4:167-76.
2. Larsson SC, Wolk A. Obesity and the risk of gallbladder cancer: A meta-analysis. Br J Cancer 2007; 96:1457-61.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-21-2048.jpg)

![Pathology
■ Sixty percent of GBC are located in the GB fundus, 30% in the body, and 10% in the
gallbladder neck.[1]
■ GBC spreads via direct invasion, lymphatic or hematogenous metastasis, perineural
invasion, and intraperitoneal or intraductal invasion.
■ LN metastases are described in 54% to 64% of pts and correlate with the depth of
invasion.
■ GBC has a predisposition to involve the liver bed because of venous drainage,
predominantly into hepatic segments IVb and V, and the anatomic approximation
that allows direct hepatic invasion.
1. Reid K, Ramos-De la Medina A, Donohue J. Diagnosis and surgical management of gallbladder cancer: A review. J Gastrointest Surg 2007; 11:671-81.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-23-2048.jpg)
![Pathogenesis
■ GBC can develop from foci of mucosal dysplasia or carcinoma in situ that progress
to adenocarcinoma.[1]
■ The time of progression of dysplasia to carcinoma is estimated to be 10 to 15
years.[2]
1. Roa I, de Aretxabala X, Araya J, et al. Preneoplastic lesions in gallbladder cancer. J Surg Oncol 2006; 93:615-23.
2. Roa I, Araya J, Villaseca M, et al. Preneoplastic lesions and gallbladder cancer: An estimate of the period required for progression. Gastroenterology 1996;
111:232-6.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-24-2048.jpg)
![Pathogenesis
■ The major pathogenic factor is inflammation.
■ Increased iNOS and COX-2 expression has been
a/with TP53 tumor suppressor gene mutations in
pts with GBC.
■ Mouse double minute 2 homolog (MDM2)
overexpression, which can result in functional
inactivation of p53, has been described in up to
80% of GBC.[1]
■ In up to 60% of pts with GBC, mutations of the K-
ras oncogene have been detected; the frequency
is highest in patients with AUPBD.[2]
■ Studies have reported up-regulation of EGFR,
HER2/ErbB2.[3,4]
References are at the end of the slides
Note: Mdm2 is an important negative regulator of the p53 tumor suppressor.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-25-2048.jpg)
![Clinical presentation & natural history
■ Patients with GBC are often asymptomatic, and the DX may be made at the time of
imaging or cholecystectomy for other indications.
■ When symptoms are present, they are nonspecific.
■ As a result, most pts with GBC present with symptoms at an advanced stage of
disease.
■ In 47% to 78% of pts, GBC is found incidentally during cholecystectomy for
presumed benign disease, reflecting the initial clinically silent nature of this
malignancy.[1,2]
■ Incidentally diagnosed GBC generally are lower in stage than symptomatic
carcinomas at the time of DX and are a/with better median survival rates.[2]
1. Duffy A, Capanu M, Abou-Alfa GK, et al. Gallbladder cancer (GBC): 10-year experience at Memorial Sloan-Kettering Cancer Centre (MSKCC). J Surg Oncol 2008;
98:485-9.
2. Mazer LM, Losada HF, Chaudhry RM, et al. Tumor characteristics and survival analysis of incidental versus suspected gallbladder carcinoma. J Gastrointest Surg
2012; 16:1311-7.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-26-2048.jpg)
![Clinical presentation & natural history
■ At presentation, gallbladder cancer is often similar to biliary colic or chronic
cholecystitis.
■ Right upper quadrant or epigastric pain is the most common symptom (54–83%),
followed by jaundice (10–46%), nausea and vomiting (15–43%), anorexia (4–41%),
and weight loss (10–39%) [1].
■ Jaundice may result either from direct invasion of the biliary tree or from metastatic
disease to the hepatoduodenal ligament [2]. Jaundice is a poor prognostic sign and
85% of pts with jaundice have unresectable tumors.
■ Only 3–8% of patients have a palpable mass [1].
■ Among pts who present symptomatically, tumours are typically advanced with 75%
being nonresectable [2].
1. C. H. E. Lai and W. Y. Lau, “Gallbladder cancer—a comprehensive review,” Surgeon, vol. 6, no. 2, pp. 101–110, 2008.
2. G. Miller and W. R. Jarnagin, “Gallbladder carcinoma,” European Journal of Surgical Oncology, vol. 34, no. 3, pp. 306–312, 2008.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-27-2048.jpg)

![Tumor markers
■ CEA and CA 19-9 are the most commonly used tumor markers for GBC.
■ At a cutoff at 4.0 ng/mL, an elevated serum CEA level has a SN and SP of 50% and
93%.
■ The SN and SP of an elevated serum CA 19-9 level at a cutoff of 20 U/mL are 79%
and 79%.[1,2]
■ These tests aid in DX but should not be relied on because levels can be elevated in
inflammatory conditions and gastroenterologic and gynecologic malignancies.
■ CEA and CA 19-9 baseline tests are useful for monitoring response to therapy. [3]
■ Other tumour markers, such as CA 242[4] and thymidine kinase[5], have been
proposed as promising or better than CEA and CA 19‐9, but are non‐specific and
require further validation.
References are at the end of the slides](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-29-2048.jpg)
![Imaging: USG
■ The primary imaging modality for the assessment of
presumed GB disease is an USG.
■ Imaging of GBC has three broad patterns. [1,2]
1. Gore RM, Shelhamer RP. Biliary tract neoplasms: Diagnosis and staging. Cancer Imaging 2007;7:S15-23.
2. Konstantinidis IT, Bajpai S, Kambadakone AR, Tanabe KK, Berger DL, Zheng H, et al. Gallbladder lesions identified on ultrasound. Lessons from the last 10 years. J
Gastrointest Surg 2012;16:549-53.
US showing a large mass (arrow) originating from the GB
wall and protruding into the lumen.
1. commonly, a hypoechoic (relative to liver) mass
obscuring the GB lumen which shows increased
flow on color doppler with or without infiltration
into the adjacent liver,
2. an intraluminal polypoidal mass >1 cm in size,
having a broad base and fixed to the wall (a sludge
ball with similar sonographic findings will move).
3. focal wall thickening and irregularity.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-30-2048.jpg)
![Imaging: USG
■ Although it has an accuracy of >80% in diagnosing GBC, it is not an useful tool for
staging the disease.[1,2]
■ However, endoscopic USG (EUS) can visualize in detail the layers of GB wall and has
a diagnostic accuracy of 100%, 75.6%, 85.3%, and 92.7% for pTis, pT1, pT2, and
pT3-4 growths, respectively.[3]
■ It can also enable FNA of any suspicious masses or aspiration of bile for cytology.
1. Gore RM, Shelhamer RP. Biliary tract neoplasms: Diagnosis and staging. Cancer Imaging 2007;7:S15-23.
2. Konstantinidis IT, Bajpai S, Kambadakone AR, Tanabe KK, Berger DL, Zheng H, et al. Gallbladder lesions identified on ultrasound. Lessons from the last 10 years. J
Gastrointest Surg 2012;16:549-53.
3. Gore RM, Thakrar KH, Newmark GM, Mehta UK, Berlin JW. Gallbladder imaging. In: Cynthia WK, editor. Gastroenterology Clinics of North America: Gallbladder
Disease. Philadelphia: Saunders; 2010. p. 265-87.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-31-2048.jpg)
![Imaging: CT
■ CECT typically shows asymmetric wall thickening with marked enhancement during
the arterial phase, which becomes isodense to liver during portal venous phase.
1. Kim SJ, Lee JM, Lee JY, et al. Accuracy of preoperative T-staging of gallbladder carcinoma using MDCT. AJR Am J Roentgenol 2008; 190:74-80.
2. Yoshimitsu K, Honda H, Shinozaki K, et al. Helical CT of the local spread of carcinoma of the gallbladder: Evaluation according to the TNM system in patients who
underwent surgical resection. AJR Am J Roentgenol 2002; 179:423-8.
CT is inferior to USG for detecting
thickening and irregularity of GB wall and
also cholelithiasis.
CT has an 83% to 86% accuracy in
assessing the local extent is therefore,
helpful in preoperative planning.[1,2]](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-32-2048.jpg)
![Imaging: MRI, MRA, and MRCP
■ The combination of MRI (magnetic resonance imaging) with MRA (magnetic
resonance angiography) and MRCP (magnetic resonance
cholangiopancreatography) is useful in detecting vascular invasion (100% SN and
87% SP), biliary tract involvement (100% SN and 89% SP), liver invasion (67% SN
and 89% SP), and lymph node involvement (56% SN and 89% SP) [1].
■ MRI has been shown to be superior to CT scan for differentiating T1a lesions from
T1b or greater and as such may be useful in preoperative management planning [2].
1. G. Miller and W. R. Jarnagin, “Gallbladder carcinoma,” European Journal of Surgical Oncology, vol. 34, no. 3, pp. 306–312, 2008.
2. S. J. Kim, J. M. Lee, E. S. Lee, J. K. Han, and B. I. Choi, “Preoperative staging of gallbladder carcinoma using biliary MR imaging,” Journal of Magnetic Resonance
Imaging, vol. 41, no. 2, pp. 314–321, 2015.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-33-2048.jpg)
![Imaging: PET
■ The role of fluorodeoxyglucose-positron emission tomography (FDG-PET) is evolving
but not routine . In pts with incidental finding of carcinoma following
cholecystectomy, PET-CT is useful in detecting local residual disease in GB fossa and
nodal and distant metastases.[4] It is also useful in postoperative follow-up to detect
any recurrence.[1]
■ The SN of PET for detecting GBC is only 75% to 78%.[2,3]
1. Kumar R, Sharma P, Kumari A, Halanaik D, Malhotra A. Role of 18F-FDG PET/CT in detecting recurrent gallbladder carcinoma. Clin Nucl Med 2012;37:431-5.
2. Anderson C, Rice M, Pinson C, et al. Fluorodeoxyglucose PET imaging in the evaluation of gallbladder carcinoma and cholangiocarcinoma. J Gastrointest Surg
2004; 8:90-7.
3. Koh T, Taniguchi H, Yamaguchi A, et al. Differential diagnosis of gallbladder cancer using positron emission tomography with fluorine-18-labeled fluoro-
deoxyglucose (FDG-PET). J Surg Oncol 2003; 84:74-81.
4. Petrowsky H, Wildbrett P, Husarik D, et al. Impact of integrated positron emission tomography and computed tomography on staging and management of
gallbladder cancer and cholangiocarcinoma. J Hepatol 2006; 45:43-50.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-34-2048.jpg)



![AJCC 8TH ed: TNM staging of GBC
In a systematic review of over
2000 incidental GBC[1], the most
frequent stage at presentation
was pT2, followed by pT3 and pT1.
1. Choi KS, Choi SB, Park P, Kim WB, Choi SY. Clinical characteristics of incidental or unsuspected gallbladder cancers diagnosed during or after cholecystectomy: a
systematic review and meta-analysis. World J Gastroenterol 2015; 21: 1315–1323.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-38-2048.jpg)
![Management
■ The management of GBC is primarily surgical. The nature of surgery depends on
the stage of the disease at DX.
■ The earlier the disease is DX and its resectability established, the better is the
prognosis.
■ Nearly 70% of the cases in the US are DX incidentally, either intraoperatively or on
the postoperative pathology; the remaining 30% are diagnosed preoperatively on
cross-sectional abdominal imaging done for nonspecific symptoms such as RUQ
abdominal pain mimicking cholecystitis, nausea, vomiting, weight loss, or
jaundice.[1-4]
1. Gore RM, Shelhamer RP. Biliary tract neoplasms: Diagnosis and staging. Cancer Imaging 2007;7:S15-23.
2. Goetze TO, Paolucci V. Adequate extent in radical re-resection of incidental gallbladder carcinoma: Analysis of the German registry. Surg Endosc 2010;24:2156-64.
3. Furlan A, Ferris JV, Hosseinzadeh K, Borhani AA. Gallbladder carcinoma update: Multimodality imaging evaluation, staging, and treatment options. AJR Am J
Roentgenol 2008;191:1440-7.
4. Miller G, Schwartz LH, D’Angelica M. The use of imaging in the diagnosis and staging of hepatobiliary malignancies. Surg Oncol Clin N Am 2007;16:343-68.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-39-2048.jpg)



![Surgery:
■ The only treatment of potentially curative intent in GBC is an aggressive R0
resection.[1]
■ Aggressive surgery entails radical cholecystectomy, which includes adequate
lymphadenectomy and adequate liver resection along with en bloc resection of any
involved viscera.
■ Adequate lymphadenectomy means clearance of the LNs and fibro fatty tissue in
the hepatoduodenal ligament to the extent that bile duct and vessels therein are
skeletonized; the hepatic artery is bared of nodes till its origin from celiac axis.
■ Nodes anterior and posterior to the head of pancreas are also cleared.[2]
1. Dixon E, Vollmer CM Jr., Sahajpal A, Cattral M, Grant D, Doig C, et al. An aggressive surgical approach leads to improved survival in patients with gallbladder cancer:
A 12-year study at a North American center. Ann Surg 2005;241:385-94.
2. Kapoor VK. Advanced gallbladder cancer: Indian “middle path”. J Hepatobiliary Pancreat Surg 2007;14:366-73.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-43-2048.jpg)
![Surgery:
■ Adequate liver resection, for no liver involvement (T1b and T2 tumors), entails either
an en bloc resection of the GB along with a 2-cm nonanatomical wedge of liver
(when this is done along with adequate lymphadenectomy, it is termed as extended
cholecystectomy) or a formal anatomical bisegmental liver resection (segments
IVb/V).For up to T1 lesions where the tumor has not penetrated the muscularis mucosa and margins are negative,
cholecystectomy is sufficient and can be curative.
• Only 15% to 47% of pts are candidates for surgical resection at the time of DX
because the stage of the disease is advanced in most cases.
• CIs to resection include multiple hepatic or distant metastases, gross vascular
invasion or encasement of major vessels, malignant ascites, and poor functional
status.[1]
1. Reid K, Ramos-De la Medina A, Donohue J. Diagnosis and surgical management of gallbladder cancer: A review. J Gastrointest Surg 2007; 11:671-81.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-44-2048.jpg)

![Gallbladder Cancer and Jaundice
■ The presence of jaundice in gallbladder cancer usually portends a poor prognosis.
These pts need careful surgical evaluation.
■ Although a relative contraindication, in select pts curative intent resection can be
attempted for resectable disease in centers with available expertise.
Randomized studies did not show a benefit of preoperative biliary decompression for jaundiced pts [1-3].
However, these studies were performed in an era in which surgical intervention mostly involved palliative bypass
of the biliary tree, and the potential benefits of preoperative decompression prior to either
pancreaticoduodenectomy or liver resection are not defined.
1. HatfieldAR, TobiasR, TerblancheJ, et al.Preoperative external biliary drainage in obstructive jaundice. A prospective controlled clinical trial. Lancet1982;2:896.
2. McPhersonGA, BenjaminIS, HodgsonHJ, et al.Pre‐operative percutaneous transhepatic biliary drainage: the results of a controlled trial. Br J Surg1984;71:371.
3. PittHA, GomesAS, LoisJF, et al.Does preoperative percutaneous biliary drainage reduce operative risk or increase hospital cost?Ann Surg1985;201:545.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-46-2048.jpg)

![Adjuvant therapy
■ Postoperative adjuvant gemcitabine and platinum-based CT and external beam
radiotherapy should be offered to pts with poorly differentiated, aggressive T2, or
higher, node/margin-positive lesions.[1]
■ Intensity-modulated radiation therapy is the newer technical advancement which
allows highly conformal radiation delivery, sparing as far as possible the normal
organs.[2]
■ Some believes that radical re-resection, if not otherwise contraindicated by the pt’s
status, may still be beneficial in terms of long-term survival even for T4 disease.[3]
1. Wang SJ, Lemieux A, Kalpathy-Cramer J, Ord CB, Walker GV, Fuller CD, et al. Nomogram for predicting the benefit of adjuvant chemoradiotherapy for resected
gallbladder cancer. J Clin Oncol 2011;29:4627-32.
2. Zhu AX, Hong TS, Hezel AF, Kooby DA. Current management of gallbladder carcinoma. Oncologist 2010;15:168-81.
3. Fong Y, Jarnagin W, Blumgart LH. Gallbladder cancer: Comparison of patients presenting initially for definitive operation with those presenting after prior
noncurative intervention. Ann Surg 2000;232:557-69.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-48-2048.jpg)
![Neoadjuvant therapy
■ Neoadjuvant therapy often is not an option due to the advanced disease at
diagnosis and is not considered a standard of care in resectable cases. Referral for
early clinical trials should be considered[1].
1. Ben-Josef E, Guthrie KA, El-Khoueiry AB, Corless CL, Zalupski MM, Lowy AM, et al. SWOG S0809: A Phase II Intergroup Trial of Adjuvant Capecitabine and
Gemcitabine Followed by Radiotherapy and Concurrent Capecitabine in Extrahepatic Cholangiocarcinoma and Gallbladder Carcinoma. J. Clin. Oncol. 2015 Aug
20;33(24):2617-22.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-49-2048.jpg)
![Approach to pts with unresectable/metastatic
disease
The National Comprehensive Cancer Network makes the following
recommendations:
■ Enrollment for clinical trials
■ Supportive care: Percutaneous or endoscopic endobiliary or gastrointestinal stents
for relieving jaundice or duodenal obstruction, narcotics for pain relief
■ Palliation for pts with jaundice before instituting chemotherapy.
■ EUS/CT-guided fine-needle aspiration biopsy for establishment of pathological DX is
required before institution of palliative nonsurgical therapy.[1]
1. Eckel F, Brunner T, Jelic S; ESMO Guidelines Working Group. Biliary cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol
2011;22 Suppl 6:vi40-4.
2. Valle J, Wasan H, Palmer DH, et al. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N Engl J Med 2010; 362:1273-81.
The standard of care for pts with unresectable GBC is chemotherapy with gemcitabine combined with cisplatin.
This recommendation is based largely on the ABC-02 trial, which included 149 pts with GBC and showed an
improvement in OS of 3.6 months. [2]](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-50-2048.jpg)
![Novel Therapies
■ GBC has been a/with the mutations of KRAS, INK4a, p53 genes, and amplification
of human epidermal growth factor receptor (EGFR), (HER)-2/Neu.[1]
■ Targeted therapies may prove to be beneficial.
■ A Phase II study with a single agent erlotinib on pts with biliary tract cancers has
shown promising results.[2]
■ Another randomized Phase II trial on pts of advanced GBC suggested the efficacy of
cetuximab followed by gemcitabine plus oxaliplatin.[3]
■ Similarly, vascular endothelial growth factor expression has been correlated with
advanced and metastatic GBCA. Bevacizumab and sorafenib were tested, with
promising results, in multicentric Phase II trials in unresectable and metastatic
cases of GBCA.[4,5]
References are at the end of the slides](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-51-2048.jpg)
![Outcome prediction and prognostic score
■ A number of factors are a/with outcome in incidental GBC. Among the most
important is the ability to achieve an R0 resection, whereas both a higher T category
and the presence of LN metastasis are strong predictors of poor survival.[1-4]
■ Several attempts at refining prognostication have been entertained, with a
Gallbladder Cancer Predictive Risk (GBPR) score developed from a multicentre
series of incidental GBC.[5].
1. Amini N, Kim Y,Wilson A, Margonis GA, Ethun CG, Poultsides G et al. Prognostic implications of lymph node status for patients with gallbladder cancer: a multiinstitutional study. Ann Surg Oncol 2016;
23: 3016–3023.
2. Saqib R, Pathak S, Smart N, Nunes Q, Rees J, Finch Jones M et al. Prognostic significance of pre-operative inflammatory markers in resected gallbladder cancer: a systematic review. ANZ J Surg 2018;
88: 554–559.
3. Amini N, Spolverato G, Kim Y, Gupta R, Margonis GA, Ejaz A et al. Lymph node status after resection for gallbladder adenocarcinoma: prognostic implications of different nodal staging/scoring
systems. J Surg Oncol 2015; 111: 299–305.
4. ZhangW, Hong HJ, Chen YL. Establishment of a gallbladder cancer-specific survival model to predict prognosis in non-metastatic gallbladder cancer patients after surgical resection. Dig Dis Sci 2018;
63: 2251–2258.
5. Ethun CG, Postlewait LM, Le N, Pawlik TM, Buettner S, Poultsides G et al. A novel pathology-based preoperative risk score to predict locoregional residual and distant disease and survival for
incidental gallbladder cancer: a 10-institution study from the U.S. Extrahepatic Biliary Malignancy Consortium. Ann Surg Oncol 2017; 24: 1343–1350.](https://image.slidesharecdn.com/cagb-190912095606/75/Tumors-of-gallbladder-52-2048.jpg)






