Thermo+rev+it.pdf
•
0 likes•9 views
The document summarizes key concepts in thermodynamics including: - State variables like pressure, volume, and temperature that describe a system's condition. - Extensive and intensive properties. - Internal energy, enthalpy, and the first law of thermodynamics. - Reversible and quasi-static processes, heat capacity, and formulas for isothermal, isobaric, isochoric, and adiabatic processes. - Entropy, the second law of thermodynamics, and entropy changes for different processes. - Gibbs free energy and conditions for spontaneity. - Types of enthalpies including formation, combustion, solution, hydration, and neutralization.
Report
Share
Report
Share
Download to read offline
Recommended
Lecture9

Thermodynamics describes the transfer of energy and changes in states of matter. The first law of thermodynamics states that energy is conserved during any process. The second law states that the entropy of the universe increases over time as energy disperses and becomes less useful. Natural systems evolve towards stable, low-energy states. Thermodynamic properties like enthalpy, heat capacity, and entropy can be used to characterize physical and chemical changes.
RAM

Thermodynamics studies energy changes that occur in systems and their surroundings. A system is the portion of the universe being studied, while the surroundings are everything outside the system. Natural systems tend toward states of minimum energy. The first law of thermodynamics states that energy is conserved during any process. The second law states that the entropy of the universe increases over time as energy moves from being concentrated to becoming dispersed.
Chemical Thermodynamics

Basic Terminology,Heat, energy and work, Internal Energy (E or U),First Law of Thermodynamics, Enthalpy,Molar heat capacity, Heat capacity,Specific heat capacity,Enthalpies of Reactions,Hess’s Law of constant heat summation,Born–Haber Cycle,Lattice energy,Second law of thermodynamics, Gibbs free energy(ΔG),Bond Energies,Efficiency of a heat engine
Thermodynamics

Thermodynamics deals with energy changes in chemical reactions and their feasibility. It describes three types of systems - open, closed, and isolated - based on their ability to exchange energy and matter with surroundings. The first law of thermodynamics states that energy in an isolated system is constant and can neither be created nor destroyed. It is expressed as a change in internal energy (ΔU) of the system equals heat (q) absorbed plus work (w) done. Entropy is a measure of randomness or disorder in a system, which typically increases for spontaneous processes that are irreversible without external influence.
Thermodynamics notes

This document discusses key concepts in thermodynamics including:
1. It defines thermodynamics as dealing with energy changes in chemical reactions and their feasibility.
2. It describes open, closed, and isolated systems and different types of processes like isothermal, isobaric, isochoric, and adiabatic processes.
3. It explains important thermodynamic properties and concepts such as internal energy, enthalpy, Gibbs free energy, entropy, and the three laws of thermodynamics.
2nd Lecture on Chemical Thermodynamics | Chemistry Part I | 12th Std

1) Internal energy (U) is the energy stored in a substance and is a state function. The first law of thermodynamics states that the total internal energy of an isolated system is constant.
2) For an isothermal process, the internal energy is constant and the work done equals the negative of heat absorbed. For an adiabatic process, no heat is exchanged.
3) Enthalpy (H) is the sum of the internal energy of a system and the product of pressure and volume. For a chemical reaction at constant pressure, the change in enthalpy equals the change in internal energy plus the product of the change in moles and the gas constant.
Ch 8 - Energy, Enthalpy, and Thermochemistry.pdf

The document discusses energy, enthalpy, and thermochemistry. It defines key concepts like energy, heat, work, internal energy, enthalpy, and explains how they relate. It also discusses state functions, the first and second laws of thermodynamics, and how to calculate enthalpy changes using standard enthalpies of formation and Hess's law. Experimental methods like calorimetry are also covered to measure energy changes during chemical reactions.
Ch6 Thermochemistry (updated)

Thermochemistry is the study of heat changes in chemical reactions. There are several types of energy including chemical, thermal, nuclear, and radiant energy. Heat is the transfer of thermal energy between objects at different temperatures. Thermochemistry examines heat absorbed or released by chemical reactions using concepts like exothermic, endothermic, enthalpy, and calorimetry. The first law of thermodynamics states that energy cannot be created or destroyed, only transferred between systems and their surroundings.
Recommended
Lecture9

Thermodynamics describes the transfer of energy and changes in states of matter. The first law of thermodynamics states that energy is conserved during any process. The second law states that the entropy of the universe increases over time as energy disperses and becomes less useful. Natural systems evolve towards stable, low-energy states. Thermodynamic properties like enthalpy, heat capacity, and entropy can be used to characterize physical and chemical changes.
RAM

Thermodynamics studies energy changes that occur in systems and their surroundings. A system is the portion of the universe being studied, while the surroundings are everything outside the system. Natural systems tend toward states of minimum energy. The first law of thermodynamics states that energy is conserved during any process. The second law states that the entropy of the universe increases over time as energy moves from being concentrated to becoming dispersed.
Chemical Thermodynamics

Basic Terminology,Heat, energy and work, Internal Energy (E or U),First Law of Thermodynamics, Enthalpy,Molar heat capacity, Heat capacity,Specific heat capacity,Enthalpies of Reactions,Hess’s Law of constant heat summation,Born–Haber Cycle,Lattice energy,Second law of thermodynamics, Gibbs free energy(ΔG),Bond Energies,Efficiency of a heat engine
Thermodynamics

Thermodynamics deals with energy changes in chemical reactions and their feasibility. It describes three types of systems - open, closed, and isolated - based on their ability to exchange energy and matter with surroundings. The first law of thermodynamics states that energy in an isolated system is constant and can neither be created nor destroyed. It is expressed as a change in internal energy (ΔU) of the system equals heat (q) absorbed plus work (w) done. Entropy is a measure of randomness or disorder in a system, which typically increases for spontaneous processes that are irreversible without external influence.
Thermodynamics notes

This document discusses key concepts in thermodynamics including:
1. It defines thermodynamics as dealing with energy changes in chemical reactions and their feasibility.
2. It describes open, closed, and isolated systems and different types of processes like isothermal, isobaric, isochoric, and adiabatic processes.
3. It explains important thermodynamic properties and concepts such as internal energy, enthalpy, Gibbs free energy, entropy, and the three laws of thermodynamics.
2nd Lecture on Chemical Thermodynamics | Chemistry Part I | 12th Std

1) Internal energy (U) is the energy stored in a substance and is a state function. The first law of thermodynamics states that the total internal energy of an isolated system is constant.
2) For an isothermal process, the internal energy is constant and the work done equals the negative of heat absorbed. For an adiabatic process, no heat is exchanged.
3) Enthalpy (H) is the sum of the internal energy of a system and the product of pressure and volume. For a chemical reaction at constant pressure, the change in enthalpy equals the change in internal energy plus the product of the change in moles and the gas constant.
Ch 8 - Energy, Enthalpy, and Thermochemistry.pdf

The document discusses energy, enthalpy, and thermochemistry. It defines key concepts like energy, heat, work, internal energy, enthalpy, and explains how they relate. It also discusses state functions, the first and second laws of thermodynamics, and how to calculate enthalpy changes using standard enthalpies of formation and Hess's law. Experimental methods like calorimetry are also covered to measure energy changes during chemical reactions.
Ch6 Thermochemistry (updated)

Thermochemistry is the study of heat changes in chemical reactions. There are several types of energy including chemical, thermal, nuclear, and radiant energy. Heat is the transfer of thermal energy between objects at different temperatures. Thermochemistry examines heat absorbed or released by chemical reactions using concepts like exothermic, endothermic, enthalpy, and calorimetry. The first law of thermodynamics states that energy cannot be created or destroyed, only transferred between systems and their surroundings.
Hsslive-XI-Cheem-Ch-6_Thermodynamics.pdf

1. Thermodynamics is the branch of science that deals with the relationship between heat and work. Chemical thermodynamics deals specifically with heat changes in chemical reactions.
2. Systems can exchange energy and matter with their surroundings and are classified as open, closed, or isolated depending on these abilities. Macroscopic properties include temperature, pressure, volume and others.
3. Enthalpy is a measure of the total heat content of a system and is a state function. The change in enthalpy of a reaction, ΔH, is the heat absorbed or released at constant pressure.
Hsslive xi-cheem-ch-6 thermodynamics (2)

1. Thermodynamics is the branch of science that deals with the relationship between heat and work, especially as applied to chemical reactions.
2. There are three types of systems - open, closed, and isolated - depending on their ability to exchange energy and matter with surroundings.
3. Important macroscopic properties include temperature, pressure, volume, internal energy, enthalpy, and entropy. Extensive properties depend on amount of matter and intensive properties do not.
Hsslive xi-cheem-ch-6 thermodynamics (1)

1. Thermodynamics is the branch of science that deals with the relationship between heat and work, especially as it relates to chemical reactions.
2. There are three main types of systems - open, closed, and isolated - depending on their ability to exchange energy and matter with surroundings. Macroscopic properties include temperature, pressure, volume, and enthalpy.
3. Key concepts in thermodynamics include state functions, path functions, the first law of thermodynamics, enthalpy, heat capacity, and Hess's law of constant heat summation. Standard enthalpies of formation and reaction are important for determining energy changes in chemical processes.
Hsslive xi-cheem-ch-6 thermodynamics

1. Thermodynamics is the branch of science that deals with the relationship between heat and work, especially as it relates to chemical reactions.
2. There are three main types of systems - open, closed, and isolated - depending on their ability to exchange energy and matter with surroundings. Macroscopic properties include temperature, pressure, volume, and enthalpy.
3. Key concepts in thermodynamics include state functions, path functions, the first law of thermodynamics, enthalpy, heat capacity, and Hess's law of constant heat summation. The standard enthalpy of reaction can be calculated from standard enthalpies of formation of reactants and products.
6.-Thermodynamics.pdf

1. Thermodynamics provides information about heat flow and properties related to large numbers of particles but does not give information about reaction rates or apply to microsystems.
2. Systems can be open, closed, or isolated depending on whether mass and heat are transferred. The universe is considered an isolated system.
3. State functions depend only on the initial and final states while path functions depend on the process path. Internal energy, enthalpy, and entropy are state functions while heat and work are path functions.
F y b. sc. ii. chemical energetics

Need of thermodynamics and the Laws of Thermodynamics.
Important principles and definitions of thermochemistry.
Concept of standard state and standard enthalpies of formations,
Integral and differential enthalpies of solution and dilution.
Calculation of bond energy, bond dissociation energy and resonance energy from thermochemical data.
Variation of enthalpy of a reaction with temperature – Kirchhoff’s equation.
Statement of Third Law of thermodynamics and calculation of absolute entropies of substances.
JFI -just for information
Latent heat and enthalpy

Latent heat is energy absorbed or released during phase changes without a change in temperature. There are two main types: latent heat of fusion during melting/freezing, and latent heat of vaporization during boiling/condensing.
Enthalpy is a measure of the total energy of a system. It is equal to the internal energy plus the product of pressure and volume. The change in enthalpy of a reaction indicates whether the reaction absorbs or releases energy. Exothermic reactions have a negative change in enthalpy as they release heat, while endothermic reactions have a positive change as they absorb heat.
THERMODYANAMICS.pptx

Thermodynamics deals with the relationship between heat and other forms of energy. The internal energy of a system is the total energy within it, including types like vibrational and translational energy. The first law of thermodynamics states that energy is conserved - it cannot be created or destroyed, only changed from one form to another. According to the first law, the change in internal energy of a system equals the heat added to the system plus the work done on the system.
chemical equilibrium and thermodynamics

1. The document discusses key concepts in chemical thermodynamics including the first law of thermodynamics. It defines important terms like system, surroundings, state functions, extensive and intensive properties.
2. The first law states that energy can be transformed but not created or destroyed. For a process, the change in internal energy of a system equals the heat transferred plus work done.
3. Enthalpy is a state function equal to the internal energy plus the product of pressure and volume. For a constant pressure process, the enthalpy change equals the heat transferred.
005 lecture f

This document discusses thermochemistry and calorimetry. It defines key concepts in thermochemistry including the various forms of energy, exothermic and endothermic processes, and systems and surroundings. It introduces the first law of thermodynamics and defines state functions. It also discusses enthalpy, thermochemical equations, and calculations involving enthalpy changes. The document explains calorimetry concepts like heat capacity, specific heat, and how to perform calculations using bomb and coffee cup calorimetry.
Chapter5.pdf

The document discusses thermochemistry and the following key points:
- Thermochemistry is the study of energy changes in chemical reactions, especially the heat evolved or absorbed.
- Enthalpy (H) is a state function that equals the internal energy (E) plus pressure-volume work (PV) for a chemical reaction. The enthalpy change (ΔH) of a reaction is the heat absorbed or released under constant pressure.
- Hess's law states that the enthalpy change for an overall reaction is equal to the sum of the enthalpy changes for the individual steps of that reaction. This allows for calculation of enthalpy changes from standard enthalpy of formation values.
CH1201-Thermodynamics.pptx

This document provides an overview of key concepts in thermodynamics, including:
- The 0th law defines temperature and thermal equilibrium.
- The 1st law concerns the conservation of energy and defines internal energy. Energy cannot be created or destroyed, only changed in form.
- The 2nd law defines entropy and the direction of spontaneous processes over time. Entropy always increases over time for isolated systems.
- An equation of state relates the state variables like pressure, volume, and temperature that define the state of a system at equilibrium.
Thermodynamics

A homogeneous thermodynamic system is one whose properties are uniform throughout. A heterogeneous system contains distinct phases.
There are several types of thermodynamic processes including isochoric (constant volume), isobaric (constant pressure), and adiabatic (no heat transfer). Extensive properties depend on amount of substance and intensive properties do not.
The first law of thermodynamics states that energy is conserved and heat and work are equivalent. For an ideal gas undergoing an adiabatic process, PVγ is constant, where γ is the heat capacity ratio.
Energy ch 16

Energy exists in different forms and can be converted between forms. Heat is a form of energy transfer between objects at different temperatures, and involves the random motion of atoms and molecules. The calorie measures the heat required to raise 1g of water by 1°C. Potential energy is due to position or composition, while kinetic energy is due to motion. Energy is conserved according to the law of conservation of energy. Specific heat and enthalpy formulas are used to quantify heat transfer and chemical reactions. Phase changes between solid, liquid and gas involve energy in the form of enthalpy of fusion and vaporization.
First Law of Thermodynamics

First law of thermodynamics as taught in introductory physical chemistry (includes general chemistry material). Covers concepts such as internal energy, heat, work, heat capacity, enthalpy, bomb calorimetry, Hess's law, thermochemical equations, bond energy, and heat of formations.
CHE 101 Thermochem.ppt

Thermochemistry is the study of heat and energy changes in chemical and physical processes. It is concerned with energy changes that accompany physical and chemical processes at constant volume or constant pressure.
The first law of thermochemistry states that the quality of heat evolved or absorbed during a chemical reaction is the same, regardless of the pathway between the initial and final states. Hess's law states that the total enthalpy change for a reaction is the sum of the enthalpy changes for the individual steps of the reaction.
Enthalpy change (ΔH) of a reaction is the heat absorbed or released when one mole of the reaction occurs at constant pressure. It can be measured using a bomb calorimeter. Common standard
Worksheet first law

The First Law of Thermodynamics states that energy cannot be created or destroyed, only transferred or changed in form. For any process, the change in internal energy of a system is equal to the heat added to the system minus the work done by the system. Heat is the transfer of energy between systems due to a temperature difference, while work is the transfer of energy by a force acting through a distance. Endothermic processes absorb heat from the surroundings, while exothermic processes release heat.
Thermodynamics

These slides are based on the notes provided by the K V Sangathan. For the revision of thermodynamics the notes are pretty awseome. I f only want submit the home work they will do so.I am sure they will help.
THANK YOU
Standard Enthalpy Changes of Reactions

1) Enthalpy is a measure of the heat absorbed or released during a chemical reaction at constant pressure. It is equal to the change in internal energy of the system plus the product of pressure and change in volume.
2) The standard enthalpy change of a reaction is the enthalpy change that occurs under standard state conditions of 1 atm pressure and 25°C temperature.
3) Standard enthalpy changes of formation, combustion, atomization, neutralization, and solution can be defined based on specific chemical processes occurring under standard state conditions.
3.biochemical thermodynamics 

Thermodynamics describes changes in heat and energy during chemical reactions and equilibria. It provides insight into reaction equilibrium, feasibility under conditions, and molecular bonding forces. The document discusses key thermodynamic concepts like open and closed systems, the laws of thermodynamics, enthalpy, entropy, and free energy - and how these can be used to determine if reactions will occur spontaneously. Living organisms are considered open systems that exchange heat and matter with their environments.
Redefining brain tumor segmentation: a cutting-edge convolutional neural netw...

Medical image analysis has witnessed significant advancements with deep learning techniques. In the domain of brain tumor segmentation, the ability to
precisely delineate tumor boundaries from magnetic resonance imaging (MRI)
scans holds profound implications for diagnosis. This study presents an ensemble convolutional neural network (CNN) with transfer learning, integrating
the state-of-the-art Deeplabv3+ architecture with the ResNet18 backbone. The
model is rigorously trained and evaluated, exhibiting remarkable performance
metrics, including an impressive global accuracy of 99.286%, a high-class accuracy of 82.191%, a mean intersection over union (IoU) of 79.900%, a weighted
IoU of 98.620%, and a Boundary F1 (BF) score of 83.303%. Notably, a detailed comparative analysis with existing methods showcases the superiority of
our proposed model. These findings underscore the model’s competence in precise brain tumor localization, underscoring its potential to revolutionize medical
image analysis and enhance healthcare outcomes. This research paves the way
for future exploration and optimization of advanced CNN models in medical
imaging, emphasizing addressing false positives and resource efficiency.
学校原版美国波士顿大学毕业证学历学位证书原版一模一样

原版一模一样【微信:741003700 】【美国波士顿大学毕业证学历学位证书】【微信:741003700 】学位证,留信认证(真实可查,永久存档)offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原海外各大学 Bachelor Diploma degree, Master Degree Diploma
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
More Related Content
Similar to Thermo+rev+it.pdf
Hsslive-XI-Cheem-Ch-6_Thermodynamics.pdf

1. Thermodynamics is the branch of science that deals with the relationship between heat and work. Chemical thermodynamics deals specifically with heat changes in chemical reactions.
2. Systems can exchange energy and matter with their surroundings and are classified as open, closed, or isolated depending on these abilities. Macroscopic properties include temperature, pressure, volume and others.
3. Enthalpy is a measure of the total heat content of a system and is a state function. The change in enthalpy of a reaction, ΔH, is the heat absorbed or released at constant pressure.
Hsslive xi-cheem-ch-6 thermodynamics (2)

1. Thermodynamics is the branch of science that deals with the relationship between heat and work, especially as applied to chemical reactions.
2. There are three types of systems - open, closed, and isolated - depending on their ability to exchange energy and matter with surroundings.
3. Important macroscopic properties include temperature, pressure, volume, internal energy, enthalpy, and entropy. Extensive properties depend on amount of matter and intensive properties do not.
Hsslive xi-cheem-ch-6 thermodynamics (1)

1. Thermodynamics is the branch of science that deals with the relationship between heat and work, especially as it relates to chemical reactions.
2. There are three main types of systems - open, closed, and isolated - depending on their ability to exchange energy and matter with surroundings. Macroscopic properties include temperature, pressure, volume, and enthalpy.
3. Key concepts in thermodynamics include state functions, path functions, the first law of thermodynamics, enthalpy, heat capacity, and Hess's law of constant heat summation. Standard enthalpies of formation and reaction are important for determining energy changes in chemical processes.
Hsslive xi-cheem-ch-6 thermodynamics

1. Thermodynamics is the branch of science that deals with the relationship between heat and work, especially as it relates to chemical reactions.
2. There are three main types of systems - open, closed, and isolated - depending on their ability to exchange energy and matter with surroundings. Macroscopic properties include temperature, pressure, volume, and enthalpy.
3. Key concepts in thermodynamics include state functions, path functions, the first law of thermodynamics, enthalpy, heat capacity, and Hess's law of constant heat summation. The standard enthalpy of reaction can be calculated from standard enthalpies of formation of reactants and products.
6.-Thermodynamics.pdf

1. Thermodynamics provides information about heat flow and properties related to large numbers of particles but does not give information about reaction rates or apply to microsystems.
2. Systems can be open, closed, or isolated depending on whether mass and heat are transferred. The universe is considered an isolated system.
3. State functions depend only on the initial and final states while path functions depend on the process path. Internal energy, enthalpy, and entropy are state functions while heat and work are path functions.
F y b. sc. ii. chemical energetics

Need of thermodynamics and the Laws of Thermodynamics.
Important principles and definitions of thermochemistry.
Concept of standard state and standard enthalpies of formations,
Integral and differential enthalpies of solution and dilution.
Calculation of bond energy, bond dissociation energy and resonance energy from thermochemical data.
Variation of enthalpy of a reaction with temperature – Kirchhoff’s equation.
Statement of Third Law of thermodynamics and calculation of absolute entropies of substances.
JFI -just for information
Latent heat and enthalpy

Latent heat is energy absorbed or released during phase changes without a change in temperature. There are two main types: latent heat of fusion during melting/freezing, and latent heat of vaporization during boiling/condensing.
Enthalpy is a measure of the total energy of a system. It is equal to the internal energy plus the product of pressure and volume. The change in enthalpy of a reaction indicates whether the reaction absorbs or releases energy. Exothermic reactions have a negative change in enthalpy as they release heat, while endothermic reactions have a positive change as they absorb heat.
THERMODYANAMICS.pptx

Thermodynamics deals with the relationship between heat and other forms of energy. The internal energy of a system is the total energy within it, including types like vibrational and translational energy. The first law of thermodynamics states that energy is conserved - it cannot be created or destroyed, only changed from one form to another. According to the first law, the change in internal energy of a system equals the heat added to the system plus the work done on the system.
chemical equilibrium and thermodynamics

1. The document discusses key concepts in chemical thermodynamics including the first law of thermodynamics. It defines important terms like system, surroundings, state functions, extensive and intensive properties.
2. The first law states that energy can be transformed but not created or destroyed. For a process, the change in internal energy of a system equals the heat transferred plus work done.
3. Enthalpy is a state function equal to the internal energy plus the product of pressure and volume. For a constant pressure process, the enthalpy change equals the heat transferred.
005 lecture f

This document discusses thermochemistry and calorimetry. It defines key concepts in thermochemistry including the various forms of energy, exothermic and endothermic processes, and systems and surroundings. It introduces the first law of thermodynamics and defines state functions. It also discusses enthalpy, thermochemical equations, and calculations involving enthalpy changes. The document explains calorimetry concepts like heat capacity, specific heat, and how to perform calculations using bomb and coffee cup calorimetry.
Chapter5.pdf

The document discusses thermochemistry and the following key points:
- Thermochemistry is the study of energy changes in chemical reactions, especially the heat evolved or absorbed.
- Enthalpy (H) is a state function that equals the internal energy (E) plus pressure-volume work (PV) for a chemical reaction. The enthalpy change (ΔH) of a reaction is the heat absorbed or released under constant pressure.
- Hess's law states that the enthalpy change for an overall reaction is equal to the sum of the enthalpy changes for the individual steps of that reaction. This allows for calculation of enthalpy changes from standard enthalpy of formation values.
CH1201-Thermodynamics.pptx

This document provides an overview of key concepts in thermodynamics, including:
- The 0th law defines temperature and thermal equilibrium.
- The 1st law concerns the conservation of energy and defines internal energy. Energy cannot be created or destroyed, only changed in form.
- The 2nd law defines entropy and the direction of spontaneous processes over time. Entropy always increases over time for isolated systems.
- An equation of state relates the state variables like pressure, volume, and temperature that define the state of a system at equilibrium.
Thermodynamics

A homogeneous thermodynamic system is one whose properties are uniform throughout. A heterogeneous system contains distinct phases.
There are several types of thermodynamic processes including isochoric (constant volume), isobaric (constant pressure), and adiabatic (no heat transfer). Extensive properties depend on amount of substance and intensive properties do not.
The first law of thermodynamics states that energy is conserved and heat and work are equivalent. For an ideal gas undergoing an adiabatic process, PVγ is constant, where γ is the heat capacity ratio.
Energy ch 16

Energy exists in different forms and can be converted between forms. Heat is a form of energy transfer between objects at different temperatures, and involves the random motion of atoms and molecules. The calorie measures the heat required to raise 1g of water by 1°C. Potential energy is due to position or composition, while kinetic energy is due to motion. Energy is conserved according to the law of conservation of energy. Specific heat and enthalpy formulas are used to quantify heat transfer and chemical reactions. Phase changes between solid, liquid and gas involve energy in the form of enthalpy of fusion and vaporization.
First Law of Thermodynamics

First law of thermodynamics as taught in introductory physical chemistry (includes general chemistry material). Covers concepts such as internal energy, heat, work, heat capacity, enthalpy, bomb calorimetry, Hess's law, thermochemical equations, bond energy, and heat of formations.
CHE 101 Thermochem.ppt

Thermochemistry is the study of heat and energy changes in chemical and physical processes. It is concerned with energy changes that accompany physical and chemical processes at constant volume or constant pressure.
The first law of thermochemistry states that the quality of heat evolved or absorbed during a chemical reaction is the same, regardless of the pathway between the initial and final states. Hess's law states that the total enthalpy change for a reaction is the sum of the enthalpy changes for the individual steps of the reaction.
Enthalpy change (ΔH) of a reaction is the heat absorbed or released when one mole of the reaction occurs at constant pressure. It can be measured using a bomb calorimeter. Common standard
Worksheet first law

The First Law of Thermodynamics states that energy cannot be created or destroyed, only transferred or changed in form. For any process, the change in internal energy of a system is equal to the heat added to the system minus the work done by the system. Heat is the transfer of energy between systems due to a temperature difference, while work is the transfer of energy by a force acting through a distance. Endothermic processes absorb heat from the surroundings, while exothermic processes release heat.
Thermodynamics

These slides are based on the notes provided by the K V Sangathan. For the revision of thermodynamics the notes are pretty awseome. I f only want submit the home work they will do so.I am sure they will help.
THANK YOU
Standard Enthalpy Changes of Reactions

1) Enthalpy is a measure of the heat absorbed or released during a chemical reaction at constant pressure. It is equal to the change in internal energy of the system plus the product of pressure and change in volume.
2) The standard enthalpy change of a reaction is the enthalpy change that occurs under standard state conditions of 1 atm pressure and 25°C temperature.
3) Standard enthalpy changes of formation, combustion, atomization, neutralization, and solution can be defined based on specific chemical processes occurring under standard state conditions.
3.biochemical thermodynamics 

Thermodynamics describes changes in heat and energy during chemical reactions and equilibria. It provides insight into reaction equilibrium, feasibility under conditions, and molecular bonding forces. The document discusses key thermodynamic concepts like open and closed systems, the laws of thermodynamics, enthalpy, entropy, and free energy - and how these can be used to determine if reactions will occur spontaneously. Living organisms are considered open systems that exchange heat and matter with their environments.
Similar to Thermo+rev+it.pdf (20)
Recently uploaded
Redefining brain tumor segmentation: a cutting-edge convolutional neural netw...

Medical image analysis has witnessed significant advancements with deep learning techniques. In the domain of brain tumor segmentation, the ability to
precisely delineate tumor boundaries from magnetic resonance imaging (MRI)
scans holds profound implications for diagnosis. This study presents an ensemble convolutional neural network (CNN) with transfer learning, integrating
the state-of-the-art Deeplabv3+ architecture with the ResNet18 backbone. The
model is rigorously trained and evaluated, exhibiting remarkable performance
metrics, including an impressive global accuracy of 99.286%, a high-class accuracy of 82.191%, a mean intersection over union (IoU) of 79.900%, a weighted
IoU of 98.620%, and a Boundary F1 (BF) score of 83.303%. Notably, a detailed comparative analysis with existing methods showcases the superiority of
our proposed model. These findings underscore the model’s competence in precise brain tumor localization, underscoring its potential to revolutionize medical
image analysis and enhance healthcare outcomes. This research paves the way
for future exploration and optimization of advanced CNN models in medical
imaging, emphasizing addressing false positives and resource efficiency.
学校原版美国波士顿大学毕业证学历学位证书原版一模一样

原版一模一样【微信:741003700 】【美国波士顿大学毕业证学历学位证书】【微信:741003700 】学位证,留信认证(真实可查,永久存档)offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原海外各大学 Bachelor Diploma degree, Master Degree Diploma
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
Advanced control scheme of doubly fed induction generator for wind turbine us...

This paper describes a speed control device for generating electrical energy on an electricity network based on the doubly fed induction generator (DFIG) used for wind power conversion systems. At first, a double-fed induction generator model was constructed. A control law is formulated to govern the flow of energy between the stator of a DFIG and the energy network using three types of controllers: proportional integral (PI), sliding mode controller (SMC) and second order sliding mode controller (SOSMC). Their different results in terms of power reference tracking, reaction to unexpected speed fluctuations, sensitivity to perturbations, and resilience against machine parameter alterations are compared. MATLAB/Simulink was used to conduct the simulations for the preceding study. Multiple simulations have shown very satisfying results, and the investigations demonstrate the efficacy and power-enhancing capabilities of the suggested control system.
The Python for beginners. This is an advance computer language.

Python language is very important language at this time. we can easily understand this language by these notes.
Modelagem de um CSTR com reação endotermica.pdf

Modelagem em função de transferencia. CSTR não-linear.
Recycled Concrete Aggregate in Construction Part II

Using recycled concrete aggregates (RCA) for pavements is crucial to achieving sustainability. Implementing RCA for new pavement can minimize carbon footprint, conserve natural resources, reduce harmful emissions, and lower life cycle costs. Compared to natural aggregate (NA), RCA pavement has fewer comprehensive studies and sustainability assessments.
Iron and Steel Technology Roadmap - Towards more sustainable steelmaking.pdf

Iron and Steel Technology towards Sustainable Steelmaking
ISPM 15 Heat Treated Wood Stamps and why your shipping must have one

For International shipping and maritime laws all wood must contain the ISPM 15 Stamp. Here is how and why.
Eric Nizeyimana's document 2006 from gicumbi to ttc nyamata handball play

this notes have been created
by Eric36 at nyamata ttc
after readings
pe book for y2sme .
Understanding Inductive Bias in Machine Learning

This presentation explores the concept of inductive bias in machine learning. It explains how algorithms come with built-in assumptions and preferences that guide the learning process. You'll learn about the different types of inductive bias and how they can impact the performance and generalizability of machine learning models.
The presentation also covers the positive and negative aspects of inductive bias, along with strategies for mitigating potential drawbacks. We'll explore examples of how bias manifests in algorithms like neural networks and decision trees.
By understanding inductive bias, you can gain valuable insights into how machine learning models work and make informed decisions when building and deploying them.
A SYSTEMATIC RISK ASSESSMENT APPROACH FOR SECURING THE SMART IRRIGATION SYSTEMS

The smart irrigation system represents an innovative approach to optimize water usage in agricultural and landscaping practices. The integration of cutting-edge technologies, including sensors, actuators, and data analysis, empowers this system to provide accurate monitoring and control of irrigation processes by leveraging real-time environmental conditions. The main objective of a smart irrigation system is to optimize water efficiency, minimize expenses, and foster the adoption of sustainable water management methods. This paper conducts a systematic risk assessment by exploring the key components/assets and their functionalities in the smart irrigation system. The crucial role of sensors in gathering data on soil moisture, weather patterns, and plant well-being is emphasized in this system. These sensors enable intelligent decision-making in irrigation scheduling and water distribution, leading to enhanced water efficiency and sustainable water management practices. Actuators enable automated control of irrigation devices, ensuring precise and targeted water delivery to plants. Additionally, the paper addresses the potential threat and vulnerabilities associated with smart irrigation systems. It discusses limitations of the system, such as power constraints and computational capabilities, and calculates the potential security risks. The paper suggests possible risk treatment methods for effective secure system operation. In conclusion, the paper emphasizes the significant benefits of implementing smart irrigation systems, including improved water conservation, increased crop yield, and reduced environmental impact. Additionally, based on the security analysis conducted, the paper recommends the implementation of countermeasures and security approaches to address vulnerabilities and ensure the integrity and reliability of the system. By incorporating these measures, smart irrigation technology can revolutionize water management practices in agriculture, promoting sustainability, resource efficiency, and safeguarding against potential security threats.
官方认证美国密歇根州立大学毕业证学位证书原版一模一样

原版一模一样【微信:741003700 】【美国密歇根州立大学毕业证学位证书】【微信:741003700 】学位证,留信认证(真实可查,永久存档)offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原海外各大学 Bachelor Diploma degree, Master Degree Diploma
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
Comparative analysis between traditional aquaponics and reconstructed aquapon...

The aquaponic system of planting is a method that does not require soil usage. It is a method that only needs water, fish, lava rocks (a substitute for soil), and plants. Aquaponic systems are sustainable and environmentally friendly. Its use not only helps to plant in small spaces but also helps reduce artificial chemical use and minimizes excess water use, as aquaponics consumes 90% less water than soil-based gardening. The study applied a descriptive and experimental design to assess and compare conventional and reconstructed aquaponic methods for reproducing tomatoes. The researchers created an observation checklist to determine the significant factors of the study. The study aims to determine the significant difference between traditional aquaponics and reconstructed aquaponics systems propagating tomatoes in terms of height, weight, girth, and number of fruits. The reconstructed aquaponics system’s higher growth yield results in a much more nourished crop than the traditional aquaponics system. It is superior in its number of fruits, height, weight, and girth measurement. Moreover, the reconstructed aquaponics system is proven to eliminate all the hindrances present in the traditional aquaponics system, which are overcrowding of fish, algae growth, pest problems, contaminated water, and dead fish.
Computational Engineering IITH Presentation

This Presentation will give you a brief idea about what Computational Engineering at IIT Hyderabad has to offer.
Unit-III-ELECTROCHEMICAL STORAGE DEVICES.ppt

Batteries -Introduction – Types of Batteries – discharging and charging of battery - characteristics of battery –battery rating- various tests on battery- – Primary battery: silver button cell- Secondary battery :Ni-Cd battery-modern battery: lithium ion battery-maintenance of batteries-choices of batteries for electric vehicle applications.
Fuel Cells: Introduction- importance and classification of fuel cells - description, principle, components, applications of fuel cells: H2-O2 fuel cell, alkaline fuel cell, molten carbonate fuel cell and direct methanol fuel cells.
2008 BUILDING CONSTRUCTION Illustrated - Ching Chapter 02 The Building.pdf

2008 BUILDING CONSTRUCTION Illustrated - Ching Chapter 02 The Building
Electric vehicle and photovoltaic advanced roles in enhancing the financial p...

Climate change's impact on the planet forced the United Nations and governments to promote green energies and electric transportation. The deployments of photovoltaic (PV) and electric vehicle (EV) systems gained stronger momentum due to their numerous advantages over fossil fuel types. The advantages go beyond sustainability to reach financial support and stability. The work in this paper introduces the hybrid system between PV and EV to support industrial and commercial plants. This paper covers the theoretical framework of the proposed hybrid system including the required equation to complete the cost analysis when PV and EV are present. In addition, the proposed design diagram which sets the priorities and requirements of the system is presented. The proposed approach allows setup to advance their power stability, especially during power outages. The presented information supports researchers and plant owners to complete the necessary analysis while promoting the deployment of clean energy. The result of a case study that represents a dairy milk farmer supports the theoretical works and highlights its advanced benefits to existing plants. The short return on investment of the proposed approach supports the paper's novelty approach for the sustainable electrical system. In addition, the proposed system allows for an isolated power setup without the need for a transmission line which enhances the safety of the electrical network
Recently uploaded (20)
Redefining brain tumor segmentation: a cutting-edge convolutional neural netw...

Redefining brain tumor segmentation: a cutting-edge convolutional neural netw...
Engineering Drawings Lecture Detail Drawings 2014.pdf

Engineering Drawings Lecture Detail Drawings 2014.pdf
Advanced control scheme of doubly fed induction generator for wind turbine us...

Advanced control scheme of doubly fed induction generator for wind turbine us...
The Python for beginners. This is an advance computer language.

The Python for beginners. This is an advance computer language.
Recycled Concrete Aggregate in Construction Part II

Recycled Concrete Aggregate in Construction Part II
Iron and Steel Technology Roadmap - Towards more sustainable steelmaking.pdf

Iron and Steel Technology Roadmap - Towards more sustainable steelmaking.pdf
ISPM 15 Heat Treated Wood Stamps and why your shipping must have one

ISPM 15 Heat Treated Wood Stamps and why your shipping must have one
Eric Nizeyimana's document 2006 from gicumbi to ttc nyamata handball play

Eric Nizeyimana's document 2006 from gicumbi to ttc nyamata handball play
A SYSTEMATIC RISK ASSESSMENT APPROACH FOR SECURING THE SMART IRRIGATION SYSTEMS

A SYSTEMATIC RISK ASSESSMENT APPROACH FOR SECURING THE SMART IRRIGATION SYSTEMS
Comparative analysis between traditional aquaponics and reconstructed aquapon...

Comparative analysis between traditional aquaponics and reconstructed aquapon...
2008 BUILDING CONSTRUCTION Illustrated - Ching Chapter 02 The Building.pdf

2008 BUILDING CONSTRUCTION Illustrated - Ching Chapter 02 The Building.pdf
Electric vehicle and photovoltaic advanced roles in enhancing the financial p...

Electric vehicle and photovoltaic advanced roles in enhancing the financial p...
Thermo+rev+it.pdf
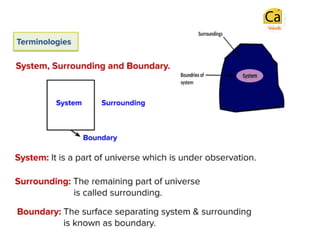
- 2. The state variables (P, V, T, n) describe the condition of a system. On changing any one or more of these variables the state of the system changes. Extensive Properties: Total Energy, volume, mass, etc. Intensive Properties: Pressure, Density, Refractive Index, etc.
- 8. Internal Energy (U) It is the sum total of the components of energy of the system due to the internal factors. U = KE + PE ΔU = q + W Enthalpy (H) It is the nnet heat content of the system. Mathematically: H = U + PV Remember: ΔH = qp ΔU = qv
- 9. First Law of Thermodynamics:
- 10. Reversible Process: A process whose direction can be changed by an infinitesimal change to the system or surroundings and which can be reversed by retracing the original path and both the system and surroundings are restored to theinitial state. Quasi Static State System is always in equilibrium with the surroundings
- 11. Heat Capacity Heat needed to raise the temperature of the system by 1K C = q/ΔT Molar Heat Capacity Heat needed to raise the temperature of one mole gas by 1K CM = q/nΔT Molar heat capacity constant pressure (CP ): CP = qP /nΔT Molar heat capacity constant volume (CV ): CV = qV /nΔT ΔH = qp = nCP ΔT ΔU = qv = nCV ΔT
- 13. Formulae: (Ideal gas, Reversible Processes) Isothermal: ΔU = ΔH =0 w = nRT ln (V1/V2) = nRT ln (P2/P1) q = nRT ln (V2/V1) = nRT ln(P1/P2) Isobaric: w = – PΔV = – nRΔT Isochoric w = 0 qv = ΔU = nCvΔT Adiabatic q = 0 w = ΔU ΔU = nCV ΔT = (P2 V2 – P1 V1 )/(γ–1) = (nRΔT)/(γ– 1) ΔH = nCP ΔT
- 14. Entropy (S): The degree of “randomness” of a system 2nd Law of Thermodynamics(2LoT): The entropy of an isolated system/Universe always tends to increase In a spontaneous process the entropy of the Universe increases Remember: In a reversible process the entropy of the Universe remains constant i.e. ΔSTotal = 0
- 15. ΔSsys = nCV ln(T2 /T1 ) + nR ln(V2 /V1 ) This expression can be simplified for the four processes: Isothermal process: ΔS = nR ln(V2 /V1 ) Isochoric process: ΔS = nCV ln(T2 /T1 ) For isobaric process: ΔS = nCP ln(T2 /T1 ) Adiabatic process: ΔS = 0 (qrev = 0)
- 16. Gibbs Free Energy: Gibbs Free energy function gives us a very convenient parameter to judge the spontaneity of a process from the system’s perspective. At constant temperature and pressure ΔG = –TΔSTOTAL For a process to be spontaneous ΔG < 0 at a constant temperature ΔGSYS = ΔH – TΔSSYS
- 17. Types of Enthalpies: Enthalpy of Formation ΔHf 0 Heat absorbed or released when one mole of a compound is formed from its constituent elements under their standard elemental forms. The enthalpy for formation of the following substances is taken to be zero under 1 bar pressure and 298 K H2 (g) + ½ O2 (g) H2 0(l) ΔHf o = -286 kJ/mol
- 18. Enthalpy of Combustion Heat released or absorbed when one mole of a substance undergoes combustion in presence of oxygen Enthalpy of Solution Heat released or absorbed when 1 mole of a compound is dissolved in excess of a solvent (water)
- 19. Enthalpy of Hydration Heat released or absorbed when 1 mole of anhydrous or partially hydrated salt undergoes hydration by the addition of water of crystallisation. Enthalpy of Neutralization Heat released or absorbed when one equivalent of an acid undergoes neutralisation with one equivalent of a base.
- 20. Bond Dissociation Enthalpy The energy needed to break the bonds of one mole molecules is called the Bond Dissociation Enthalpy of the substance H2 H + H BDE = 436kJ/mol Resonance Energy Many compounds exhibit resonance. Due to resonance they exist in a structure which is different from the expected one and more Stable. It is a negative value.
- 21. Hess’ Law Uses the fact that Enthalpy is a state function
- 22. Free Expansion: Expansion of gas in an isolated system in Vaccum Pext =0 w=0 q=0 ΔT = 0 ΔU= 0 ΔH= 0 ΔS > 0 Polytropic Process Generalized form of any thermodynamic process where PV n = constant, (n is a real number) For an isothermal process n = 1 For an adiabatic process n = γ