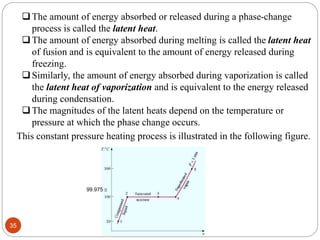
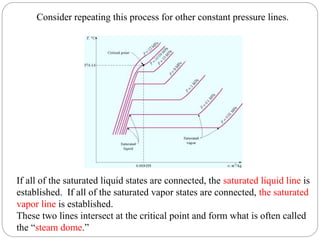
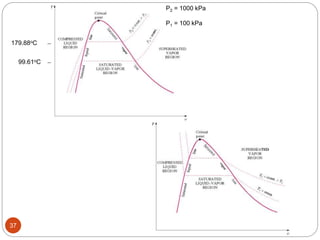
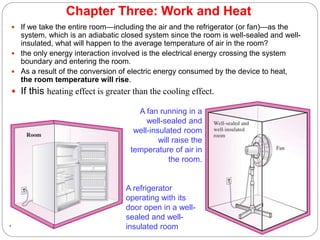
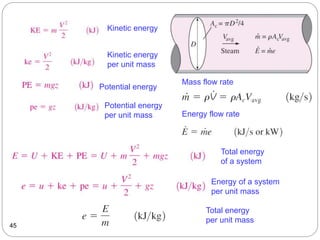
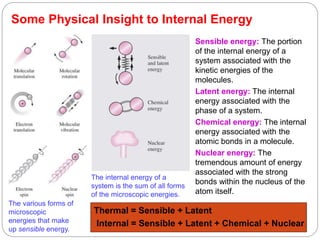
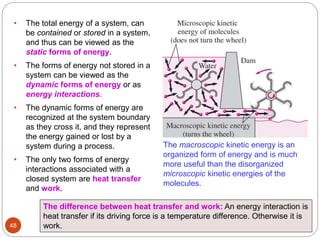
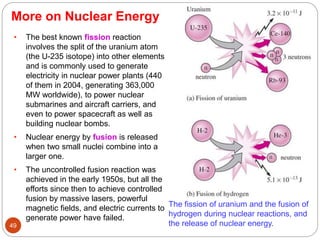
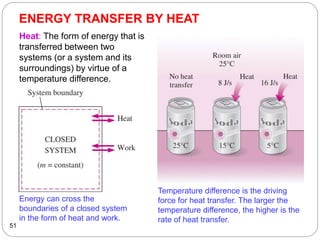
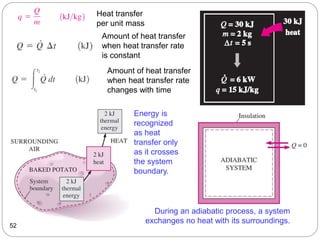
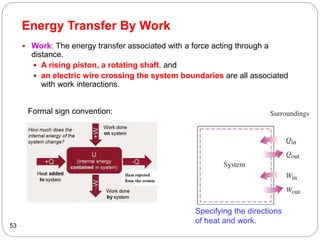
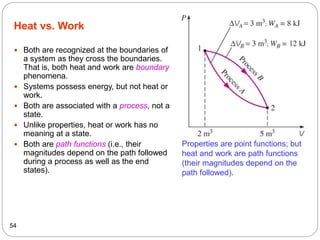
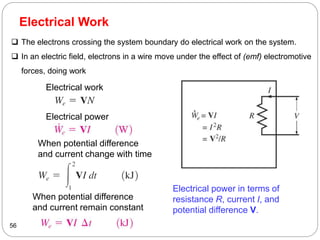
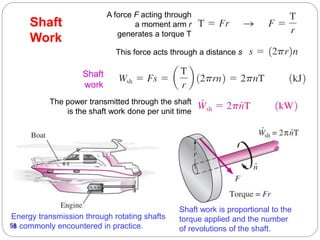
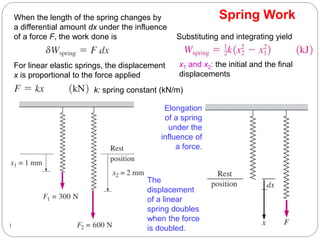
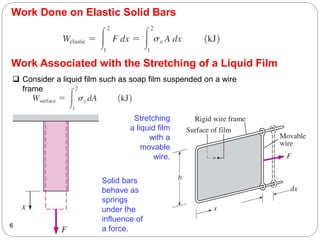
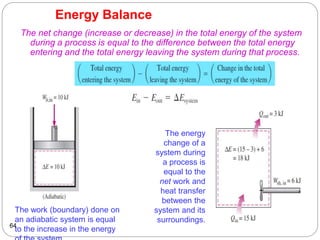
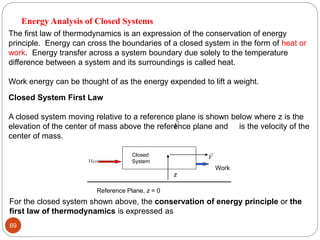
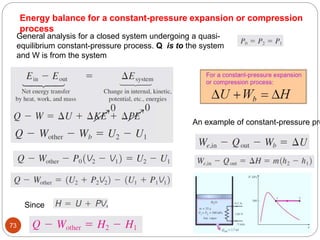
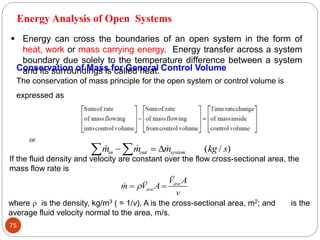
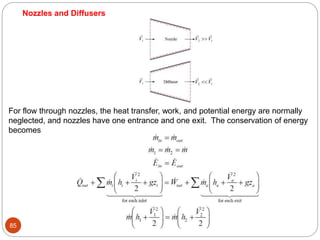
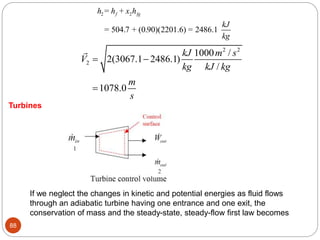
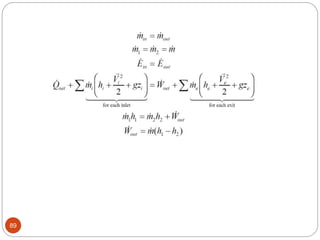
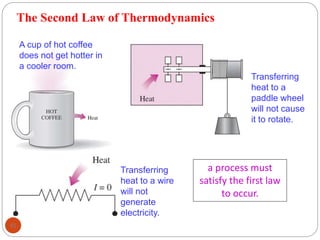
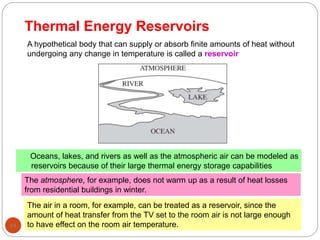
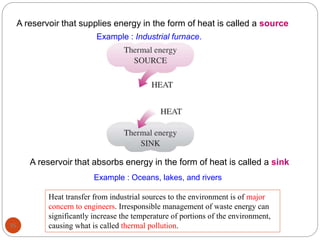
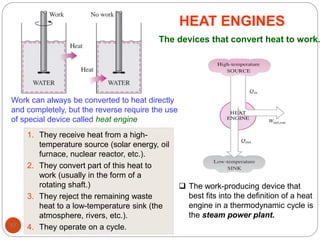
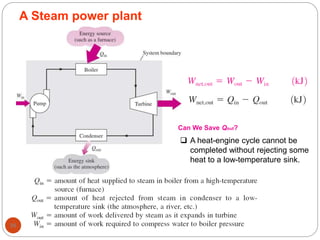
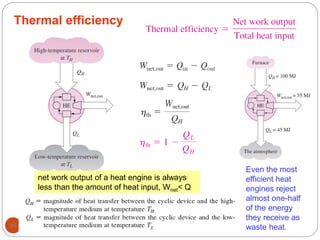
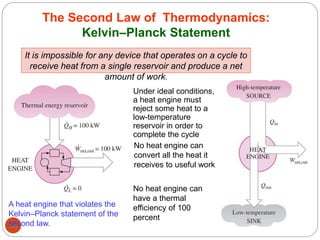
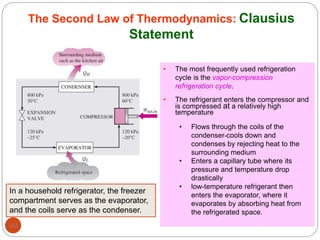
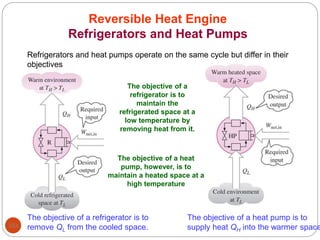
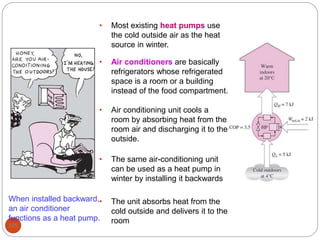
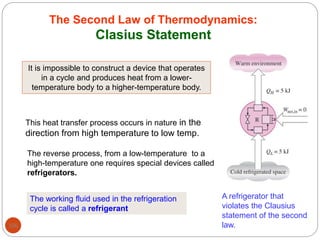
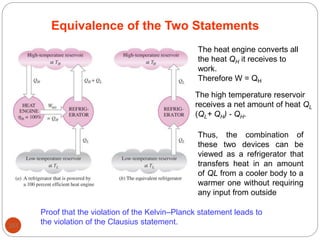
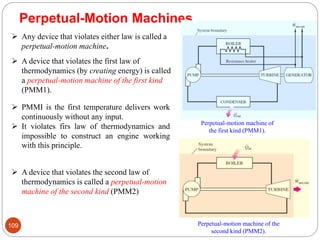
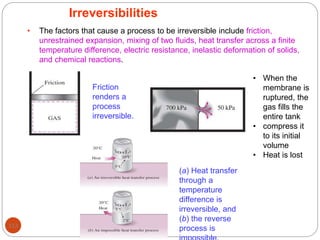
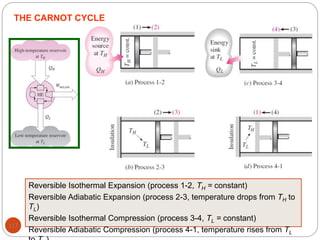
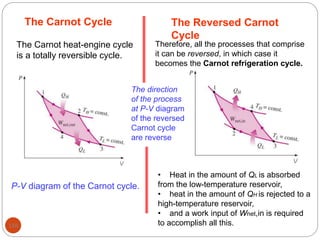
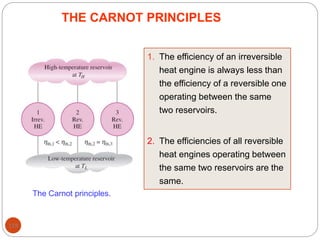
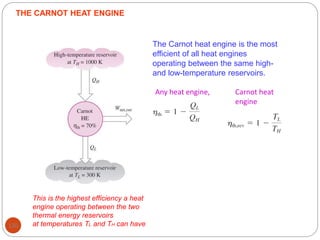
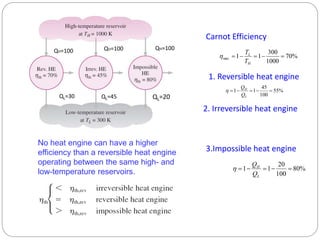
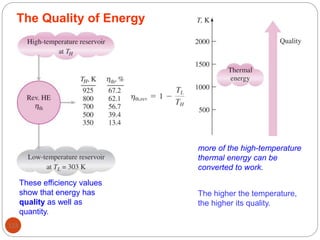
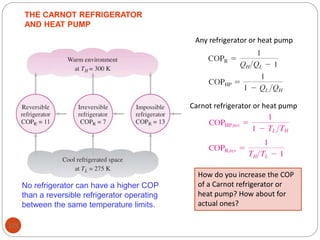
This document provides an overview of thermodynamics concepts including:
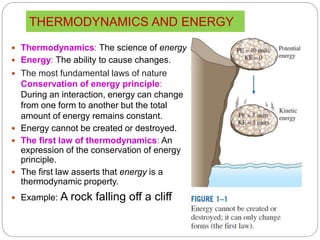
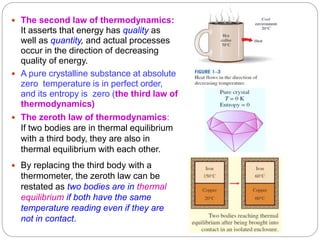
- The four laws of thermodynamics and what they assert about energy and its transformation.
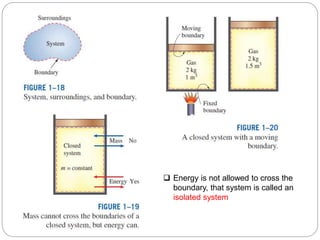
- Systems, surroundings, boundaries, open and closed systems.
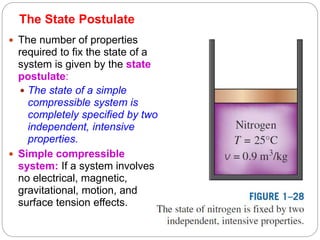
- Properties of systems including intensive, extensive, specific properties, and state properties.
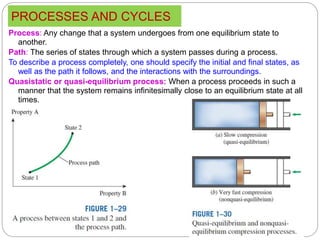
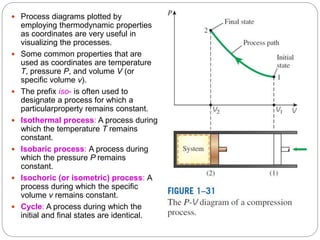
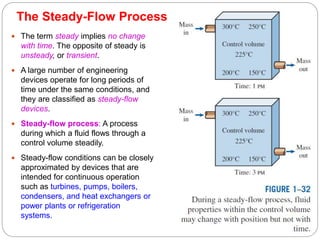
- Equilibrium states, processes, cycles, and steady-flow processes.
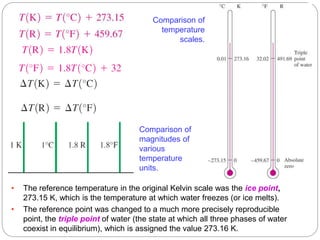
- Temperature scales including Celsius, Fahrenheit, Kelvin, and Rankine scales.
- Concepts related to pure substances including homogeneous substances, phases, and examples.