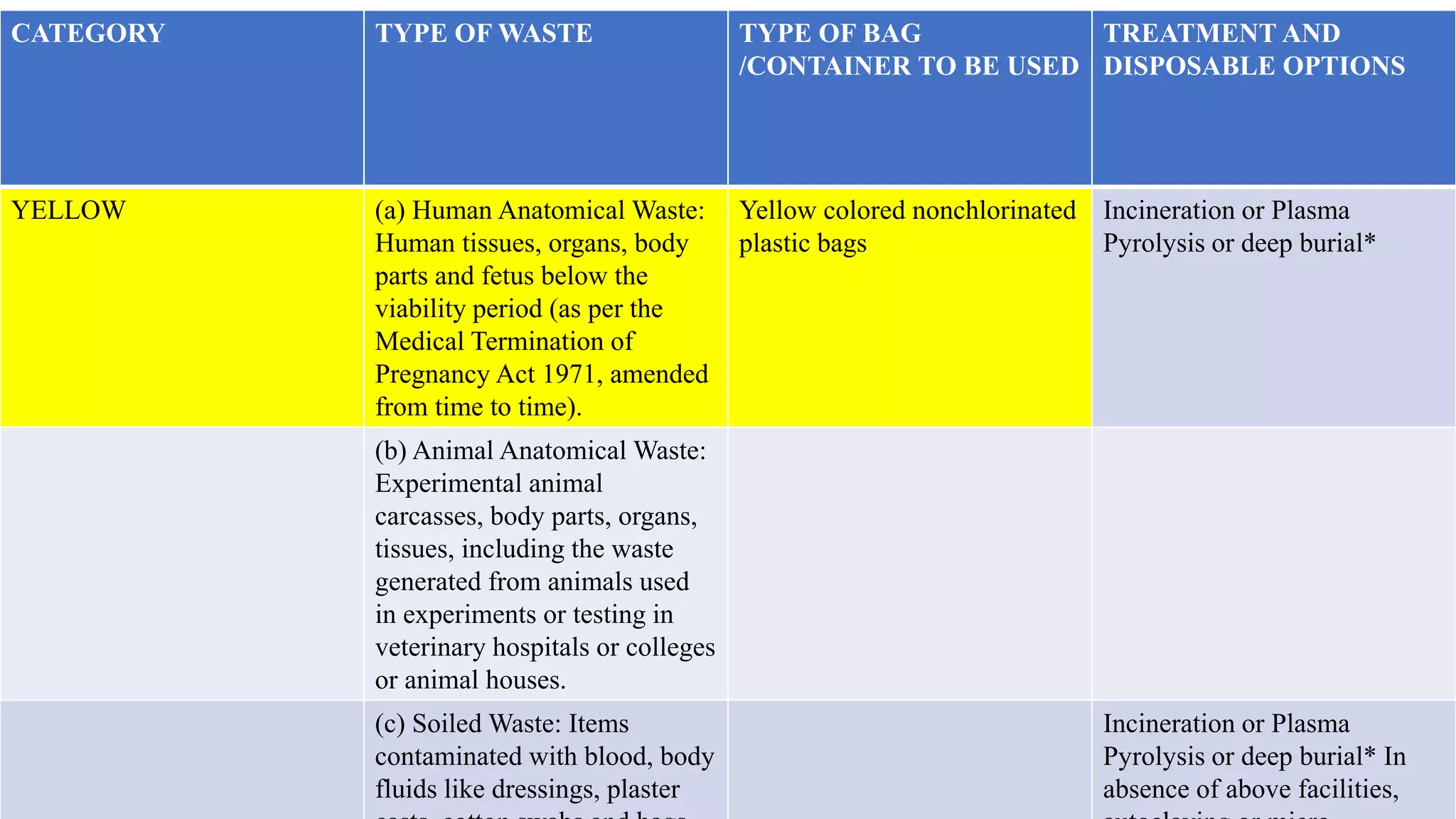
The document discusses guidelines for proper storage, cleaning, and waste management in a labour room. It recommends establishing a Central Sterile Supply Department (CSSD) to ensure sterile supplies are provided from one centralized location. The CSSD should have specific areas for receiving, cleaning, sterilizing, storing, and distributing supplies. It also outlines categories for proper biomedical waste segregation (e.g. yellow, red, white, black bins), treatment and disposal options to safely manage waste and prevent infections.