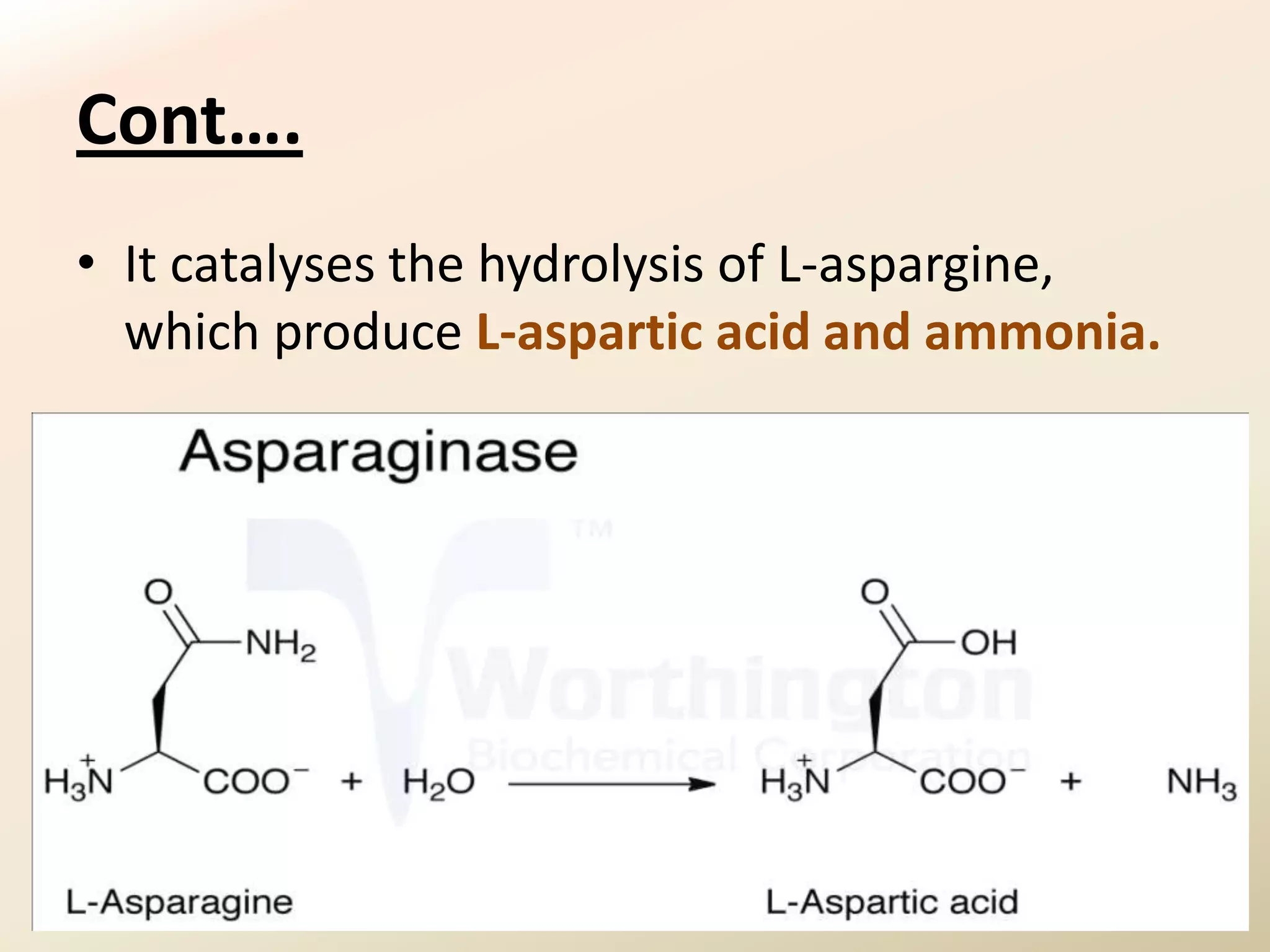
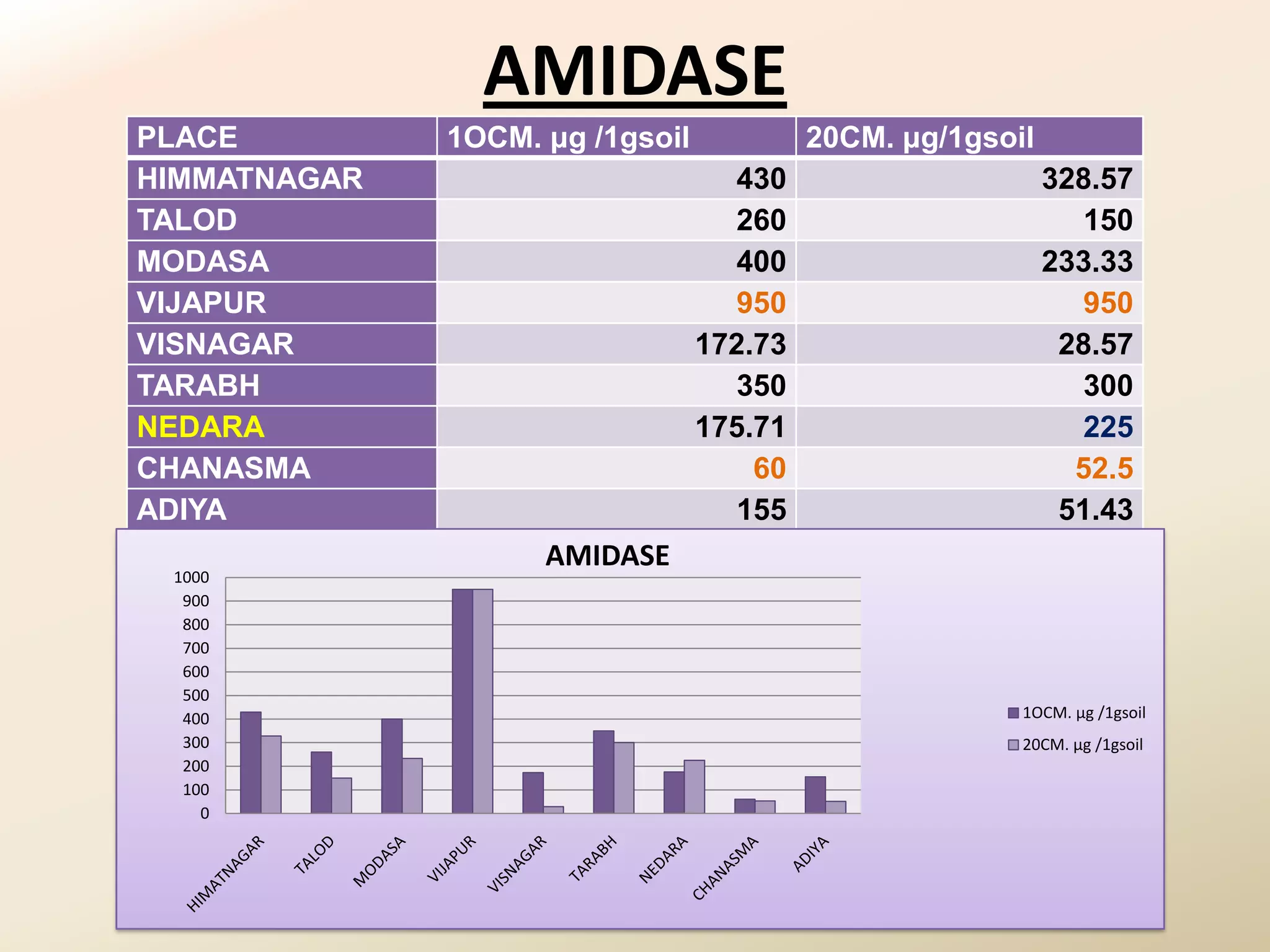
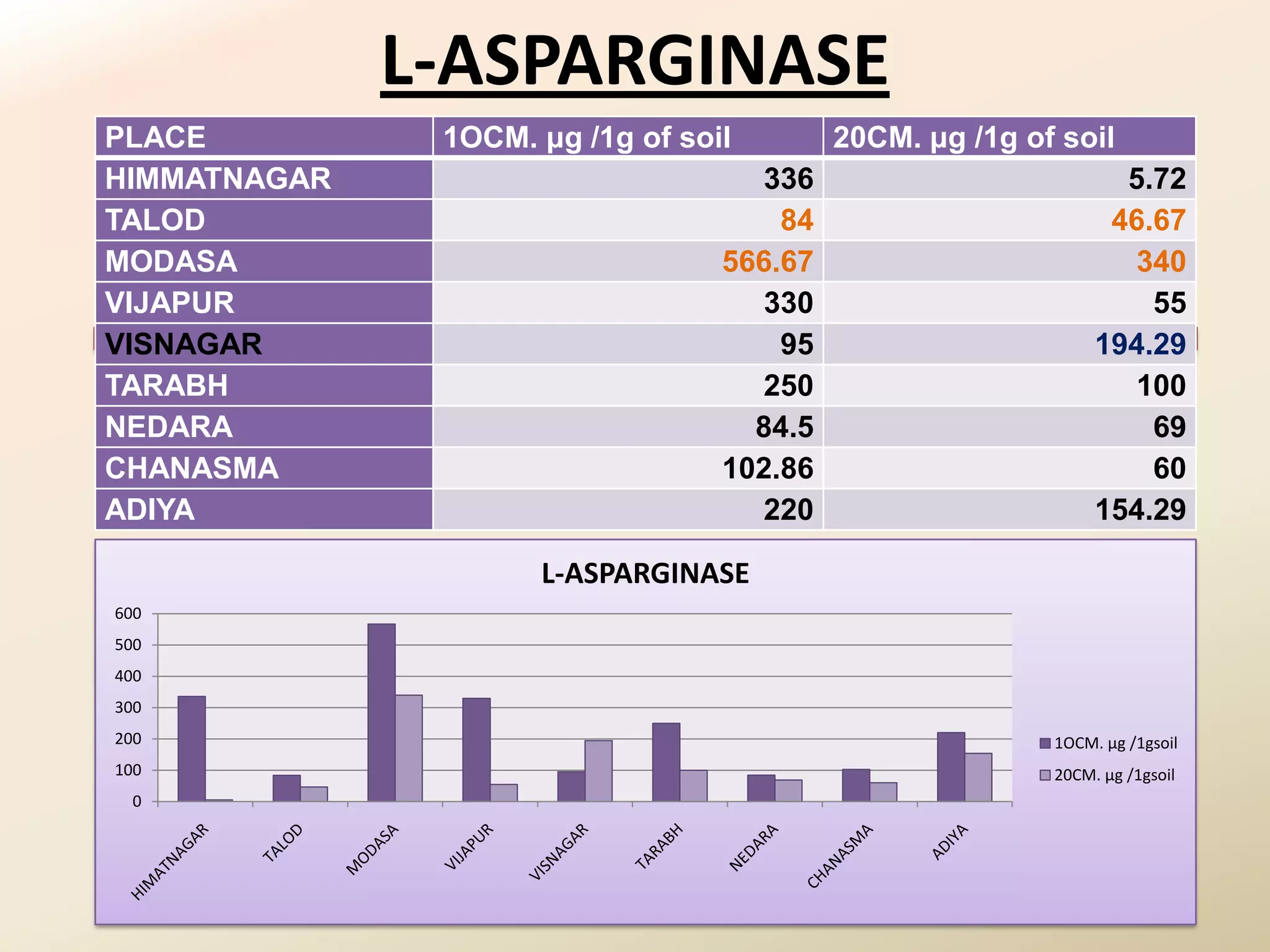
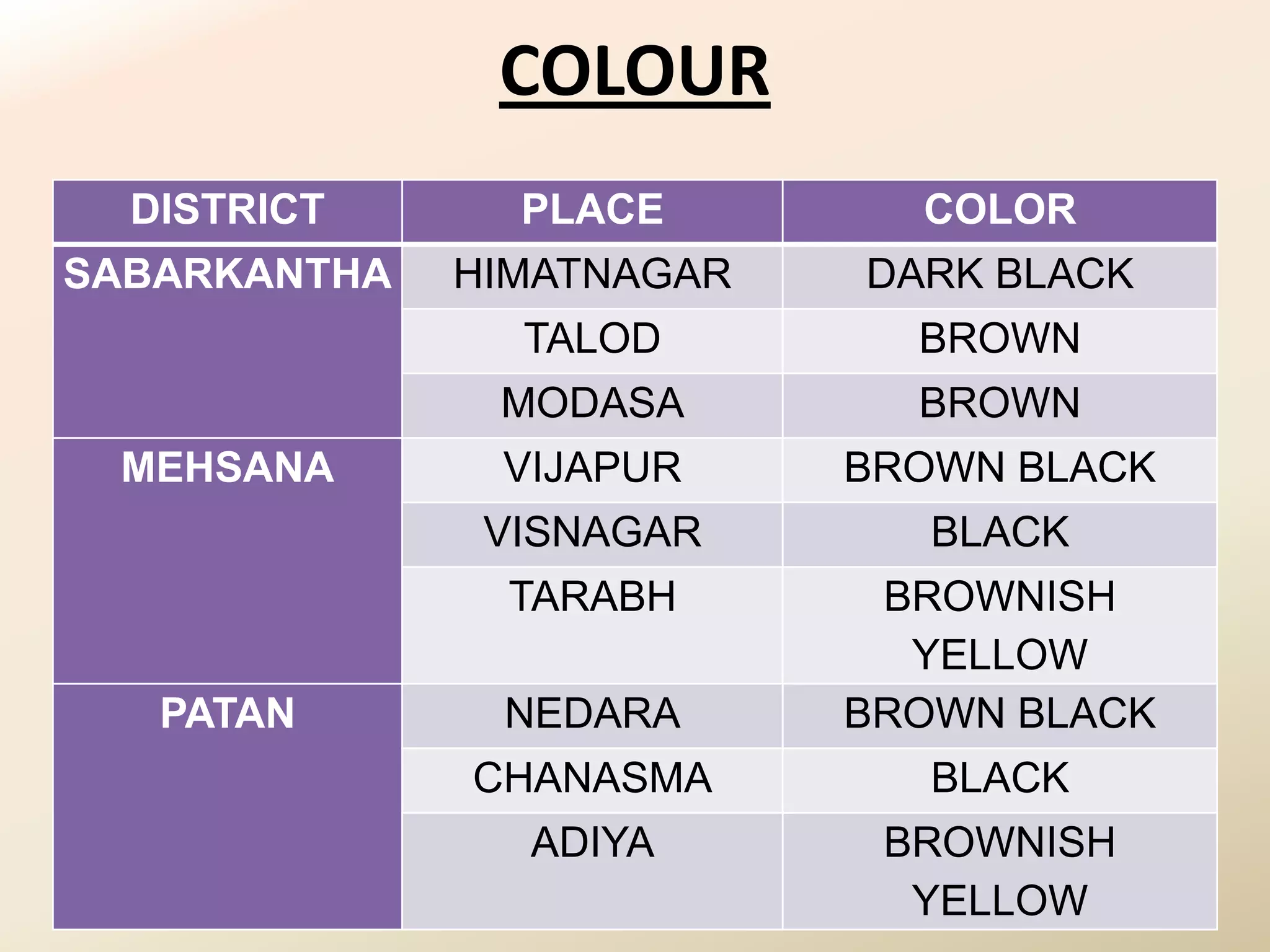
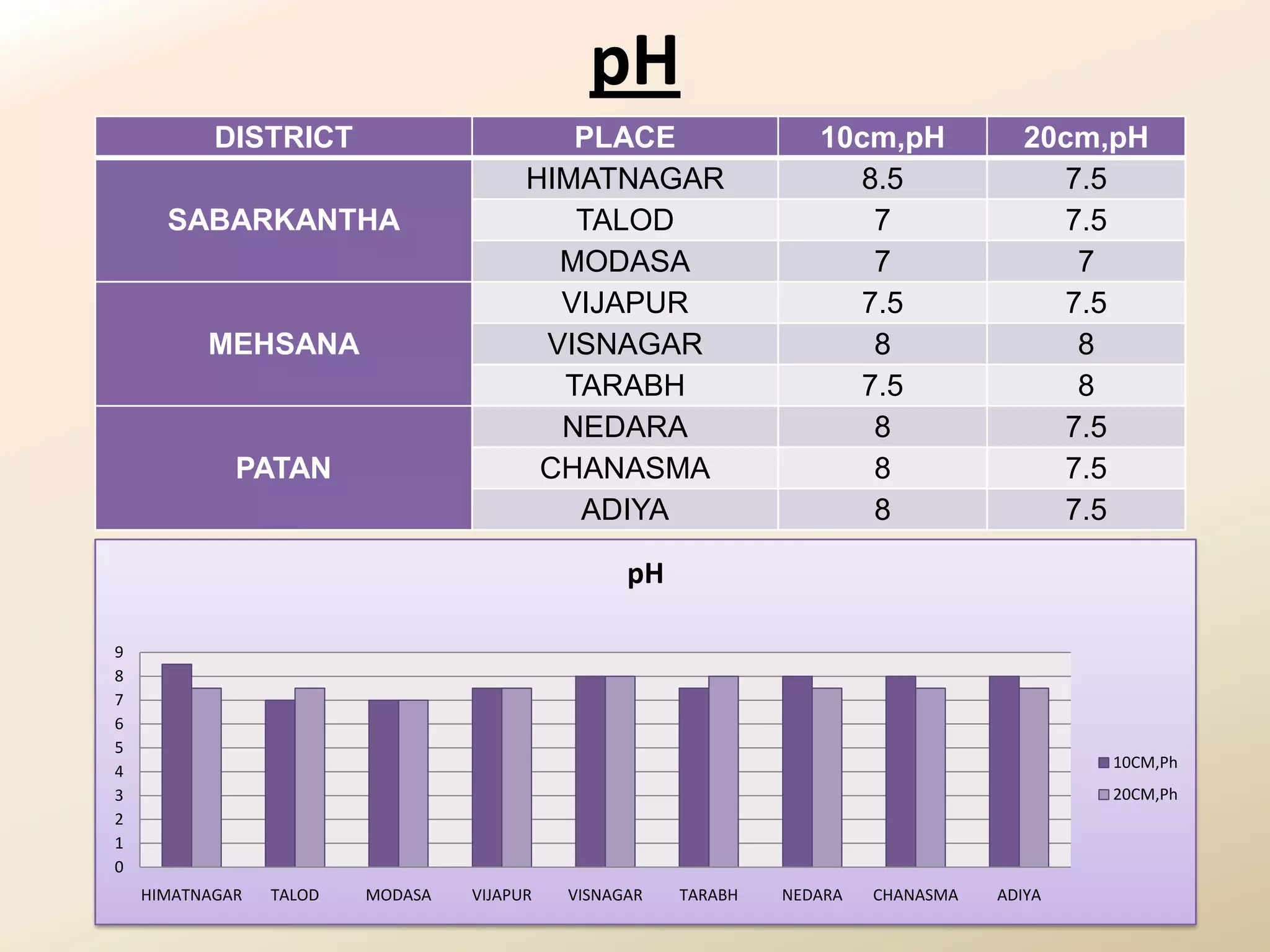
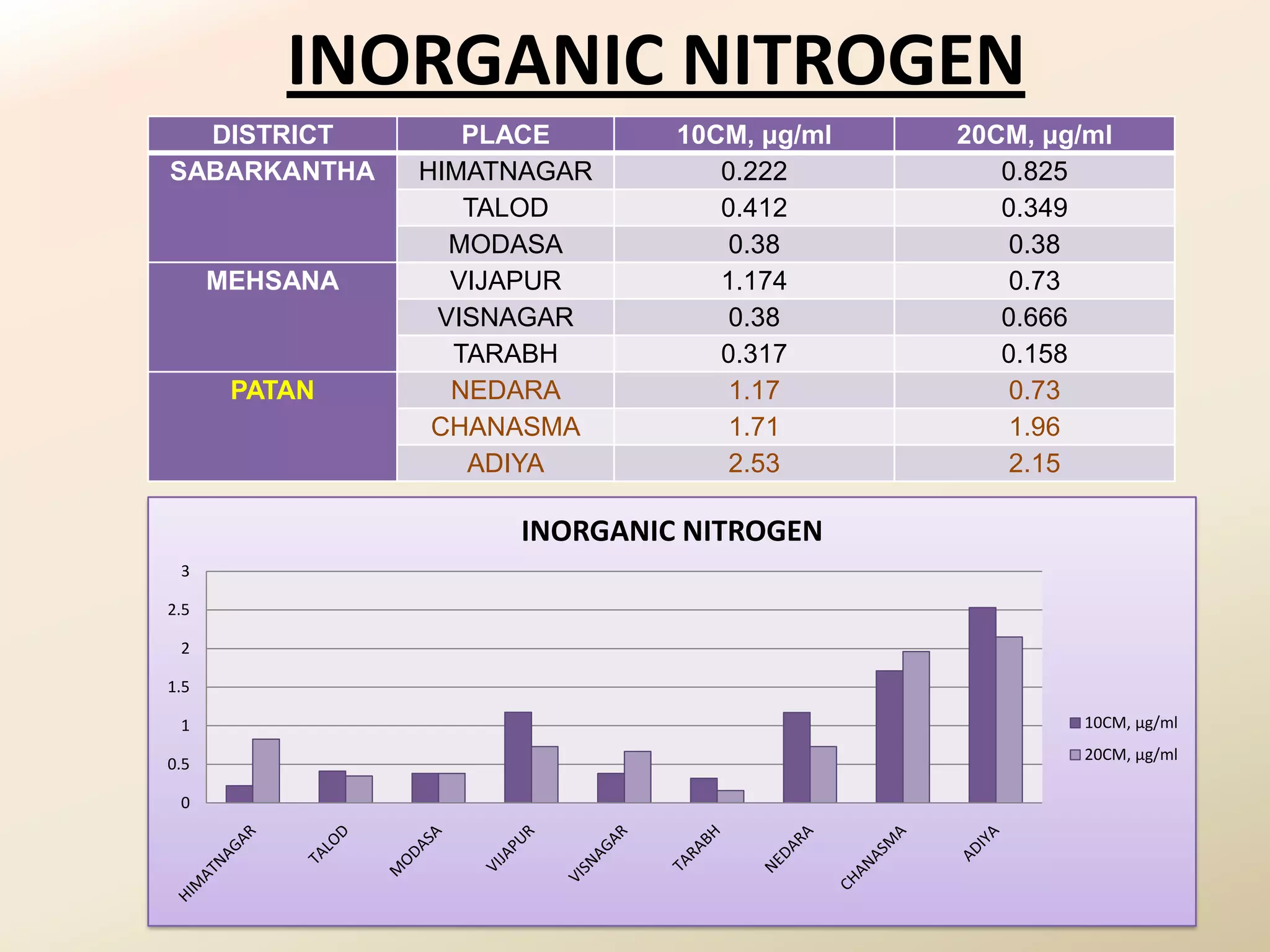
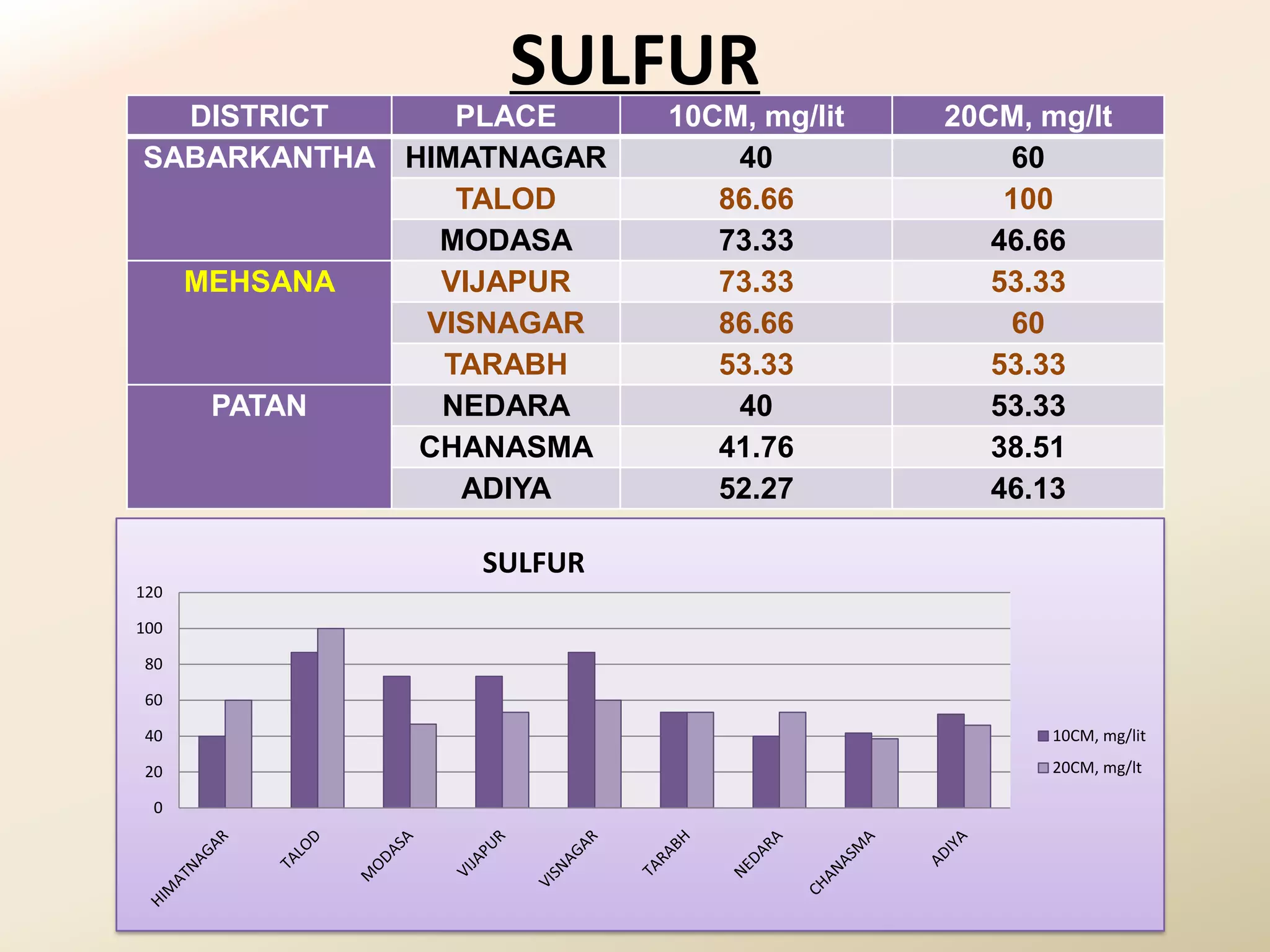
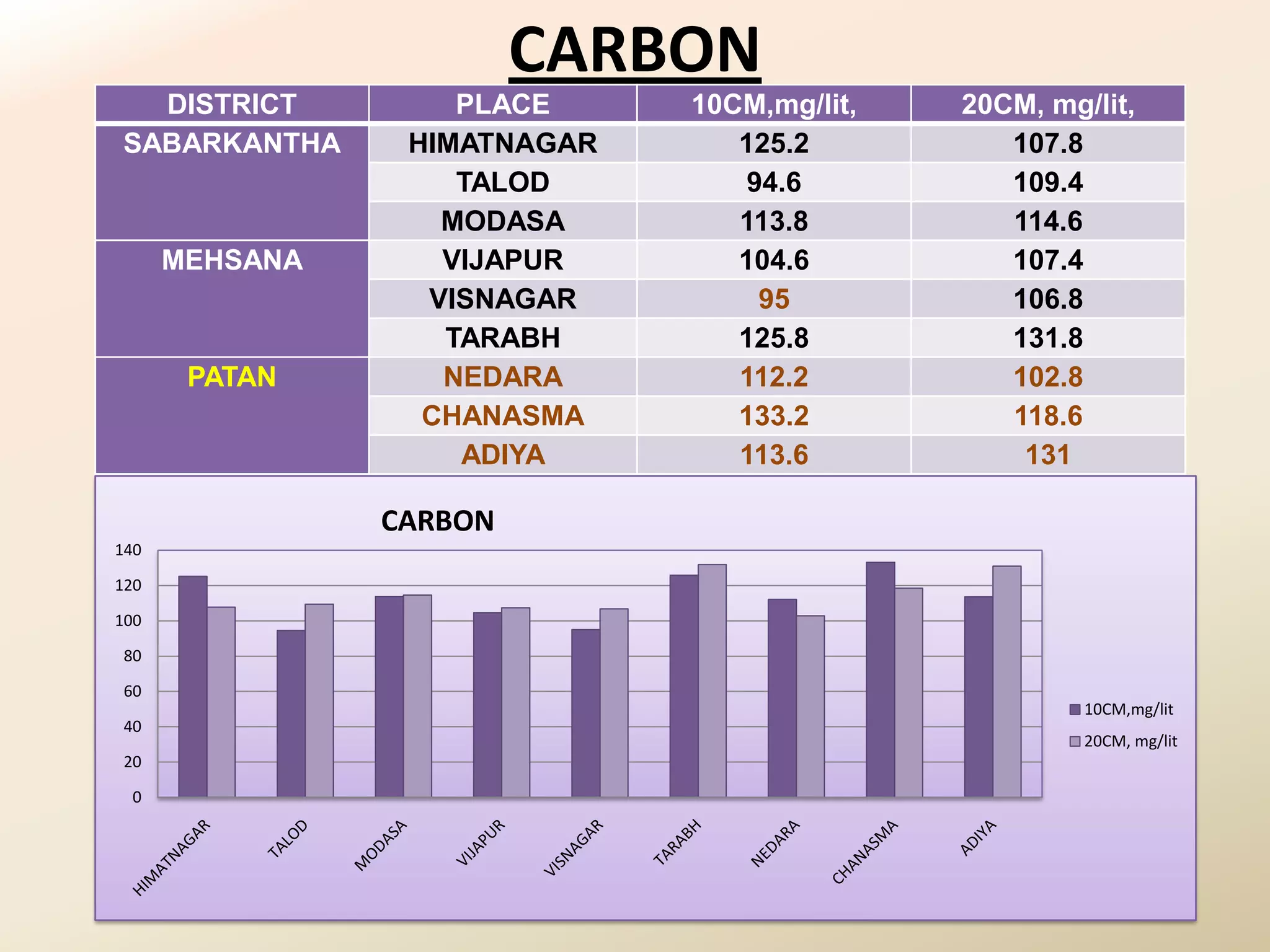
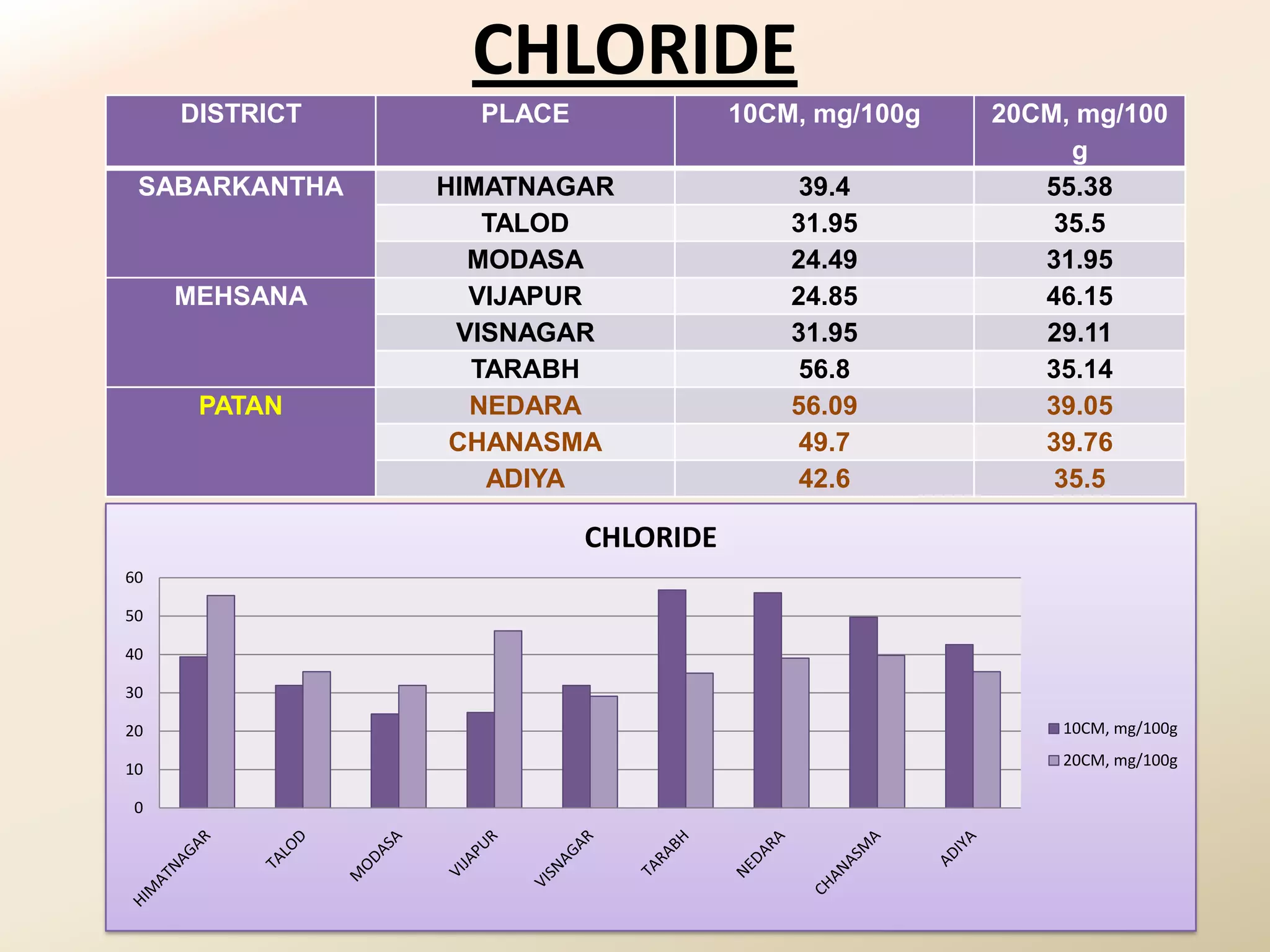
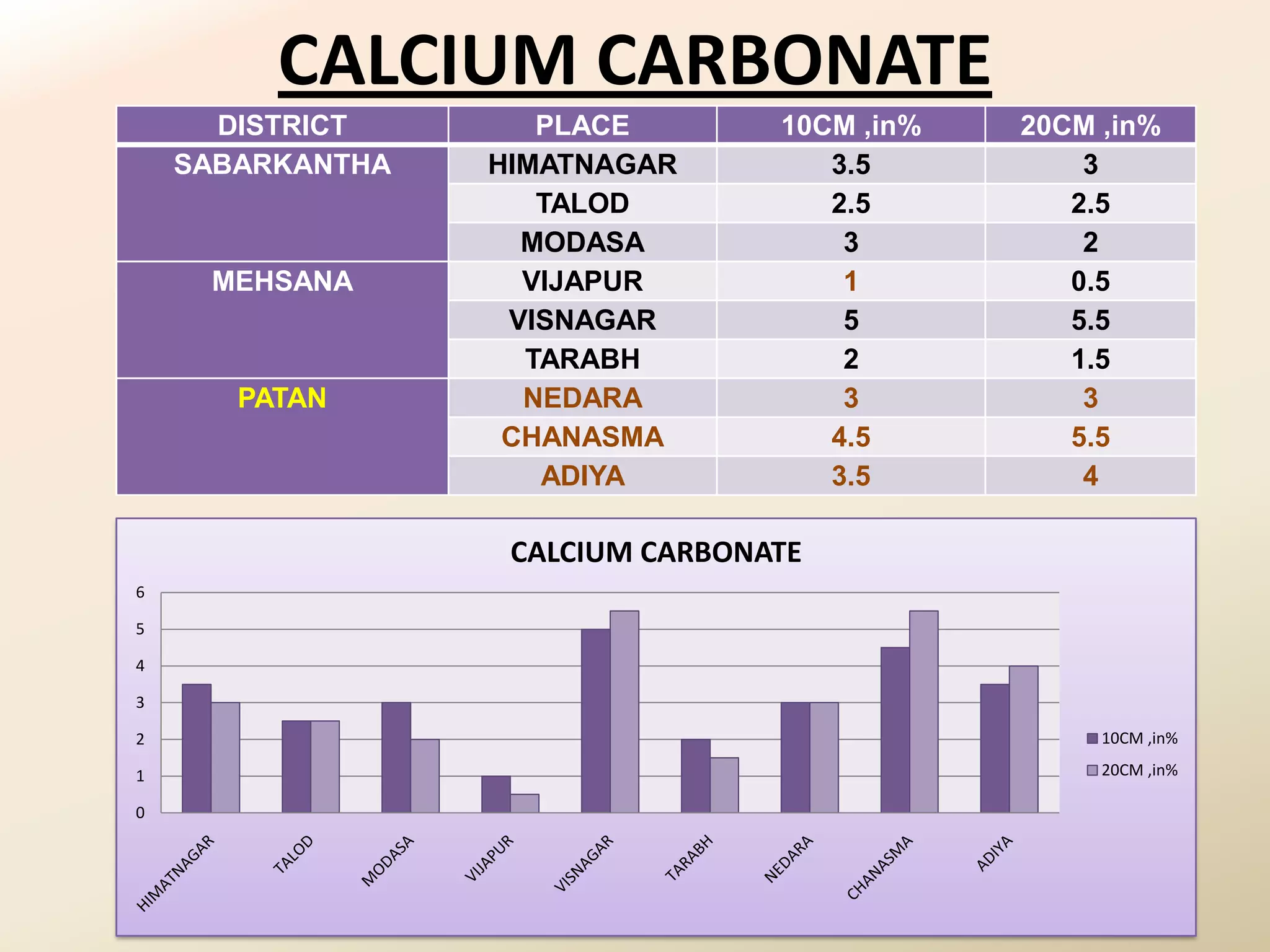
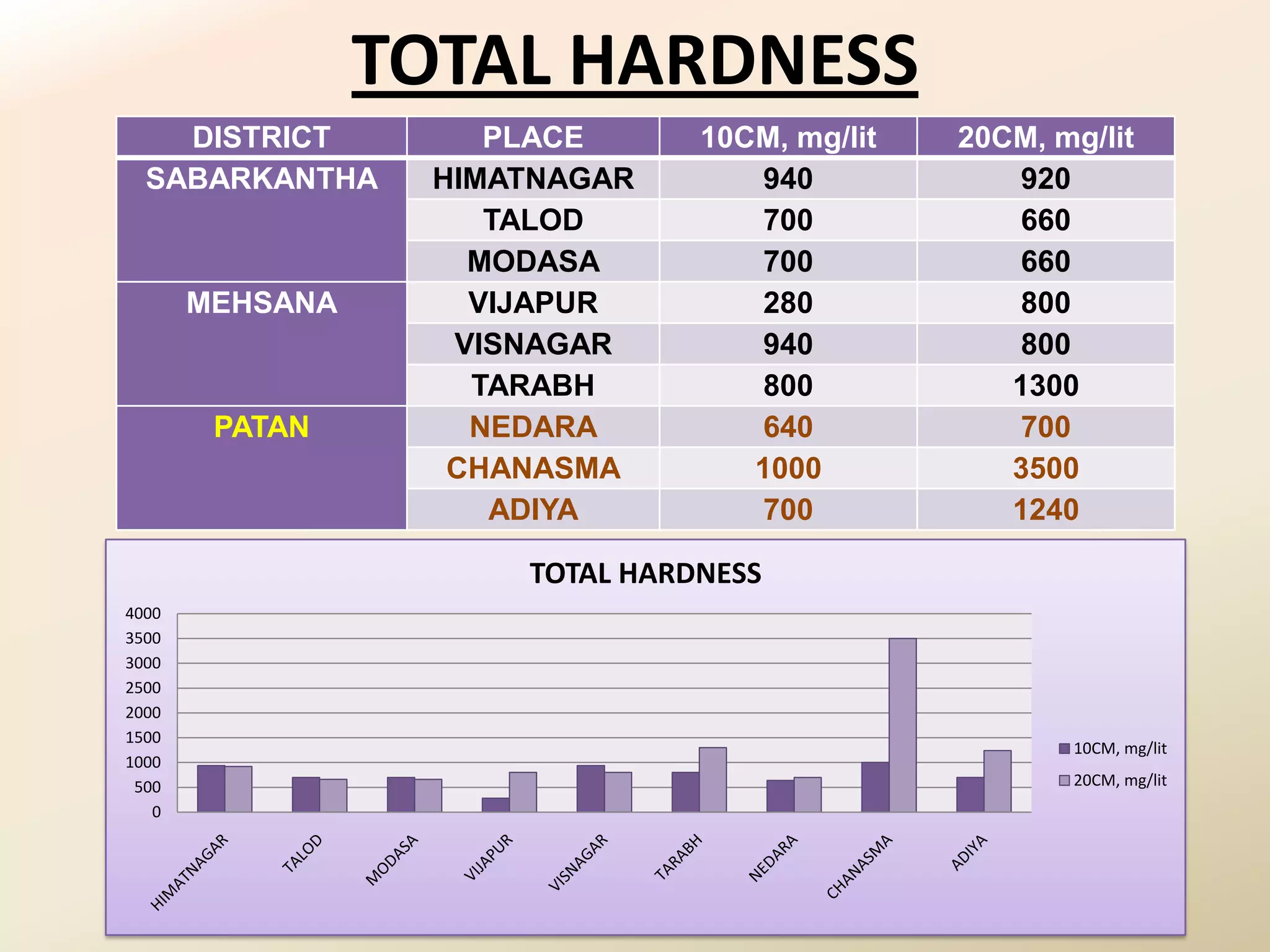
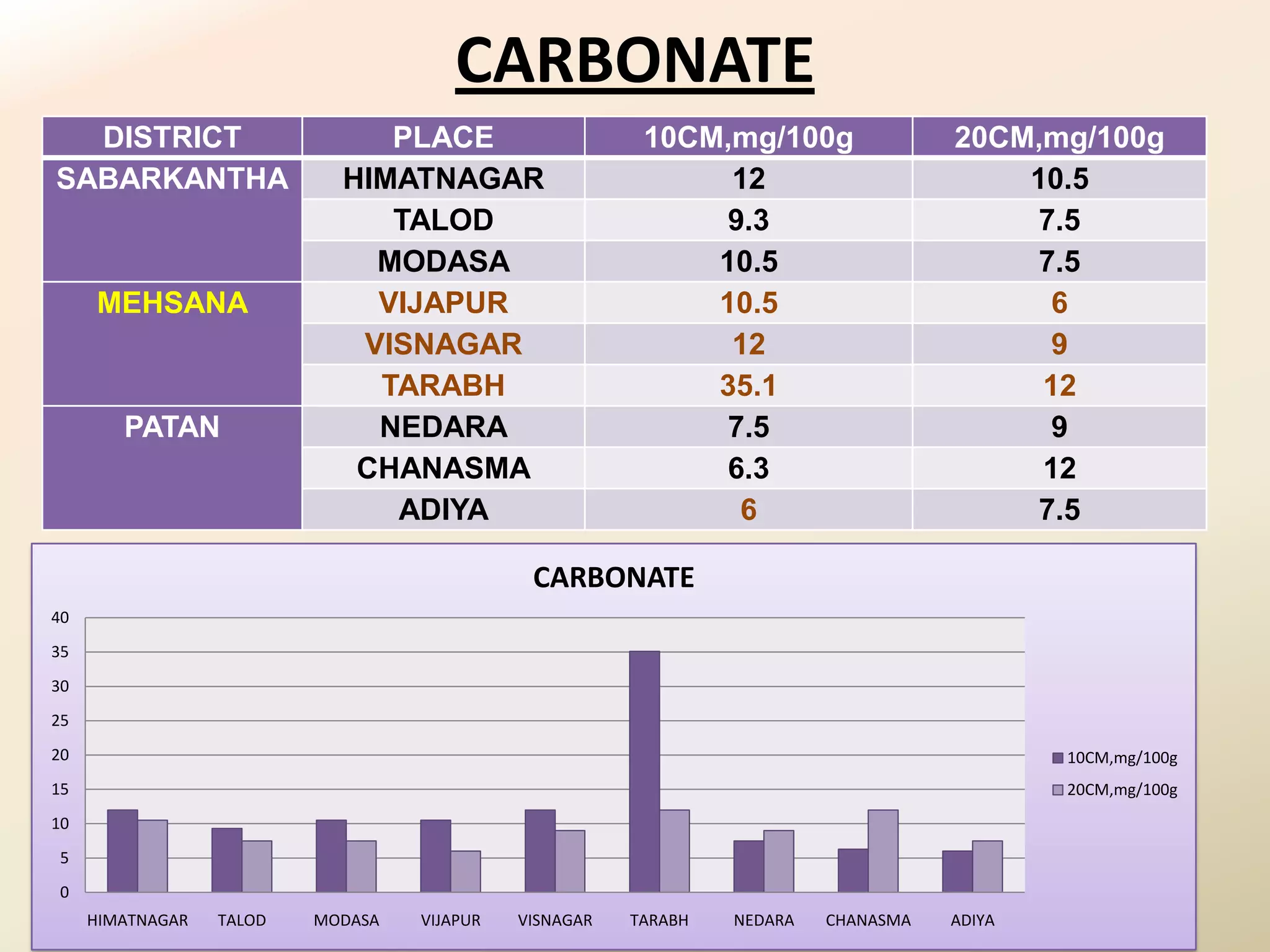
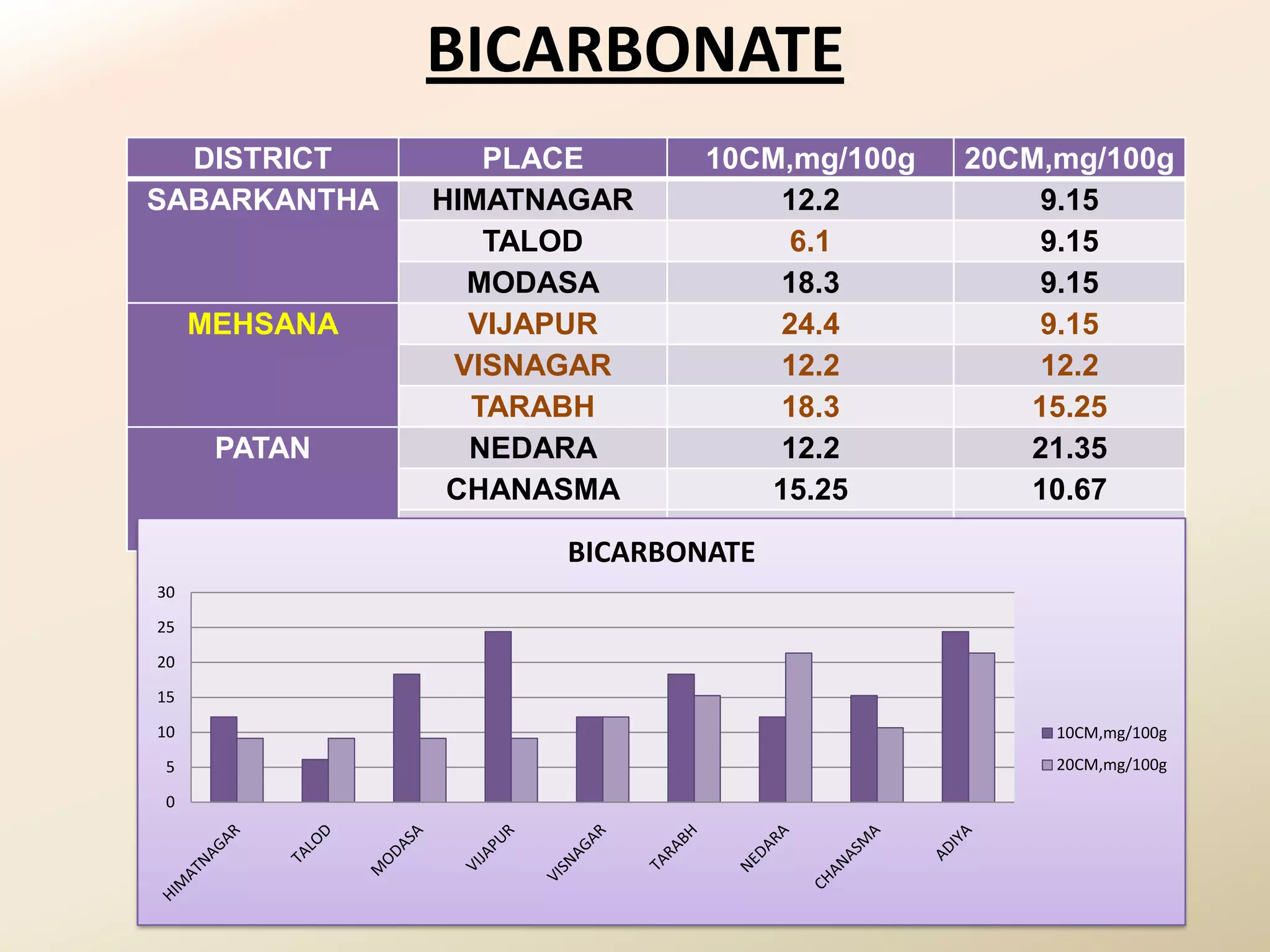
The document summarizes the results of profiling amidohydrolase enzymes and analyzing physicochemical properties of soil samples collected from various locations in North Gujarat, India. Amidohydrolase enzymes like amidase and L-asparaginase were assayed in the soil samples. Various physicochemical parameters of the soil like color, pH, organic carbon, nitrogen, sulfur, chloride, calcium carbonate and total hardness were also analyzed. The results showed variation in enzyme activities and physicochemical properties across different sampling locations.