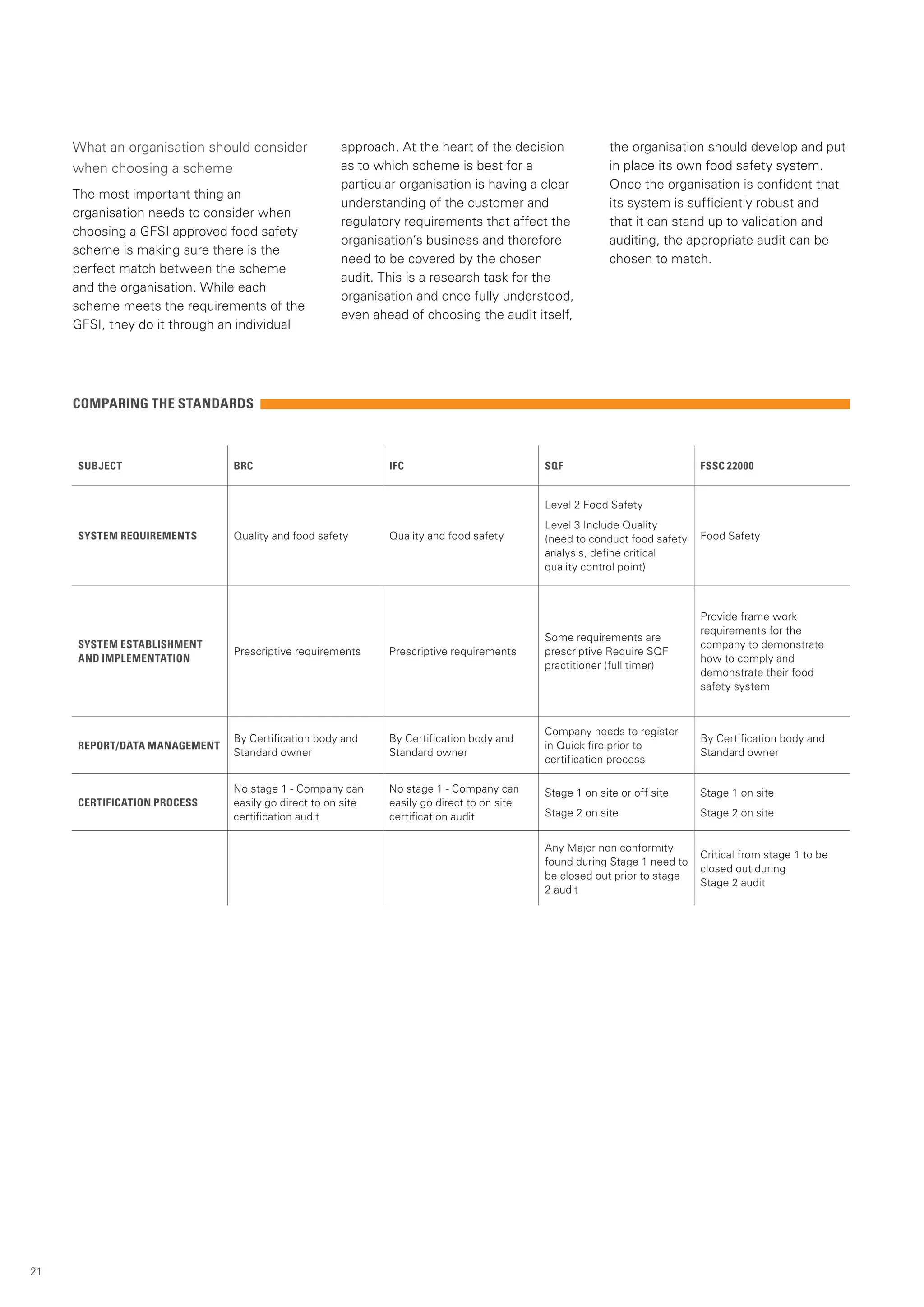
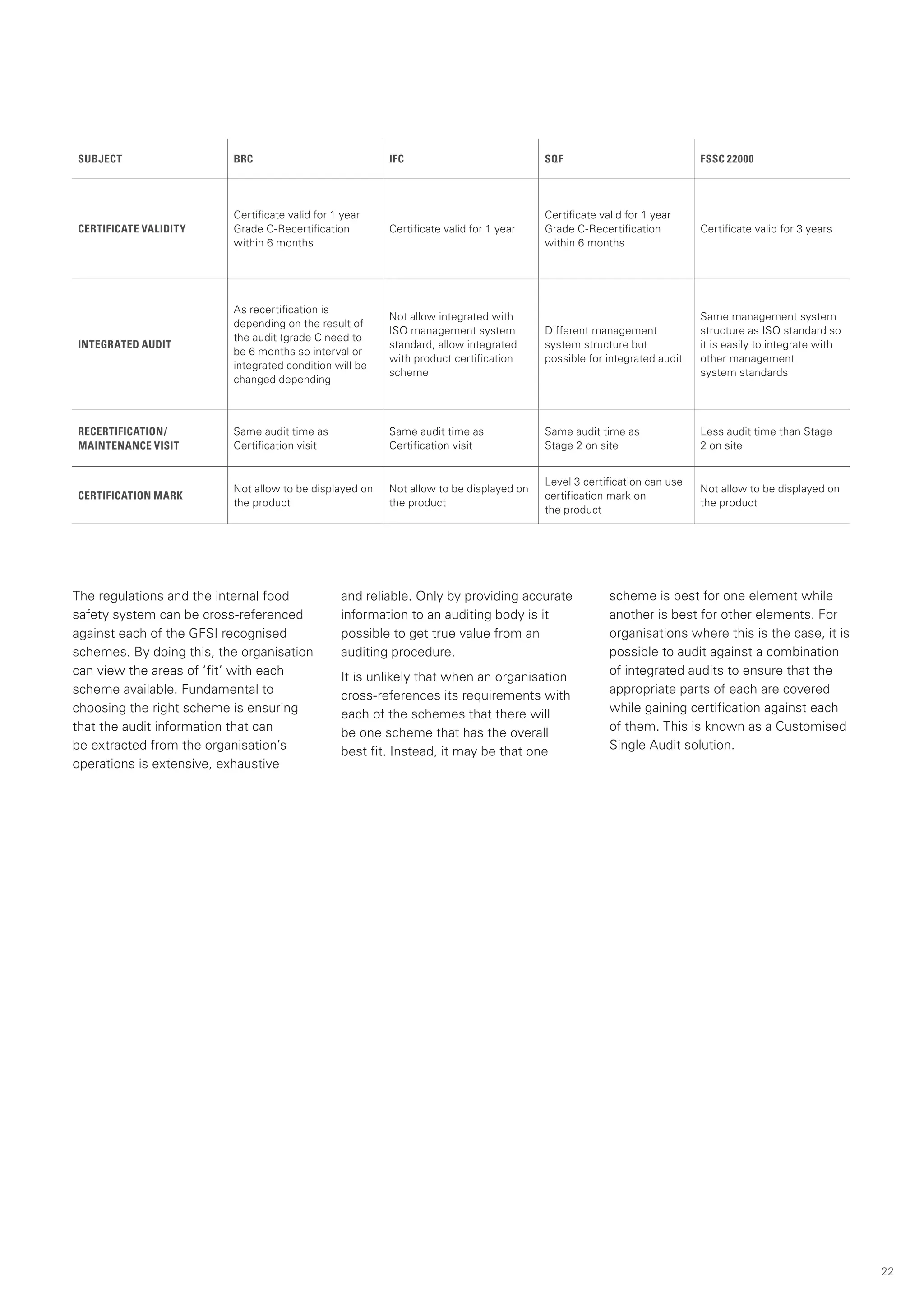
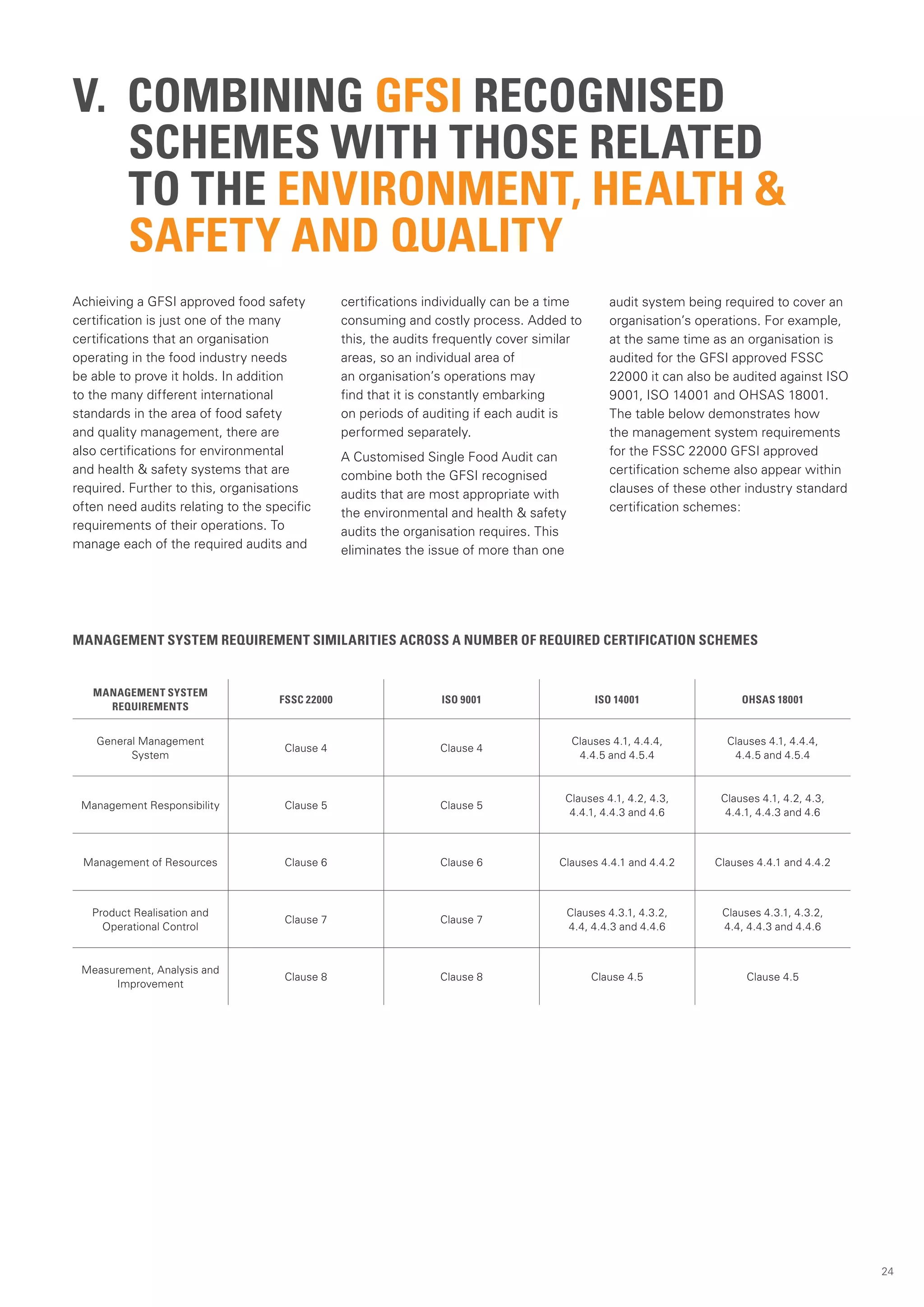
This document provides an overview of the Global Food Safety Initiative (GFSI) and compares various GFSI-recognized food safety standards. The GFSI aims to improve food safety, reduce audits, and increase transparency through a benchmarking process. It recognizes several standards that meet its criteria for ensuring safe food production globally. The document analyzes key standards like BRC, FSSC 22000, IFS Food, SQF 2000, and GLOBALG.A.P., comparing their requirements, certification processes, and suitability for different organizations. It also discusses potential benefits of combining standards to conduct a single audit addressing multiple needs.