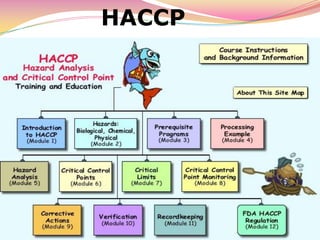
HACCP (Hazard Analysis and Critical Control Points) is a systematic preventative approach to food safety that identifies potential food safety hazards, finds ways of preventing them, and establishes procedures to reduce risks to safe levels. It involves identifying potential hazards at specific points in the food production process, establishing controls to prevent hazards, and monitoring these control points to ensure effectiveness and safety of the food. The Codex Alimentarius Commission establishes international food standards, guidelines and codes of practice to protect consumer health and ensure fair practices. HACCP was developed in the 1960s and has since been recognized internationally as an effective tool for modern, science-based food safety systems.